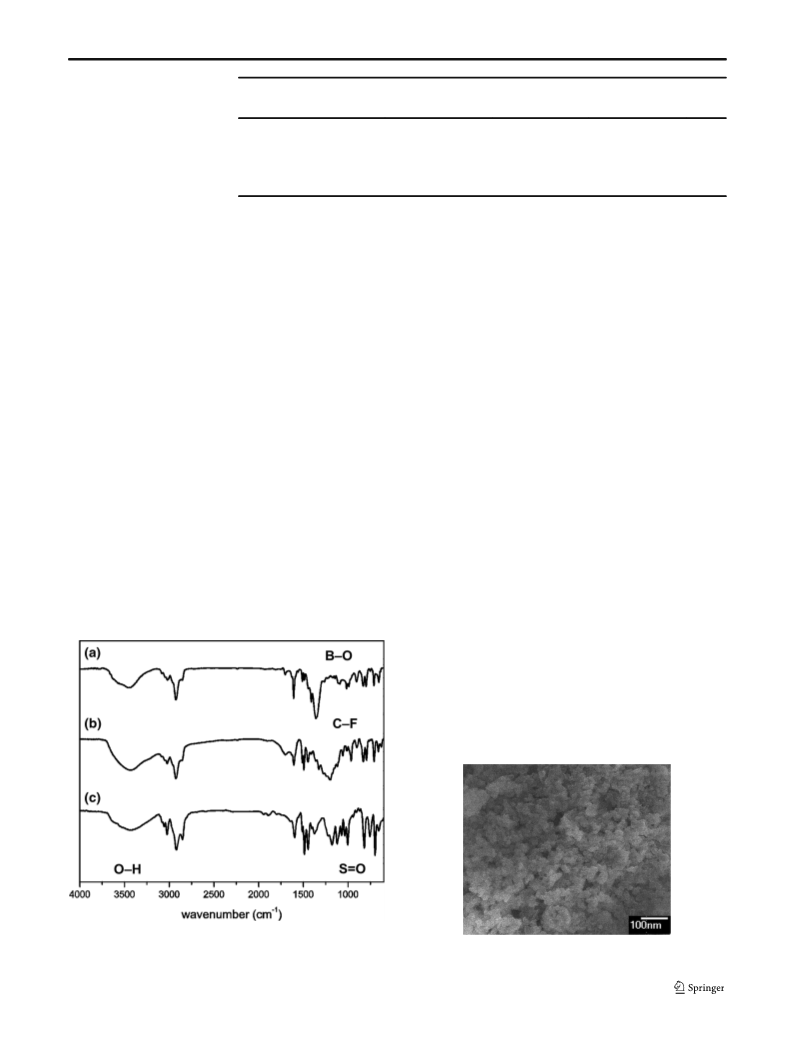
Struct Chem
0
.5 wt% loading of catalyst (Fig. 7). Although the same
The methodology of pre-formed porous polymers using
a porous silica template can offer a convenient synthetic
platform for creation of solid catalysts with a variety of
micro- and meso-porous structures that will be used in
broader catalysis applications.
weight percent of catalysts was employed, A15 has a much
more acidic groups per gram because of its higher loading
capacity (4.8 mmol/g). This value is almost five times higher
than that of SAC1 (1.02 mmol/g). Nafion has a similar molar
loading capacity of 0.90 mmol/g but it does not have a porous
structure. Due to the high acid loading capacity, A15 showed
good conversion throughout the reaction, achieving complete
conversion in 4 h. With the same 0.5 wt% of catalyst loading,
the activity of SAC1 was comparable to that of A15 under the
entire reaction period. This is possibly due to the mesoporous
structure of SAC1 which allows easy access of reactants into
the pores of the catalyst and facilitates mass diffusion of reac-
tants and product. Additionally, the hydrophobic surface of
pores made of polystyrene with a high degree of cross-
linking density would be helpful to achieve good catalytic
performance by repelling water that is formed as a byproduct,
otherwise it may push the reaction back to an equilibrium [22].
In spite of having a similar acidity strength of SAC1, NR50
showed much lower conversion because of its non-porous
structure and low surface area [23].
Acknowledgments The authors appreciate the generous support of the
Rensselaer Polytechnic Institute (Bae startup) for this work.
References
1
.
2. Melero JA, Iglesias J, Morales G (2009) Heterogeneous acid cata-
lysts for biodiesel production: current status and future challenges.
Green Chem 11(9):1285–1308
3
.
Sherrington DC (1998) Preparation, structure and morphology of
polymer supports. Chem. Comm. (21):2275–2286
4. Okay O (2000) Macroporous copolymer networks. Prog Polym Sci
25(6):711–779
5
.
Kirschning A (2004) Immobilized catalysts: solid phases, immobi-
lization and applications, vol 242. Springer Science & Business
Media
Reusability of solid acid catalysts to reduce environmental
impact is an important criteria for practical application. In
reusability study, after first run of esterification with
6
.
.
Svec F, Fréchet JM (1996) New designs of macroporous polymers and
supports: from separation to biocatalysis. Science 273(5272):205
Melero JA, van Grieken R, Morales G (2006) Advances in the synthe-
sis and catalytic applications of organosulfonic-functionalized
mesostructured materials. Chem Rev 106(9):3790–3812
7
0
.5 wt% of SAC1, the solid catalyst was filtered, washed
and dried, and used for the next cycle of reaction without
any additional treatment. The recycled catalyst showed almost
the same level of catalytic activity and achieved full conver-
sion within 6 h up to 3 cycles, demonstrating reusability of the
catalyst in acid-catalyzed organic reaction. After the third cy-
cle, the catalyst showed a slight decrease in activity.
8
.
.
Ishihara K, Hasegawa A, Yamamoto H (2001) Polystyrene-Bound
Tetrafluorophenylbis (triflyl) methane as an Organic-Solvent-
Swellable and Strong Brønsted Acid Catalyst. Angew Chem
113(21):4201–4203
9
Yin Y, Zhao G, Li G-L (2005) Synthesis of polystyrene-bound
perfluoroalkyl sulfonic acids and the application of their ytter-
bium salts in multicomponent reactions (MCRs). Tetrahedron
6
1(51):12042–12052
1
0. Olah GA, Prakash GS, Sommer J (1979) Acids up to billions of
times stronger than sulfuric acid have opened up fascinating new
areas of chemistry. Chemistry 1:3
Conclusion
Two solid acid catalysts with well-defined porosity and
different acid strength were synthesized, and their catalyt-
ic activities in esterification were compared with commer-
cially available solid acid catalysts. The use of borylated
precursor polymer allowed convenient preparation of sol-
id acid catalysts with different acid strength. The solid
acid catalyst with perfluoroalkyl sulfonate (SAC1)
showed a greater activity compared to the solid acid cat-
alyst with less acidic aryl sulfonate (SAC2) and the dif-
ference in activity becomes wider as decreasing tempera-
ture. Despite high degree of cross-linking and lower acid
loading, the mesoporosity and superacidity of SAC1
afforded good conversion rate of esterification which is
comparable to that of commercially available Amberlyst
11. Chang Y, Brunello GF, Fuller J, Hawley M, Kim YS, Disabb-Miller
M, Hickner MA, Jang SS, Bae C (2011) Aromatic ionomers with
highly acidic sulfonate groups: acidity, hydration, and proton con-
ductivity. Macromolecules 44(21):8458–8469
1
2. Shin J, Jensen SM, Ju J, Lee S, Xue Z, Noh SK, Bae C (2007)
Controlled functionalization of crystalline polystyrenes via activa-
tion of aromatic CH bonds. Macromolecules 40(24):8600–8608
3. Miyaura N, Suzuki A (1995) Palladium-catalyzed cross-coupling
reactions of organoboron compounds. Chem Rev 95(7):2457–2483
4. Popeney CS, Levins CM, Guan Z (2011) Systematic Investigation
of Ligand Substitution Effects in Cyclophane-Based Nickel (II) and
Palladium (II) Olefin Polymerization Catalysts (1). Organometallics
1
1
3
0(8):2432–2452
1
5. Zhang Y, Zhao L, Patra PK, Ying JY (2008) Synthesis and catalytic
applications of mesoporous polymer colloids in olefin
hydrosilylation. Advanced Synthesis & Catalysis 350(5):662–666
16. Kim J-H, Park EJ, Lim D-K, Singh B, Bae C, Song S-J (2015)
Fabrication of Dense Cerium Pyrophosphate-Polystyrene
Composite for Application as Low-Temperature Proton-
Conducting Electrolytes. J Electrochem Soc 162(10):F1159–F1164
1
5 with higher acid loading. After the reaction, catalyst
was easily recovered and recycled up to three times with-
out additional treatment, demonstrating a potential for use
as a recyclable solid acid catalyst in organic reactions.
1
7. Miguel, Yd, Rohr T, Sherrington DC (2005) Structure,
Morphology, Physical Formats and Characterization of Polymer

 Park, Eun Joo
Park, Eun Joo
 Bae, Chulsung
Bae, Chulsung