Please do not adjust margins
Chemical Science
Page 7 of 7
DOI: 10.1039/C6SC00932H
Edge Article
Chemical Science
Okumura and K. Kawata, J. Am. Chem. Soc., 1968, 90, 1650- 22 (a) G. Valero and A. Moyano, In Comprehensive
1
1
651; (h) S. Hiari, K. Kawata and W. Nagata, Chem. Commun.,
968, 1016-1017; (i) T. Imanishi, N. Yagi, H. Shin and M.
Enantioselective Organocatalysis: Catalysts, Reactions, and
Applications, (Eds.: P. I. Dalko), Wiley-VCH Verlag GmbH & Co.
KGaA, 2013, pp. 1191-1224; (b) A. McNally, B. Evans and M. J.
Gaunt, Angew. Chem., Int. Ed., 2006, 45, 2116-2119.
Hanaoka, Tetrahedron Lett., 1981, 22, 4001-4004; (j) T.
Imanishi and N. Yagi, M. Hanaoka, Chem. Pharm. Bull., 1985,
3
3, 4202-4211; (k) G. R. Krow, D. A. Shaw, B. Lynch, W. Lester, 23 (a) H. G. Lindwall and J. S. Maclennan, J. Am. Chem. Soc., 1932,
S. W. Szczepanski, K. Raghavachari and A. E. Derome, J. Org.
Chem., 1988, 53, 2258-2262; (l) C. Herdeis and C. Hartke-
Karger, Liebigs Ann. Chem., 1991, 99-104; (m) P. Rosenmund,
W. H. Haase, J. Bauer and R. Frische, Chem. Ber., 1975, 108,
54, 4739-4744; (b) S. Mukherjee, J. W. Yang, S. Hoffmann and
B. List, Chem. Rev., 2007, 107, 5471-5569 and references cited
therein; (c) D. W. C. MacMillan, Nature, 2008, 455, 304-308
and references cited therein.
1
6
1
871-1895; (n) S. I. Sallay, J. Am. Chem. Soc., 1967, 89, 6762- 24 A proposed mechanism for 16-catalyzed formation of 6a:
763; (o) M. Ikezaki, T. Wakamastu and Y. Ban, J. Chem. Soc. D,
969, 88-89; (p) K. J. Henrey, P. A. Grieco, Jr. and W. J. DuBay,
Tetrahedron Lett., 1996, 37, 8289-8292; (q) W. G. Bornmann
and M. E. Kuhene, J. Org. Chem., 1992, 57, 1752-1760; (r) A. C.
Kruegel, S. Rakshit, X. Li and D. Sames, J. Org. Chem., 2015, 80,
2
062-2071.
B. M. Trost, S. A. Godleski and J. P. Genêt, J. Am. Chem. Soc.,
978, 100, 3930-3931.
(a) D. M. Hodgson and J.-M. Galano, Org. Lett., 2005, 7, 2221-
1
1
1
2
1
2
8
224; (b) S. Höck and H.-J. Borschberg, Helv. Chim. Acta., 2006,
9, 542-557; (c) L. Moisan, P. Thuéry, M. Nicolas, E. Doris and
B. Rousseau, Angew. Chem., Int. Ed., 2006, 45, 5334-5336; (d)
M. Harada, K. N. Asaba, M. Iwai, N. Kogure, M. Kitajima and H.
Takayama, Org. Lett., 2012, 14, 5800-5803; (e) K. Ishihara, H.
o
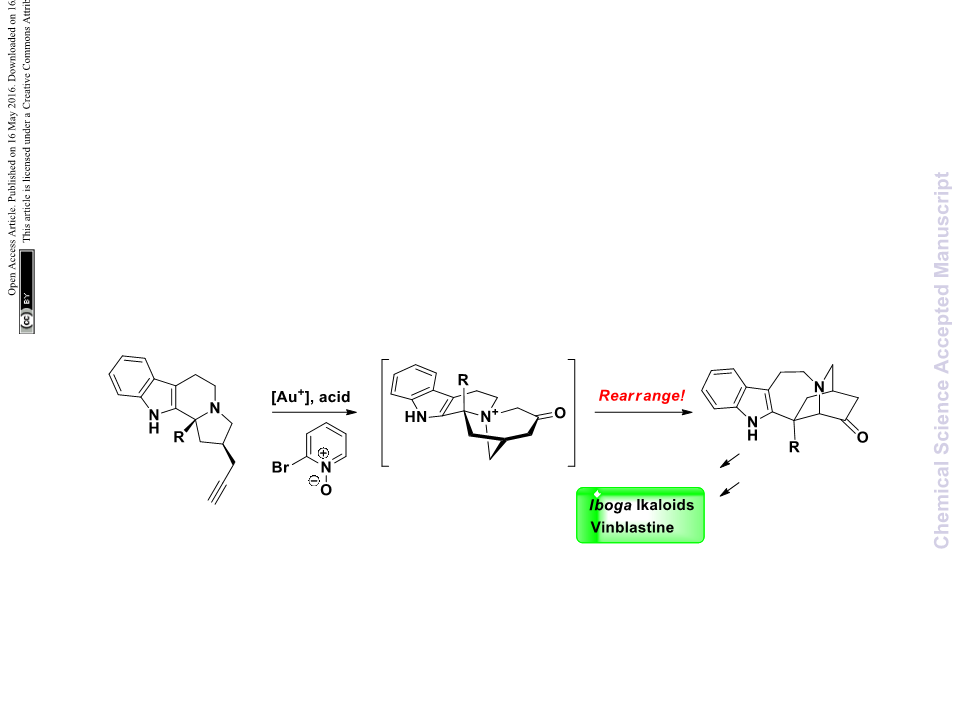
When 8a and 16 were heated at 80 C in HFIP, the formation
of enamine intermediate A was supported by LCMS analysis.
Yamada and M. Akakura, Chem. Commun., 2014, 50, 6357- 25 For a recent review, see: G. B. Dudley, R. Richert and A. E.
6
360; (f) M. Hatano, Y. Goto, A. Izumiseki, M. Akakura and K.
Stiegman, Chem. Sci., 2015, 6, 2144-2152.
Ishihara, J. Am. Chem. Soc., 2015, 137, 13472-13475.
(a) J. D. White and Y. Choi, Org. Lett., 2000, 2, 2373-2376; (b)
H. Mizoguchi, H. Oikawa and H. Oguri, Nat. Chem., 2014, 6,
26 The synthesis of racemic epiibogamine has been reported.
See: (a) G. Büchi, D. L. Coffen, K. Kocsis, P. E. Sonnet and F. E.
Ziegler, J. Am. Chem. Soc., 1966, 88, 3099-3109; (b) G. K. Jana
and S. Sinha, Tetrahedron Lett., 2012, 53, 1671-1674.
1
3
5
7-64.
1
1
4
5
J. W. Beatty and C. R. J. Stephenson, J. Am. Chem. Soc., 2014, 27 (a) K. Iwasaki, K. K. Wan, A. Oppedisano, S. W. M. Crossley and
R. A. Shenvi, J. Am. Chem. Soc., 2014, 136, 1300-1303; b) S. M.
1
36, 10270-10273 and references cited therein.
King, X. Ma and S. B. Herzon, J. Am. Chem. Soc., 2014, 136,
884-6887.
(a) J. C. Lo, Y. Yabe and P. S. Baran, J. Am. Chem. Soc., 2014,
36, 1304-1307; (b) J. C. Lo, J. Gui, Y. Yabe, C.-M. Pan and P. S.
Baran, Nature, 2014, 516, 343-348.
O. H. Oldenziel, D. Van Leusen and A. M. Van Leusen, J Org
Chem, 1977, 42, 3114-3118.
(a) T. S. Stevens, E. M. Creighton, A. B. Gordon and M.
MacNicol, J. Chem. Soc., 1928, 3193-3197; (b) I. E. Markó in
Comprehensive Organic Synthesis, Vol. 3 (Eds.: B. M. Trost, I.
Fleming), Pergamon, Oxford, 1991, pp. 913-974; (c) M. G.
Charest, C. D. Lerner, J. D. Brubaker, D. R. Siegel and A. G.
Myers, Science, 2005, 308, 395-398; (d) J. A. Vanecko, H. Wan
and F. G. West, Tetrahedron, 2006, 62, 1043-1062; (e) P.
Tuzina and P. Somfai, Org. Lett., 2009, 11, 919-921; (f) T. M.
Bott, J. A. Vanecko and F. G. West, J. Org. Chem., 2009, 74,
6
2
8
1
2
3
9
0
CCDC-1433182 (19), CCDC-1433188 (20) and CCDC-1433180
(
S4) contain the supplementary crystallographic data for this
paper. These data can be obtained free of charge from The
Cambridge Crystallographic Data Centre via
www.ccdc.cam.ac.uk/data_request/cif.
2
832-2836; (g) M. Valpuesta, M. Ariza, A. Díaz and R. Suau,
Eur. J. Org. Chem., 2010, 4393-4401; (h) L. Palombi, Catal.
Commun., 2011, 12, 485-488; (i) J. Clayden, M. Donnard, J.
Lefranc. and D. J. Tetlow, Chem. Commun., 2011, 47, 4624-
3
3
1
2
M. Ito, C. W. Clark, M. Mortimore, J. B. Goh and S. F. Martin, J.
Am. Chem. Soc., 2001, 123, 8003-8010.
a) H. Ishikawa, D. A. Colby, S. Seto, P. Va, A. Tam, H. Kakei, T. J.
Rayl, I. Hwang and D. L. Boger, J. Am. Chem. Soc., 2009, 131,
4
639; (j) S. Harada, M. Kono, T. Nozaki, Y. Menjo, T. Nemoto
and Y. Hamada, J. Org. Chem., 2015, 80, 10317-10333.
(a) W. He, L. Xie, Y. Xu, J. Xiang and L. Zhang, Org. Biomol.
Chem., 2012, 10, 3168-3171; (b) E. L. Noey, Y. Luo, L. Zhang
and K. N. Houk, J. Am. Chem. Soc., 2012, 134, 1078-1084.
I. T. Raheem, P. S. Thiara, E. A. Peterson and E. N. Jacobsen, J.
Am. Chem. Soc., 2007, 129, 13404-13405.
(a) P. Magnus, M. Ladlow, C. S. Kim and P. Boniface,
Heterocycles, 1989, 28, 951-956; (b) P. Magnus, J. S. Mendoza,
A. Stamford, M. Ladlow and P. Willis, J. Am. Chem. Soc., 1992,
1
6
4904-4916. b) E. Giovanelli, S. Leroux, L. Moisan, H. Carreyre,
P. Thuéry, D.-A. Buisson, A. Meddour, J.-M. Coustard, S.
Thibaudeau, B. Rousseau, M. Nicolas, P. Hellier and E. Doris,
Org. Lett., 2011, 13, 4116-4119.
1
1
7
8
3
3
3
3
4
5
L. Zhu, C. Ni, Y. Zhao and J. Hu, Tetrahedron, 2010, 66, 5089-
5100.
T. Miyazaki, S. Yokoshima, S. Simizu, H. Osada, H. Tokuyama
and T. Fukuyama, Org. Lett., 2007, 9, 4737-4740.
(a) B. Gigant, C. Wang, R. B. G. Ravelli, F. Roussi, M. O.
Steinmetz, P. A. Curmi, A. Sobel and M. Knossow, Nature,
1
14, 10232-10245.
1
2
2
9
0
1
(a) P. de Frémont, N. Marion and S. P. Nolan, J. Organomet.
Chem., 2009, 694, 551-560; (b) R. L. Lalonde, W. E. Brenzovich,
D. Benitez, E. Tkatchouk, K. Kelley, W. A. Goddard and F. D.
Toste, Chem. Sci., 2010, 1, 226-233.
2005, 435, 519-522; (b) Y. Wang, F. W. Benz, Y. Wu, Q. Wang,
Y. Chen, X. Chen, H. Li, Y. Zhang, R. Zhang and J. Yang, Mol.
Pharmacol. 2016, 89, 233-242.
E. K. Leggans, K. K. Duncan, T. J. Barker, K. D. Schleicher and D.
L. Boger, J. Med. Chem. 2013, 56, 628-639.
(a) L. Ye, L. Cui, G. Zhang and L. Zhang, J. Am. Chem. Soc., 2010,
3
6
1
32, 3258-3259; (b) E. P. A. Talbot, M. Richardson, J. M.
McKenna and F. D. Toste, Adv. Synth. Catal. 2014, 356, 687-
91.
6
J. Han, N. Shimizu, Z. Lu, H. Amii, G. B. Hammond and B. Xu,
Org. Lett., 2014, 16, 3500-3503.
6
| Chem. Sci., 2015, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Zhang, Yun
Zhang, Yun
 Xue, Yibin
Xue, Yibin
 Li, Gang
Li, Gang
 Yuan, Haosen
Yuan, Haosen
 Luo, Tuoping
Luo, Tuoping