
Journal of the American Chemical Society p. 11914 - 11917 (2014)
Update date:2022-08-30
Topics:
 Reid, Robert C.
Reid, Robert C.
 Yau, Mei-Kwan
Yau, Mei-Kwan
 Singh, Ranee
Singh, Ranee
 Lim, Junxian
Lim, Junxian
 Fairlie, David P.
Fairlie, David P.
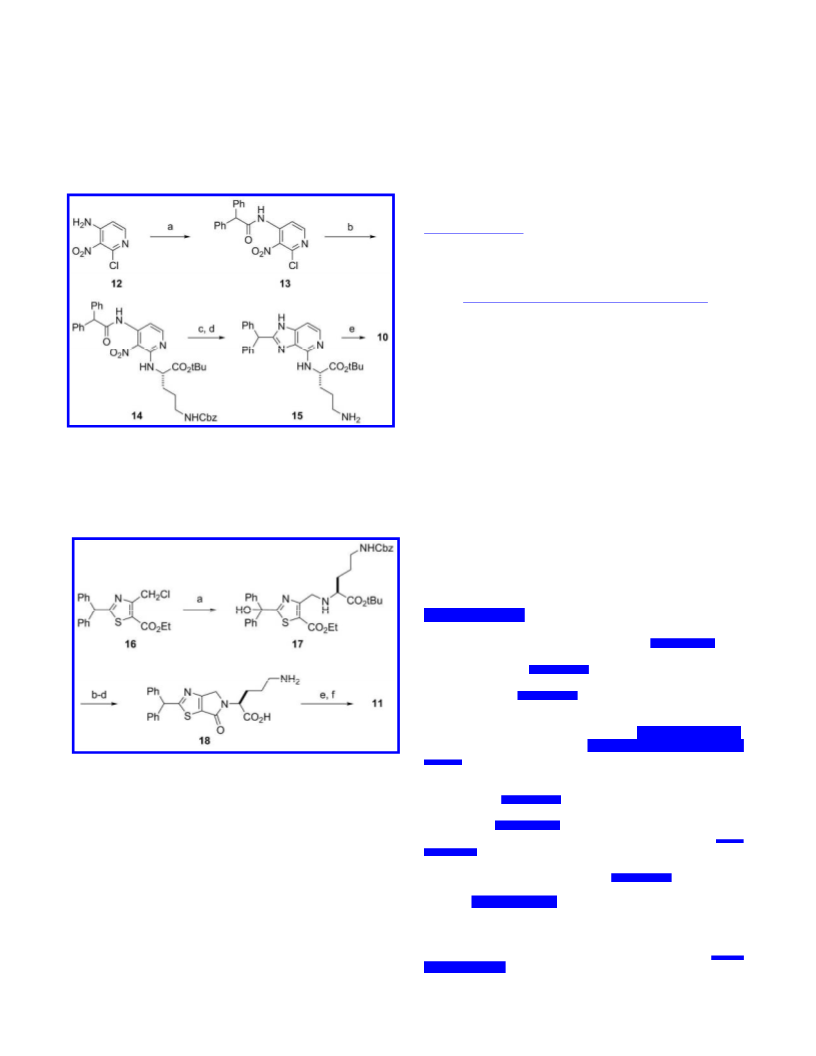
Heterocycles adjacent to amides can have important influences on molecular conformation due to stereoelectronic effects exerted by the heteroatom. This was shown for imidazole- and thiazole-amides by comparing low energy conformations (ab initio MP2 and DFT calculations), charge distribution, dipole moments, and known crystal structures which support a general principle. Switching a heteroatom from nitrogen to sulfur altered the amide conformation, producing different three-dimensional electrostatic surfaces. Differences were attributed to different dipole and orbital alignments and spectacularly translated into opposing agonist vs antagonist functions in modulating a G-protein coupled receptor for inflammatory protein complement C3a on human macrophages. Influences of the heteroatom were confirmed by locking the amide conformation using fused bicyclic rings. These findings show that stereoelectronic effects of heterocycles modulate molecular conformation and can impart strikingly different biological properties.
View More




Zhejiang PRIMAR Import & Export Trade Co., Ltd.
Contact:86-570-3630818
Address:No.1Puzhuyuan,Quhua,Zhejiang Province,China324004
Taizhou volsen chemical Co., Ltd
website:http://www.volsenchem.com
Contact:+86-576-88869393
Address:Jiaojiang District, Taizhou, Zhejiang, China.
Tianjin Crest Pharmaceutical R&D Co., Ltd. (Tianjin Yao Technology Development Co., Ltd.)
Contact:+86-22-66211386
Address:Building B5-405, No, 80 4th Avenue, TEDA, Tianjin, China P.R. 300457
Contact:0571-86821378 ,86820258,56836287,56830923,
Address:Block D ,20F, Tianyuan Building,No.508, Wensan RD, 310013,Hangzhou Zhejiang China
Contact:+1-284-4950244
Address:Box 3069, Road Town, Tortola, British Virgin Islands
Doi:10.1016/S0040-4039(00)84691-2
(1986)Doi:10.1007/BF01184884
(1994)Doi:10.1002/jhet.2459
(2016)Doi:10.1039/JR9510000311
()Doi:10.1021/jo01097a603
(1958)Doi:10.1016/j.tet.2018.12.022
(2019)