B.M. Choudary et al. / Journal of Catalysis 218 (2003) 191–200
199
(d) J.V. Sinisterra, J.M. Marinas, F. Riquelme, M.S. Arias, Tetrahe-
dron 44 (1988) 1431;
(e) B. Deschamps, J.P. Lampin, F. Mathey, J. Seyden-Penne, Tetrahe-
dron Lett. 13 (1977) 1137;
(f) E. Breuer, D.M. Bannet, Tetrahedron 34 (1978) 997;
(g) K. Ando, Tetrahedron Lett. 36 (1995) 4105;
(h) Y. Nakamura, C. Shin, Synthesis (1994) 552;
(i) P. Allevi, P. Ciuffreda, D. Colombo, D. Monti, G. Speranza,
J. Chem. Soc., Perkin Trans. 1 (1989) 1281;
(j) K. Ando, J. Org. Chem. 62 (1997) 1934;
(k) K. Ando, J. Org. Chem. 63 (1998) 8411;
(l) G. Jones, R.F. Maisey, Chem. Commun. (1968) 543;
(m) J.A. Marshell, C.P. Hagan, G.A. Flynn, J. Org. Chem. 40 (1975)
1162;
(n) G. Jones, R.F. Maisey, Chem. Commun. (1968) 543;
(o) K. Ando, Synlett. (2001) 1272;
(p) J. Motoyoshiya, T. Kusaura, K. Kokin, Y. Sei-ichi, Y. Takaguchi,
S. Narita, H. Aoysma, Tetrahedron 57 (2001) 1715;
(q) F. Texier-Boulett, A. Foucaud, Tetrahedron Lett. 21 (1980) 2161;
(r) S. Sano, T. Ando, K. Yokoyama, Y. Nagao, Synlett. (1998) 777;
(s) W.C. Still, C. Gennari, Tetrahedron Lett. 24 (1983) 4405;
(t) H. Rehwinkel, J. Skupsch, H. Vorbruggen, Tetrahedron Lett. 29
(1988) 1775;
Scheme 4. The plausible mechanism for the Wadsworth–Emmons reaction
catalyzed by HT–O Bu (cat. B).
t
(u) D.H. Wadsworth, O.E. Schupp, E.J. Seus, J.A. Ford, J. Org.
Chem. 30 (1965) 680.
[4] F. Texier-Boullet, D. Villemin, M. Ricard, H. Moison, A. Foucaud,
with the (EtO)2P(O)OH to manifest the catalytic activity.
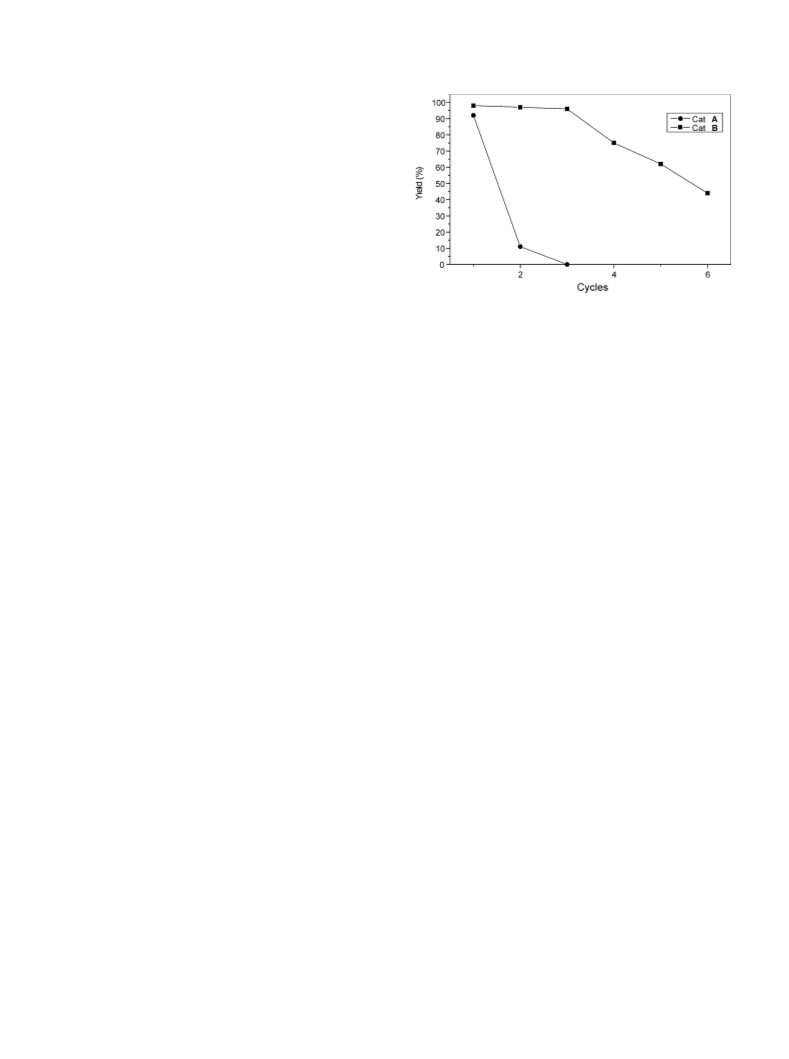
The higher activity and turnover number displayed by the
cat. B is ascribed to the smaller crystallites and high surface
area. All these results demonstrate that the WE reactions
could be conducted with high atom utilization using solid
base catalysts rather than soluble bases, which require near-
stoichiometric quantities.
Tetrahedron 41 (1985) 1259;
(b) M. Moison, F. Texier-Boullet, A. Foucaud, Tetrahedron 43 (1987)
537.
[5] (a) F. Cavani, F. Trifiro, A. Vaccari, Catal. Today 11 (1991) 173;
(b) F. Trifiro, A. Vaccari, in: G. Alberti, T. Bein (Eds.), Comprehen-
sive Supramolecular Chemistry, Vol. 7, Pergmon Press, Oxford, 1996,
p. 251;
(c) B. Sels, D. De Vos, M. Buntinx, F. Pierard, A. Kirsch-De Mes-
maeker, P. Jacobs, Nature 400 (1999) 855;
(d) B.M. Choudary, M. Lakshmi Kantam, A. Rahman, Ch. Venkat
Reddy, K.K. Rao, Angew. Chem., Int. Ed. 40 (2001) 763;
(e) B.M. Choudary, N.S. Chowdari, M. Lakshmi Kantam, K.V. Ragha-
van, J. Am. Chem. Soc. 123 (2001) 9220;
(f) B.M. Choudary, B. Bharathi, Ch. Venkat Reddy, M. Lakshmi Kan-
tam, K.V. Raghavan, Chem. Commun. (2001) 1736;
(g) T. Matsushita, K. Ebitani, K. Kaneda, Chem. Commun. (1999) 265;
(h) M. Ballabeni, R. Ballini, F. Bigi, R. Maggi, M. Parrini, G. Predieri,
G. Sartori, J. Org. Chem. 64 (1999) 1029;
(i) B.M. Choudary, N.S. Chowdari, M. Sateesh, M. Lakshmi Kantam,
Angew. Chem., Int. Ed. 40 (2001) 4620;
(j) R.K. Allada, A. Navrotsky, H.T. Berbeco, W.H. Casey, Science 296
(2002) 721;
Acknowledgments
This work was realized in the frame of an Indo-French
co-operative program, funded by IFCPAR (Project No.
IFC/1106-2/96/2460). Ch.V.R. and B.B. thank the Council
of Scientific and Industrial Research (CSIR), India, for the
award of Senior Research Fellowship. We also thank Dr. B.
Jagadeesh (Center for NMR spectroscopy, IICT) for record-
ing 27Al MAS NMR spectra.
(k) B.M. Choudary, N.S. Chowdari, K. Jyothi, M. Lakshmi Kantam,
J. Am. Chem. Soc. 124 (2002) 5341.
References
[6] (a) V. Prevot, C. Forano, J.P. Besse, J. Mat. Chem. 9 (1999) 155;
(b) B.M. Choudary, M. Lakshmi Kantam, V. Neeraja, K.K. Rao, F.
Figueras, L. Delmotte, Green. Chem. 3 (2001) 257.
[1] (a) W.E. Wadsworth, W.D. Emmons, J. Am. Chem. Soc. 83 (1961)
1733;
(b) W.E. Wadsworth, Org. React. 25 (1977) 73.
[7] (a) W.T. Reichle, J. Catal. 94 (1985) 547;
[2] (a) A.W. Johnson, in: Ylides and Imines of Phosphorus, Wiley, New
York, 1993, p. 307;
(b) J.G. Numan, P.B. Himelfarb, R.G. Herman, K. Klier, C.E. Bogdan,
G.W. Simmons, Inorg. Chem. 28 (1989) 3868;
(c) C. Busetto, G. Delpiero, G. Manara, F. Trifiro, A. Vaccari,
J. Catal. 85 (1984) 260;
(d) K. Yamaguchi, K. Ebitani, T. Yoshida, H. Yoshida, K. Kaneda,
J. Am. Chem. Soc. 121 (1999) 4526;
(e) V.J. Bulbule, V.H. Deshpande, S. Velu, A. Sudalai, S. Sivasankar,
V.T. Sathe, Tetrahedron 55 (1999) 9325;
(f) E. Suzuki, Y. Ono, Bull. Chem. Soc. Jpn. 61 (1988) 1008;
(g) E. Suzuki, M. Okamoto, Y. Ono, J. Mol. Catal. 61 (1990) 283;
(h) C. Cativiela, F. Figueras, J.I. Garcia, J.A. Mayoral, M. Zurbano,
Synth. Commun. 25 (1995) 1745;
(b) B.J. Walker, in: J.I.G. Cadogan (Ed.), Organophosphorus Reagents
in Organic Synthesis, Academic Press, London, 1979, p. 155, Chap. 3;
(c) J. Boutagy, R. Thomas, Chem. Rev. 74 (1974) 87;
(d) B.E. Maryanoff, A.B. Reitz, Chem. Rev. 89 (1989) 863;
(e) K.C. Nicolaou, M.W. Harter, J.L. Gunzner, A. Nadin, Liebigs
Ann./Recueil. (1997) 1283.
[3] (a) F. Texier-Boullet, A. Foucaud, Synthesis (1979) 884;
(b) J.V. Sinisterra, Z. Mouloungui, M. Delmas, A. Gaset, Synthesis
(1985) 1097;
(c) M.S. Climent, J.M. Marinas, Z. Mouloungui, Y. LeBigot, M. Del-
mas, A. Gaset, J.V. Sinisterra, J. Org. Chem. 54 (1989) 3695;

 Choudary, Boyapati M.
Choudary, Boyapati M.
 Kantam, Mannepalli L.
Kantam, Mannepalli L.
 Reddy, Chinta Reddy V.
Reddy, Chinta Reddy V.
 Bharathi, Balagam
Bharathi, Balagam
 Figueras, Francois
Figueras, Francois