D. R o¨ me et al. / Tetrahedron Letters 48 (2007) 635–638
637
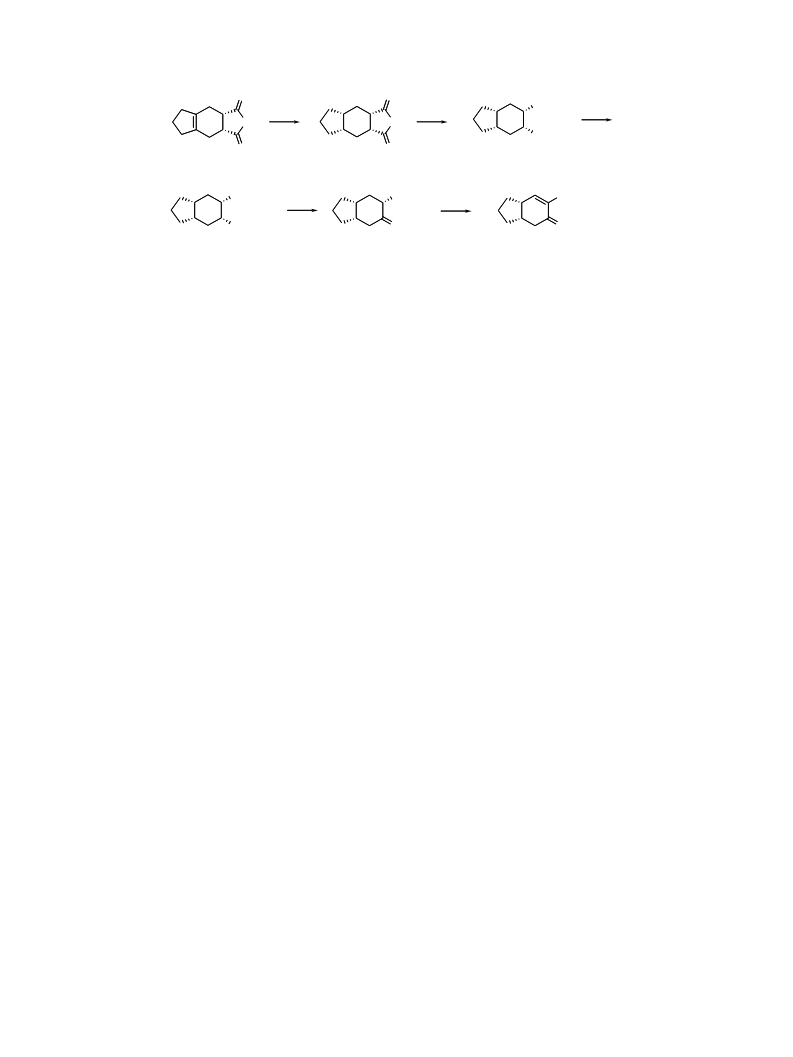
O
O
CO Me
2
b
c,d
a
O
O
CO H
2
O
O
4
5
16
CO Me
CO Me
CO Me
2
2
2
e,f
g
CHO
O
O
17
18
3
Scheme 3. Reagents and conditions: (a) Pt
rt, overnight; (d) Pd/C, H
CH Cl
2
O, H
2
(1 atm), THF, rt, overnight; (b) (DHQD)
2
AQN, MeOH, Et
2
O, ꢀ18 ꢁC, 3 d; (c) (COCl)
2
, toluene,
, MeOH/
2
(40 psi), 2,6-lutidine, THF, overnight; (e) ZnCl , Et N, TMSCl, benzene, rt (0.5 h) to 40 ꢁC, overnight; (f) O
(4:1), ꢀ7 ꢁC, 39% from 5; (g) (i) PhSeCl, pyridine, CH Cl , 0 ꢁC, 0.5 h; (ii) 35% H , CH Cl , 0 ꢁC, 0.5 h, 48%.
2
3
3
2
2
2
2
2
O
2
2
2
Attempts to form the silylenol ether by conventional
methods, for example, using a base and then chlorotri-
methylsilane, failed. However, enolization of aldehyde
5. Becker, H.; Sharpless, K. B. Angew. Chem., Int. Ed. 1996,
5, 448.
3
6
. (a) Blomquist, A. T.; Wolinsky, J.; Meinwald, Y. C.;
Longon, D. T. J. Am. Chem. Soc. 1956, 77, 6057–6063; (b)
Nguyet, A. L.; Jones, M., Jr.; Bickelhaupt, F.; De Wolf,
W. H. J. Am. Chem. Soc. 1989, 111, 8491–8493; (c)
Sustmann, R.; Daute, P.; Sauer, R.; Sommer, A.; Traha-
novsky, W. S. Chem. Ber. 1989, 122, 1551–1558.
1
7 in the presence of chlorotrimethylsilane using trieth-
1
8
ylamine-zinc chloride complex was successful, and the
1
9
product was immediately ozonolyzed to yield b-keto
ester 18 in a 39% yield and 6 steps from 5. Introduction
of the a,b-unsaturation in 18 to yield a,b-unsaturated b-
7
. (a) Bailey, W. J.; Sorenson, W. R. J. Am. Chem. Soc. 1989,
76, 5421–5423; (b) Bartlett, P. D.; Wingrove, A. S.;
Owyang, R. J. Am. Chem. Soc. 1968, 90, 6067–6070.
2
keto ester 3 was done as previously described using
PhSeCl and hydrogen peroxide. Keto ester 3 was ob-
2
0
tained in a 90% enantiomeric excess and 3 has previ-
8. Harmata, M.; Bohnert, G. J. Org. Lett. 2003, 5, 59–
1.
6
ously been transformed to the target dialdehyde 2 in a
2
9. Barco, A.; Benetti, S.; Casolari, A.; Menfredini, S.; Pollini,
2
5% overall yield.
G. P.; Polo, E.; Zanirato, V. Tetrahedron 1989, 45, 3935–
3
944.
In summary, we have developed a facile method for the
asymmetric synthesis of an important intermediate in
the synthesis of biologically active dialdehydes utilizing
a nonenzymatic asymmetric desymmetrization (ADS)
reaction. We have also shown that reactive exocylic
dienes can be readily synthesized from quatenary amines
using recyclable ion-exchange resins, eliminating the
need for expensive silver salts.
1
0. Hegedus, L. S.; Kambe, N.; Ishii, Y.; Mori, A. J. Org.
Chem. 1985, 50, 2240–2243.
1. Martin, H. D.; Mayer, B. Tetrahedron Lett. 1979, 2351–
1
2
352.
12. (a) Wilkening, D.; Mundy, B. P. Synth. Commun. 1984, 14,
227–238; (b) Padwa, A.; Hornbuckle, S. F.; Fryxell, G. E.;
Stull, P. D. J. Org. Chem. 1989, 54, 817–824.
3. Hanessian, S.; Ugolini, A.; Dub e´ , D.; Glamyan, A. Can. J.
Chem. 1984, 62, 2146–2147.
1
1
4. Activation/regeneration of ion-exchange resin: The resin
should be activated prior to use according to the method
described by the supplier, DOWEX 550A OH; regenerant,
Acknowledgement
4
–8% NaOH; rinse requirement, 2–5 Bed volumes; tem-
Financial support from the Swedish Natural Science
Council is gratefully acknowledged.
perature, ambient or up to 60 ꢁC.
1
1
5. Ishizumi, K.; Antoku, F.; Maruyama, I.; Kojima, A.
European Patent Application 1986, EP 86-104190, CAN
1
06-33119.
6. Based on the enantiomeric excess of the final product 3 ee
was determined using chiral chromatography: Daicel
CHIRAL PAK AD-RH column (4.6 · 150 mm, particle
size 5 lM), Waters 2705 Separations Module, Waters 2996
Photodiode Array Detector and acetonitrile/water eluent.
References and notes
1
. (a) Anke, H.; Sterner, O. Planta Med. 1991, 57, 344–346;
b) Sterner, O.; Bergman, R.; Kihlberg, J.; Wickberg, B. J.
(
Nat. Prod. 1985, 48, 279–288; (c) Sterner, O.; Carter, R.;
Nilsson, L. Mutat. Res. 1987, 188, 169–174; (d) Gustafs-
son, J.; Jonassohn, M.; Kahnberg, P.; Anke, H.; Sterner,
O. Nat. Prod. Lett. 1997, 9, 253–258.
17. Houpis, I. N.; Molina, A.; Reamer, R. A.; Lynch, J. E.;
Volante, R. P.; Reider, P. J. Tetrahedron Lett. 1993, 34,
2593–2596.
18. Simoneau, B.; Brassard, P. Tetrahedron 1988, 44, 1015–
1022.
2
3
4
. Aujard, I.; R o¨ me, D.; Arzel, E.; Johansson, M.; De Vos,
D.; Sterner, O. Bioorg. Med. Chem. 2005, 13, 6145–
19. (a) Arai, Y.; Hayashi, Y.; Yamamoto, M.; Takayema, H.;
Koizumi, T. J. Chem. Soc., Perkin Trans. 1 1988, 3133–
3140; (b) Corey, E. J.; Peterson, R. T. Tetrahedron Lett.
1985, 26, 5025–5028.
6
150.
. (a) Spivey, A. C.; Andrews, B. I. Angew. Chem., Int. Ed.
001, 40, 3131–3134; (b) Willis, M. C. J. Chem. Soc.,
2
Perkin Trans. 1 1999, 1765–1784.
. (a) Chen, Y.; Tian, S. K.; Deng, L. J. Am. Chem. Soc.
20. Methyl
indene-5-carboxylate (3). PhSeCl (201 mg, 1.05 mmol)
was dissolved in CH Cl (30 ml) and cooled to 0 ꢁC.
Pyridine (89 ll, 1.10 mmol) was added and the orange
(3aS,7aS)-6-oxo-2,3,3a,6,7,7a-hexahydro-1H-
2
000, 122, 9542–9543; (b) Hiratake, J.; Yamamoto, Y.;
2
2
Oda, J. J. Chem. Soc., Chem. Commun. 1985, 1717–1719.

 R?me, Daniel
R?me, Daniel
 Johansson, Martin
Johansson, Martin
 Sterner, Olov
Sterner, Olov