
European Journal of Medicinal Chemistry p. 289 - 301 (2015)
Update date:2022-08-17
Topics:
 Nirogi, Ramakrishna
Nirogi, Ramakrishna
 Mohammed, Abdul Rasheed
Mohammed, Abdul Rasheed
 Shinde, Anil K.
Shinde, Anil K.
 Bogaraju, Narsimha
Bogaraju, Narsimha
 Gagginapalli, Shankar Reddy
Gagginapalli, Shankar Reddy
 Ravella, Srinivasa Rao
Ravella, Srinivasa Rao
 Kota, Laxman
Kota, Laxman
 Bhyrapuneni, Gopinadh
Bhyrapuneni, Gopinadh
 Muddana, Nageswara Rao
Muddana, Nageswara Rao
 Benade, Vijay
Benade, Vijay
 Palacharla, Raghava Chowdary
Palacharla, Raghava Chowdary
 Jayarajan, Pradeep
Jayarajan, Pradeep
 Subramanian, Ramkumar
Subramanian, Ramkumar
 Goyal, Vinod Kumar
Goyal, Vinod Kumar
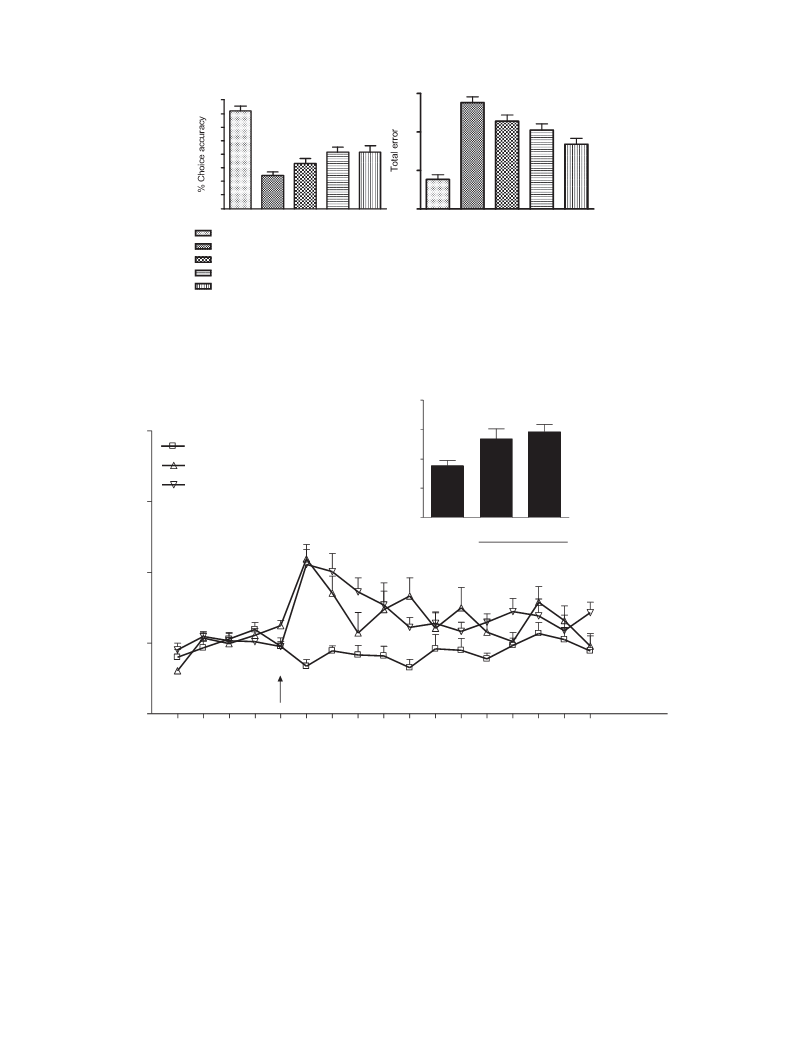
Alzheimer's disease (AD) is a neurodegenerative disease which has a higher prevalence and incidence in older people. The need for improved AD therapies is unmet. The 5-hydroxytryptamine4 receptor (5-HT4R) partial agonists may be of benefit for both the symptomatic and disease-modifying treatment of cognitive disorders associated with AD. Herein, we report the design, synthesis and SAR of imidazo[1,5-a] pyridine derivatives as 5-HT4R partial agonists. The focused SAR, optimization of ADME properties resulted the discovery of compound 5a as potent, selective, brain penetrant 5-HT4 partial agonist as a lead compound with good ADME properties and efficacy in both symptomatic and disease modifying animal models of cognition.
View More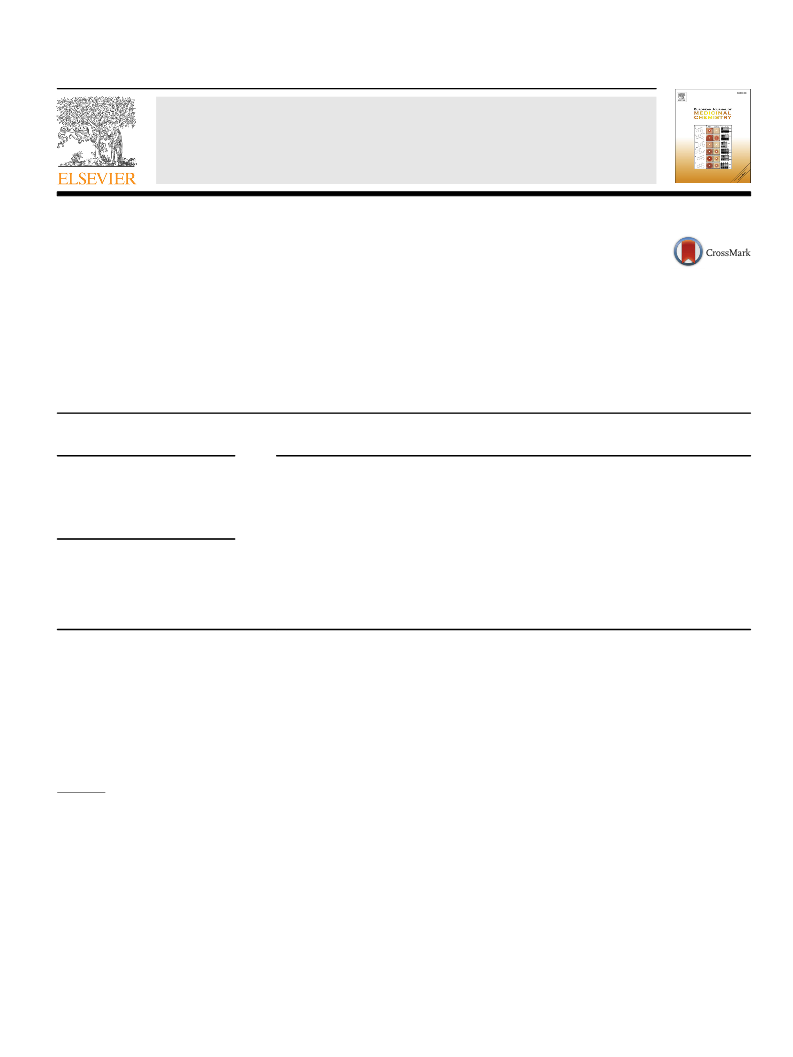



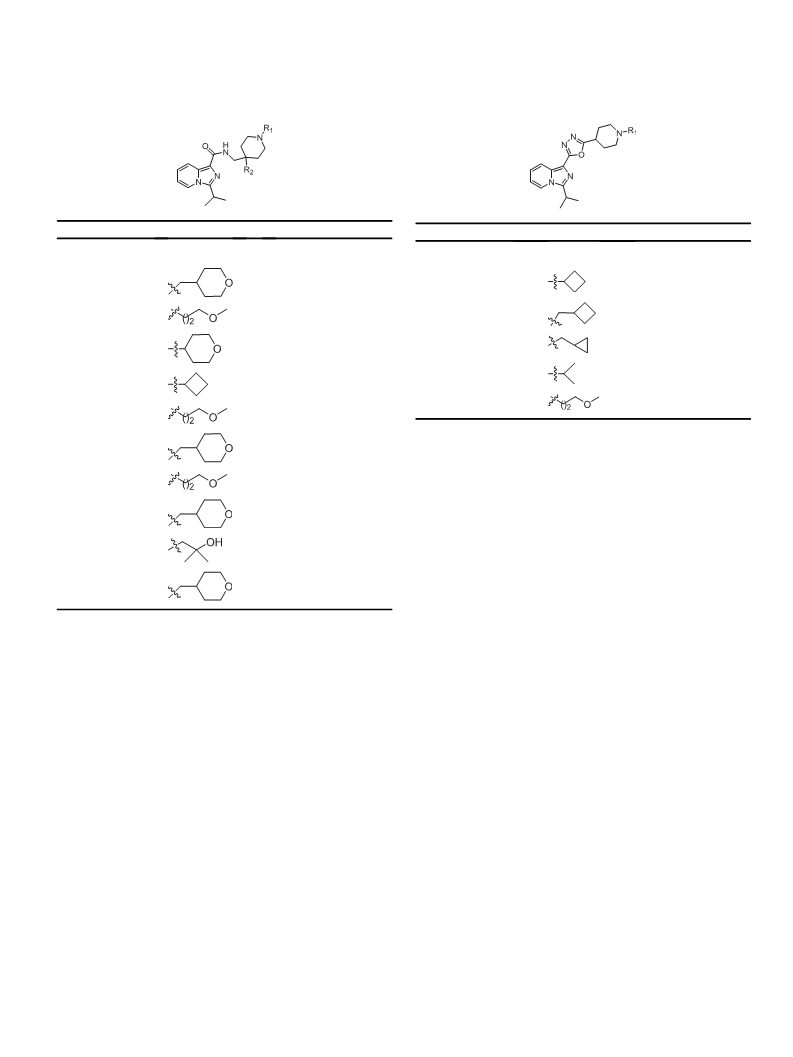





Shanghai Bocimed Pharmaceutical Co., Ltd.
website:http://www.bocimed.com
Contact:+86-21-68861632
Address:Building 1, Lane 647, Songtao Road, Zhangjiang High-Tech Park, Shanghai
Contact:+91 9963263336
Address:Plot#146A, IDA Mallapur, Hyderabad - 500072
Dongying J&M Chemical Co., Ltd,
Contact:546-8551108
Address:Room 1219, Zisheng Mansion, Zibo Road, Dongying, Shandong, China
Shanghai Rainbow Chemistry Co., Ltd.
Contact:+86-21-64968086-5815/5812
Address:3rd floor, Building 7, 251 Faladi Road, Zhangjiang Hi-Tech Park, Pudong District, Shanghai, P.R. China
Hangzhou Deli Chemical Co.,Ltd.
website:http://www.dlchemical.com
Contact:86 571 28006267
Address:Tangxi Industrial Area, Yuhang District, Hangzhou
Doi:10.1016/j.bmc.2011.02.040
(2011)Doi:10.1016/j.ejmech.2020.112203
(2020)Doi:10.1246/bcsj.34.1045
(1961)Doi:10.3390/molecules20045360
(2015)Doi:10.1021/acsmedchemlett.6b00311
(2017)Doi:10.1021/jo00039a036
(1992)