3450
B. Baek et al. / Tetrahedron Letters 44 (2003) 3447–3450
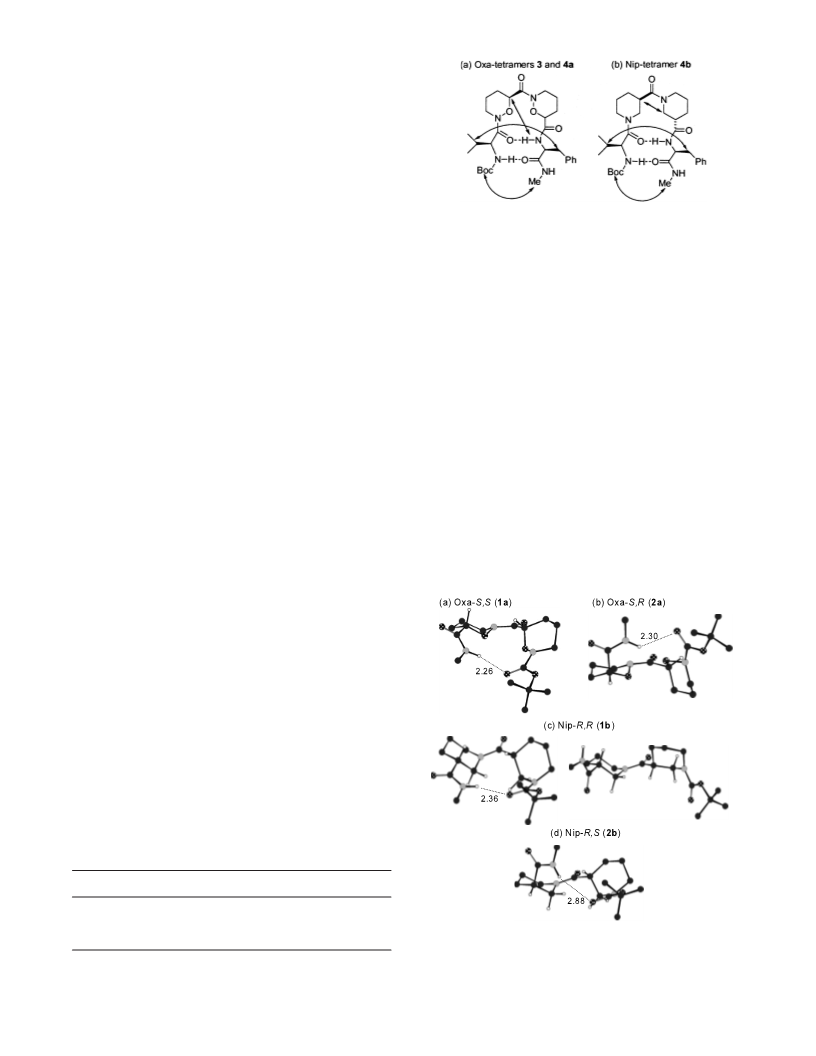
Oxa,L formed folded structures, however, only L,(R)-
Nip,(S)-Nip,L promoted the stable folded conforma-
tions.
5. Since N-acetylated peptidomimetics possessing di-oxa-
nipecotic acids were sparingly soluble in nonpolar sol-
vents such as methylene chloride and chloroform, which
are ideal for examining hydrogen bond formations by
FT-IR and NMR, Boc-protected compounds were pre-
pared for structural studies.
Acknowledgements
6. According to FT-IR and NMR studies of four Boc-pro-
tected Nip-tetramers [L,(R)-Nip,(S)-Nip,L, L,(R)-
Nip,(R)-Nip,L, L,(S)-Nip,(S)-Nip,L, L,(S)-Nip,(R)-
Nip,L], only L,(R)-Nip,(S)-Nip,L (4b) exhibited folded
conformations. Accordingly, we selected 4b for these
studies. In fact, this result is a little different from previ-
ous results that heterochiral acetylated Nip-tetramers
(L,(R)-Nip,(S)-Nip,L and L,(S)-Nip,(R)-Nip,L) promote
turn formations.4c We assumed that carbonyl of Boc
group is a worse hydrogen bond acceptor to C-terminal
NH than that of acetyl group and thus only L,(R)-
Nip,(S)-Nip,L forms the folded conformation.
This work was supported by a grant of the Center for
Integrated Molecular Systems (KOSEF). We thank Dr.
Yong Jun Chung for several helpful discussions con-
cerning IR studies.
References
1. (a) Yang, D.; Li, B.; Ng, F.-F.; Yan, Y.-L.; Qu, J.; Wu,
Y.-D. J. Org. Chem. 2001, 66, 7303; (b) Yang, D.; Qu, J.;
Li, W.; Zhang, Y.-H.; Ren, Y.; Wang, D.-P.; Wu, Y.-D.
J. Am. Chem. Soc. 2002, 124, 12410; (c) Yang, D.; Zhang,
Y.-H.; Zhu, N.-Y. J. Am. Chem. Soc. 2002, 124, 9966.
2. (a) Shin, I.; Lee, M.-r.; Lee, J.; Jung, M.; Lee, W.; Yoon,
J. J. Org. Chem. 2000, 65, 7667; (b) Shin, J.; Lee, J.
Synlett 2000, 1297; (c) Shin, I.; Park, K. Org. Lett. 2002,
4, 869; (d) Lee, M.-r.; Lee, J.; Shin, I. Synlett 2002, 1463;
(e) Lee, M.-r.; Lee, J.; Baek, B.-h.; Shin, I. Synlett 2003,
325; (f) Baek, B.-h.; Lee, M.-r.; Kim, K.-Y.; Cho, U.-I;
Boo, D. W.; Shin, I. Org. Lett. 2003, in press; (g) Lee,
M.-r.; Kim, K.-Y.; Cho, U.-I; Boo, D. W.; Shin, I. Chem.
Commun. 2003, in press.
3. For reviews, see: (a) Seebach, D.; Mattews, J. L. Chem.
Commun. 1997, 2015; (b) Gellman, S. H. Acc. Chem. Res.
1998, 31, 173; (c) Hill, D. J.; Mio, M. J.; Prince, R. B.;
Hughes, T. S.; Moore, J. S. Chem. Rev. 2001, 101, 3893;
(d) Cheng, R. P.; Gellman, S. H.; DeGrado, W. F. Chem.
Rev. 2001, 101, 3219.
7. See Supplementary Material.
8. (a) Maxfield, F. R.; Leach, S. J.; Stimson, E. R.; Powers,
S. P.; Scheraga, H. A. Biopolymers 1979, 18, 2507; (b)
Gardner, R. R.; Liang, G.-B.; Gellman, S. H. J. Am.
Chem. Soc. 1999, 121, 1806.
9. We also performed DMSO titration and concentration
dependence experiments for all the tetramers. See Supple-
mentary Material.
10. Gaussian 98: Frisch, M. J.; Trucks, G. W.; Schlegel, H.
B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.;
Zakrzewski, V. G.; Montgomery, J. A.; Stratmann, R. E.;
Burant, J. C.; Dapprich, S.; Millam, J. M.; Daniels, A.
D.; Kudin, K. N.; Strain, M. C.; Farkas, O.; Tomasi, J.;
Barone, V.; Cossi, M.; Cammi, R.; Mennucci, B.;
Pomelli, C.; Adamo, C.; Clifford, S.; Ochterski, J.;
Petersson, G. A.; Ayala, P. Y.; Cui, Q.; Morokuma, K.;
Malick, D. K.; Rabuck, A. D.; Raghavachari, K.; Fores-
man, J. B.; Cioslowski, J.; Ortiz, J. V.; Stefanov, B. B.;
Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Gom-
perts, R.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham,
M. A.; Peng, C. Y.; Nanayakkara, A.; Gonzalez, C.;
Challacombe, M.; Gill, P. M. W.; Johnson, B. G.; Chen,
W.; Wong, M. W.; Andres, J. L.; Head-Gordon, M.;
Replogle, E. S.; Pople, J. A. Gaussian, Inc., Pittsburgh,
PA, 1998.
4. (a) Chung, Y. J.; Christianson, L. A.; Stanger, H. E.;
Powell, D. R.; Gellman, S. H. J. Am. Chem. Soc. 1998,
120, 10555; (b) Chung, Y. J.; Huck, B. R.; Christianson,
L. A.; Stanger, H. E.; Krautha¨user, S.; Powell, D. R.;
Gellman, S. H. J. Am. Chem. Soc. 2000, 122, 3996; (c)
Huck, B. R.; Fisk, J. D.; Gellman, S. H. Org. Lett. 2000,
2, 2607.

 Baek, Bong-Hyeon
Baek, Bong-Hyeon
 Lee, Myung-Ryul
Lee, Myung-Ryul
 Kim, Kwang-Yon
Kim, Kwang-Yon
 Cho, Ung-In
Cho, Ung-In
 Boo, Doo Wan
Boo, Doo Wan
 Shin, Injae
Shin, Injae