Full Paper
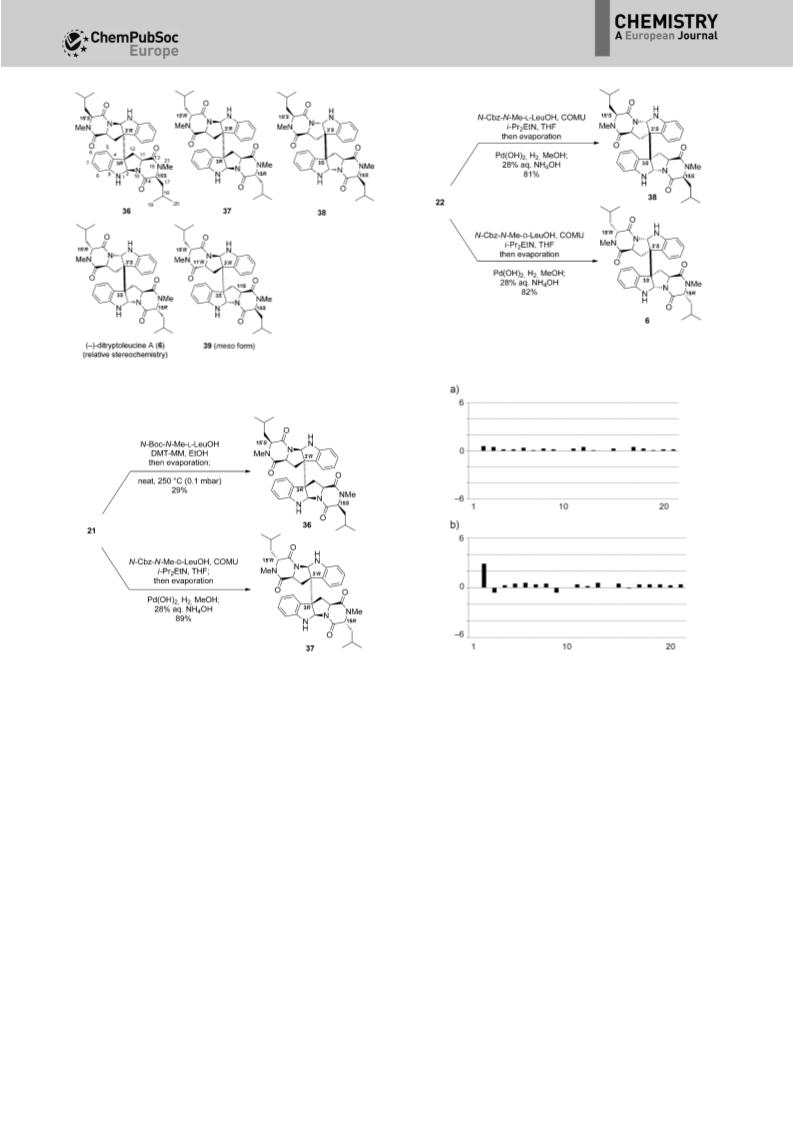
(4),[6] 1’-(2-phenylethylene)-ditryptophenaline (5),[7] ditryptoleu-
cine A (6),[8] WIN 64745 (7),[2a] cristatumin C (8),[9] and asperdi-
min (9),[10] which possess vicinal quaternary stereocenters
joined though C3(sp3)ÀC3(sp3) bonds. On the other hand,
a C3(sp3)ÀC7(sp2) bridge has been observed in the structure in
naseseazine A (10) and naseseazine B (11).[11] Furthermore, in
2008, pestalazine B (12) possessing a C3(sp3)ÀN1 bridge was
isolated.[4,12] These alkaloids exhibit a unique architecture
derived from an indole oxidation reaction of tryptophan in the
are not easy because common organic compounds are usually
insoluble in water, and sometimes the water reacts with the
substrates or reagents. With alkaloids that contain basic amine
portions in the molecule, such situations occur under neutral
or basic conditions. In contrast, under acidic conditions, basic
alkaloids form water-soluble salts. In addition, salt formation
possibly prevents side reactions derived from the nucleophilici-
ty of the lone pair of the amine without any protective groups.
Thus, acids would play an important role in aqueous biosyn-
thesis of alkaloids. Recently, Boger and co-workers reported an
elegant bioinspired radical-mediated oxidative coupling
reaction of vinblastine and related alkaloids in aqueous HCl
and 2,2,2-trifluoroethanol.[14]
biosynthesis and
a wide variety of biological activities;
accordingly, several biomimetic and non-biomimetic ap-
proaches to the synthesis of these alkaloids have
been reported.[9b,10b,11b,12,13]
A number of pioneering researchers have adopted a proce-
dure to define bioinspired oxidative coupling reactions of
tryptamine or tryptophan units. In 1981, Nakagawa and co-
workers reported a bioinspired coupling protocol enlisting
a thallium(III)-mediated oxidative dimerization reaction for the
first total synthesis of ditryptophenaline (4).[13a] Although the
yield of the key coupling reaction was not excellent (3% yield),
the work made a huge contribution to the recognition of the
radical-mediated biosynthetic pathway, the determination of
the absolute stereochemistry, and the first attempt at a direct
dimerization reaction with an unprotected indole core of
a tryptophan derivative. In 2008, Movassaghi and co-workers
reported elegant syntheses of WIN 64821 (1), ditryptophena-
line (4), and 1’-(2-phenylethylene)-ditryptophenaline (5)
through cobalt(I)-mediated bioinspired dimerization reactions
with C3-brominated pyrollidinoindoline derivatives.[13e] The
method of Movassaghi et al. has been employed for several
syntheses of the same class of alkaloids by his own and other
groups. In addition, the group of Movassaghi also published
an asymmetrically connected dimerization reaction through di-
azine fragmentation in 2011.[13k] In 2008, de Lera and co-work-
ers accomplished an efficient synthesis of WIN 64821 (1) with
the method of Movassaghi et al. as a bioinspired dimerization
reaction in a seven-step sequence with a fully protected d-
tryptophan derivative and expanded it to the preparation of
related natural products, including WIN 64745 (7), cristatu-
min C (8), and asperdimin (9).[9b,10b] More recently, an alterna-
tive approach by using a nickel-catalyzed reductive dimeriza-
tion reaction of C3-brominated pyrrolidinoindoline derivatives
to prepare WIN 64821 (1) was developed by Oguri and co-
workers.[13p] The first total synthesis and structure revision of
naseseazine A (10) and naseseazine B (11) were achieved by
Kim and Movassaghi in 2011 by using a Friedel–Crafts-type
coupling reaction as a bioinspired heterodimerization reac-
tion.[11b] Reisman and co-workers also reported an elegant and
non-biomimetic synthesis of naseseazine A (10) and
naseseazine B (11) that involved a CuI-catalyzed arylation
reaction of tryptophan derivatives.[13n]
Our proposed biosynthetic pathway for dimeric diketopiper-
azine alkaloids is shown in Scheme 1. After the water-soluble
salt of tryptophan is formed under acidic conditions, it receives
selective one-electron oxidation on the indole core without un-
desired oxidation of the primary amine. The generated radical
compounds have several resonance hybrids, such as active
species of the radical on the C3, C7, or N1 atom (A, B, or C in
Scheme 1). These intermediates dimerize to provide the corre-
sponding natural product scaffolds: A+A for compounds 1–9,
A+B for compounds 10 and 11, and A+C for compound 12.
Recently, Watanabe and co-workers reported an enzymatic bio-
synthesis of ditryptophenaline (4) through a radical-mediated
coupling reaction with cytochrome P450.[15]
Scheme 1. Proposed dimerization reaction of the tryptophan units in the
biosynthetic pathway.
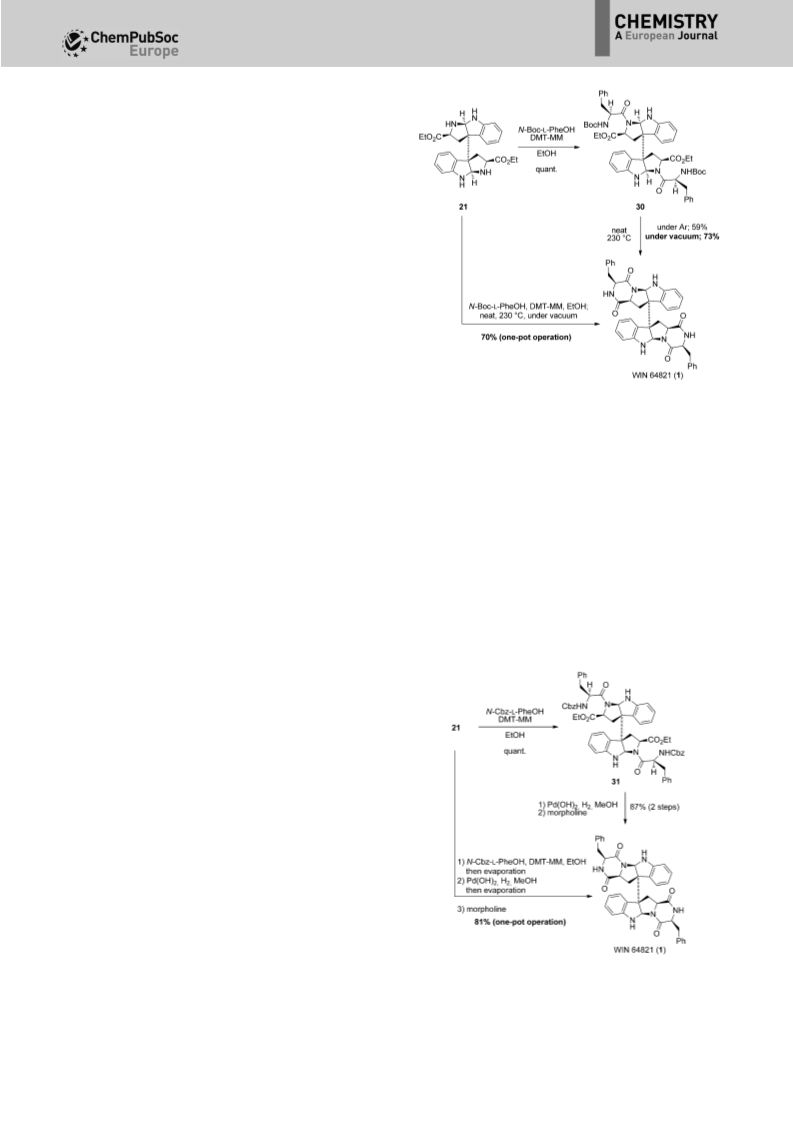
In 2013, we reported two one-pot or three-step syntheses of
WIN 64821 (1), ditryptophenaline (4), and naseseazine B (11) by
using an originally developed, bioinspired dimerization
reaction in acidic aqueous media along with our proposed
biosynthetic pathway.[16] Full details of the development of the
bioinspired dimerization reaction of tryptophan derivatives in
aqueous acidic media are provided herein. In addition, total
Despite all of these elegant strategies, we were convinced
that a more direct bioinspired solution to the preparation of
tryptophan-based diketopiperazine alkaloids was possible. In
a true biosynthesis, direct dimerization methods of tryptophan
without a special protective group on the substrates in aque-
ous media should be performed. Organic reactions in water
Chem. Eur. J. 2016, 22, 1277 – 1291
1278
ꢀ 2016 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Tadano, Shinji
Tadano, Shinji
 Sugimachi, Yukihiro
Sugimachi, Yukihiro
 Sumimoto, Michinori
Sumimoto, Michinori
 Tsukamoto, Sachiko
Tsukamoto, Sachiko
 Ishikawa, Hayato
Ishikawa, Hayato