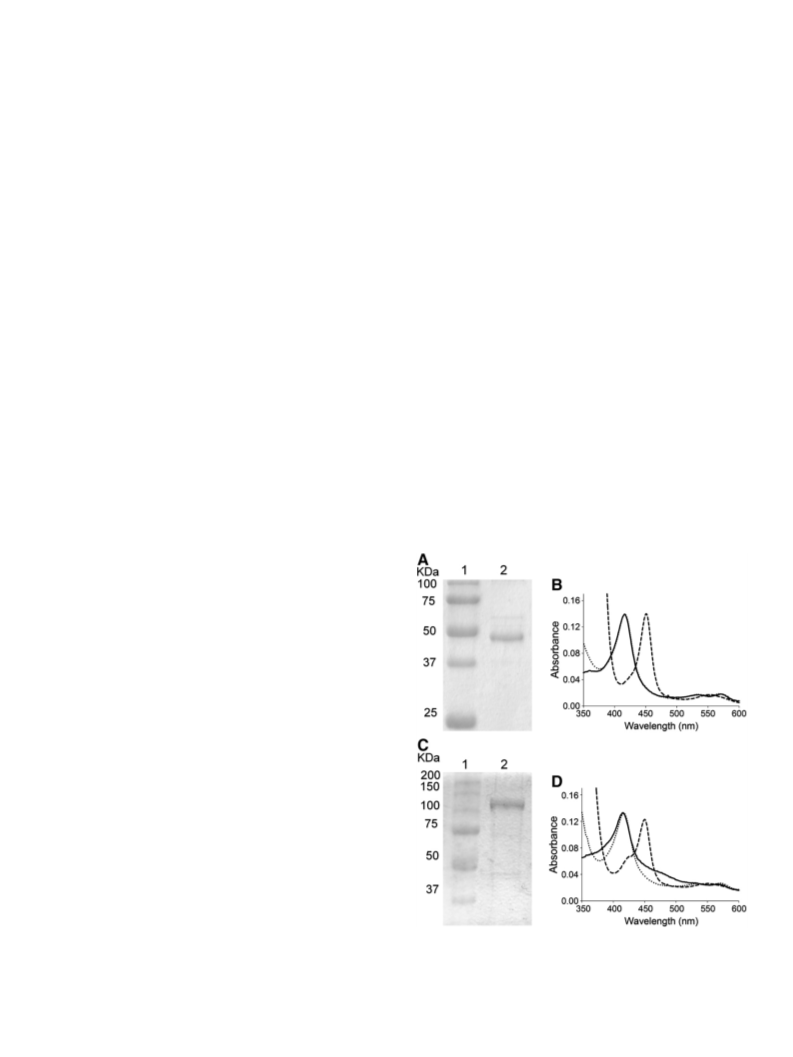
284
F. Rua et al. / Journal of Inorganic Biochemistry 117 (2012) 277–284
[8] M. Fairhead, S. Giannini, E.M. Gillam, G. Gilardi, J. Biol. Inorg. Chem. 10 (2005)
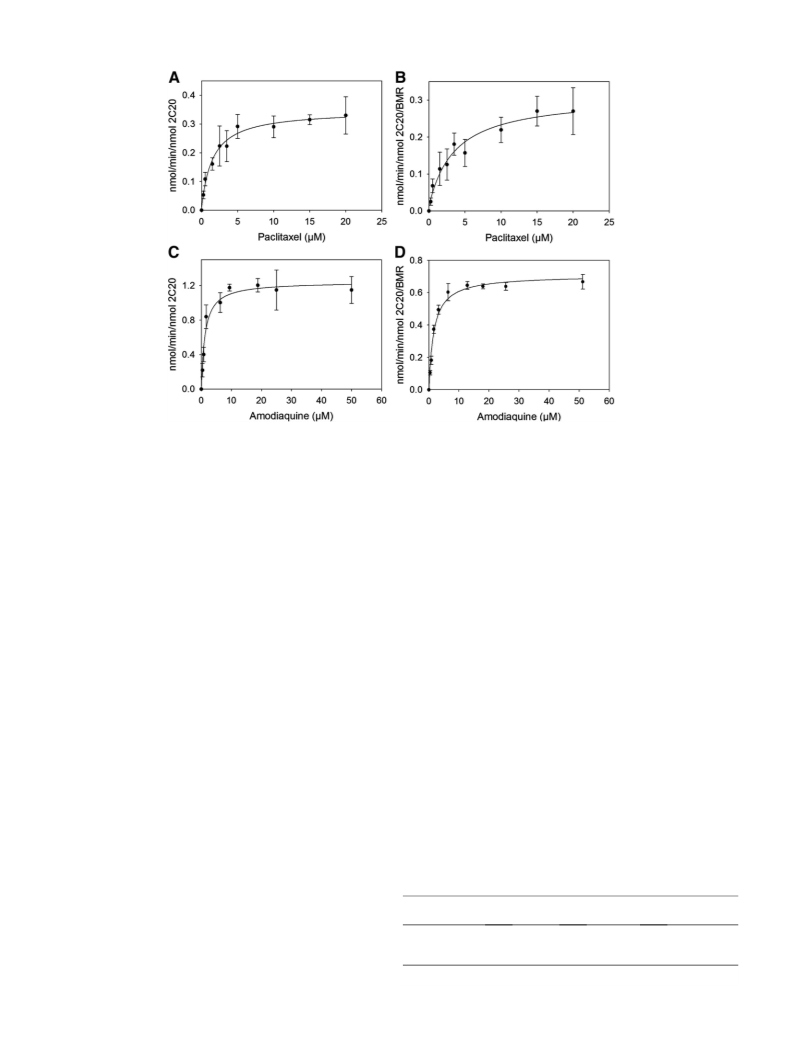
The electrochemical platforms were tested using paclitaxel and
842–853.
amodiaquine as substrates and the electrocatalytic properties of the
P450 2C20 immobilized on different surfaces was compared to the
P450 reconstituted system in presence of the human P450 reductase
and NADPH in solution. The best electrochemical platform was found
to be the one where P450 2C20 was immobilized on glassy carbon elec-
trodes using AuNPs.
When the AuNPs were used to increase the sensitivity of the system it
was possible to obtain a platform where the protein functioned as well as
in the in vitro assay with KM values of 3.0 0.5 μM and 1.4 0.4 μM for
paclitaxel and amodiaquine, respectively, in perfect agreement with the
P450 2C20 values.
In conclusion, starting from the cloning of an intriguing animal
P450 we have developed two alternative methods to study the
M. fascicularis P450 2C20 enzyme in vitro. Both the chimeric P450
2C20/BMR and the electrochemical platforms are representative of
the catalytic properties of the physiological monooxygenase system.
Through the P450 2C20/BMR chimera and the immobilization on
electrode surfaces we have bypassed the requirement for the redox
partner and NADPH as electron source, the two main obstacles for de-
veloping P450 enzymes for biotechnological applications. The data
presented are a step forward toward alternative in vitro tests which
will become more predictive of the in vivo situation and should be
used wherever possible to replace animal models for rapid assess-
ment of drug biotransformation and toxicity [41].
[9] V.R. Dodhia, A. Fantuzzi, G. Gilardi, J. Biol. Inorg. Chem. 11 (2006) 903–916.
[10] A. Fantuzzi, M. Fairhead, G. Gilardi, J. Am. Chem. Soc. 28 (2004) 5040–5041.
[11] D.L. Johnson, B.C. Lewis, D.J. Elliot, J.O. Miners, L.L. Martin, Biochem. Pharmacol. 15
(2005) 1533–1541.
[12] V.R. Dodhia, C. Sassone, A. Fantuzzi, G. Di Nardo, S.J. Sadeghi, G. Gilardi, Electrochem.
Commun. 10 (2008) 1744–1747.
[13] S.J. Sadeghi, S. Fererro, G. Di Nardo, G. Gilardi, Bioelectrochemistry 86 (2012)
87–91.
[14] C. Helvig, J.H. Capdevila, Biochemistry 39 (2000) 5196–5205.
[15] J. Spratlin, M.B. Sawyer, Crit. Rev. Oncol. Hematol. 61 (2007) 222–229.
[16] X.Q. Li, A. Björkman, T.B. Andersson, M. Ridderström, C.M. Masimirembwa,
J. Pharmacol. Exp. Ther. 300 (2002) 399–407.
[17] T. Omura, R. Sato, J. Biol. Chem. 239 (1964) 2370–2378.
[18] C.L. Crespi, T.K. Chang, D.J. Waxman, Methods Mol. Biol. 320 (2006) 103–107.
[19] M. Ntale, M. Mahindi, J.W. Ogwal-Okeng, L.L. Gustafsson, O. Beck, J. Chromatogr. B
Analyt. Technol. Biomed. Life Sci. 859 (2007) 137–140.
[20] S. Castrignanò, S.J. Sadeghi, G. Gilardi, Anal. Bioanal. Chem. 398 (2010)
1403–1409.
[21] V.E. Ferrero, L. Andolfi, G. Di Nardo, S.J. Sadeghi, A. Fantuzzi, S. Cannistraro, G.
Gilardi, Anal. Chem. 80 (2008) 8438–8446.
[22] S.J. Sadeghi, R. Meirinhos, G. Catucci, V.R. Dodhia, G. Di Nardo, G. Gilardi, J. Am.
Chem. Soc. 132 (2009) 458–459.
[23] L.H. Mak, S.J. Sadeghi, A. Fantuzzi, G. Gilardi, Anal. Chem. 82 (2010) 5357–5362.
[24] G.A. Schoch, J.K. Yano, S. Sansen, P.M. Dansette, C.D. Stout, E.F. Johnson, J. Biol.
Chem. 20 (2008) 17227–17237.
[25] E. Krieger, G. Vriend, Bioinformatics 18 (2002) 315–318.
[26] R.A. Laskowski, J.A. Rullmannn, M.W. MacArthur, R. Kaptein, J.M. Thornton, J. Biomol.
NMR 8 (1996) 477–486.
[27] T. Cresteil, B. Monsarrat, J. Dubois, M. Sonnier, P. Alvinerie, F. Gueritte, Drug
Metab. Dispos. 30 (2002) 438–445.
[28] A. Rahman, K.R. Korzekwa, J. Grogan, F.J. Gonzalez, J.W. Harris, Cancer Res. 54
(1994) 5543–5546.
[29] R.L. Walsky, E.A. Gaman, R.S. Obach, J. Clin. Pharmacol. 45 (2005) 68–78.
[30] N. Hanioka, K. Matsumoto, Y. Saito, S. Narimatsu, Basic Clin. Pharmacol. Toxicol.
107 (2010) 565–569.
Acknowledgements
[31] S.J. Sadeghi, A. Fantuzzi, G. Gilardi, Biochim. Biophys. Acta 1814 (2011) 237–248.
[32] A. Fantuzzi, L.H. Mak, E. Capria, V.R. Dodhia, P. Panicco, S. Collins, G. Gilardi, Anal.
Chem. 15 (2011) 3831–3839.
This work was supported by the Region Piedmont CIPE 2006 (CYP-
TECH project, Italy) and Nanobiodesign s.r.l. (Italy).
[33] P. Panicco, V.R. Dodhia, A. Fantuzzi, G. Gilardi, Anal. Chem. 15 (2011) 2179–2186.
[34] E. Laviron, J. Electroanal. Chem. 101 (1979) 19–23.
[35] X. Cao, Y. Ye, S. Liu, Anal. Biochem. 417 (2011) 1–16.
[36] B.D. Fleming, Y. Tian, S.G. Bell, L.L. Wong, V. Urlacher, H.A.O. Hill, Eur. J. Biochem.
270 (2003) 4082.
References
[1] J.E. May, J. Xu, H.R. Morse, N.D. Avent, C. Donaldson, Br. J. Biomed. Sci. 66 (2009)
160–165.
[2] O. Varga, A.K. Hansen, P. Sandøe, I.A. Olsson, EMBO Rep. 11 (2010) 500–503.
[3] F.P. Guenguerich, Chem. Res. Toxicol. 21 (2008) 70–83.
[4] M. Ebeling, E. Küng, A. See, C. Broger, G. Steiner, M. Berrera, T. Heckel, L. Iniguez, T.
Albert, R. Schmucki, H. Biller, T. Singer, U. Certa, Genome Res. 21 (2011) 1746–1756.
[5] X.S. Lai, L.P. Yang, X.T. Li, J.P. Liu, Z.W. Zhou, S.F. Zhou, Curr. Drug Metab. 10
(2009) 1009–1047.
[37] A.K. Udit, N. Hindoyan, M.G. Hill, F.H. Arnold, H.B. Gray, Inorg. Chem. 44 (2005)
4109–4111.
[38] Y.O. Rudakov, V.V. Shumyantseva, T.V. Bulko, E.V. Suprun, G.P. Kuznetsova, N.F.
Samenkova, A.I. Archakov, J. Inorg. Biochem. 102 (2008) 2020–2025.
[39] S.Y. Rhieu, D.R. Ludwig, V.S. Siu, T.R. Palmore, Electrochem. Commun. 11 (2009)
1857–1860.
[6] Y. Uno, H. Fujino, G. Kito, T. Kamataki, R. Nagata, Mol. Pharmacol. 70 (2006)
477–486.
[7] F. Roussel, D.B. Duignan, M.P. Lawton, R.S. Obach, C.A. Strick, D.J. Tweedie, Arch.
Biochem. Biophys. 357 (1998) 27–36.
[40] D.L. Johnson, A.J. Conley, L.L. Martin, J. Mol. Endocrinol. 36 (2006) 349–359.
[41] M. Martignoni, G.M. Groothuis, R. de Kanter, Expert Opin. Drug Metab. Toxicol. 2
(2006) 875–894.

 Rua, Francesco
Rua, Francesco
 Sadeghi, Sheila J.
Sadeghi, Sheila J.
 Castrignanò, Silvia
Castrignanò, Silvia
 Di Nardo, Giovanna
Di Nardo, Giovanna
 Gilardi, Gianfranco
Gilardi, Gianfranco