
Journal of Pharmaceutical Sciences p. 497 - 501 (1980)
Update date:2022-08-10
Topics:
 Testa
Testa
 Salvesen
Salvesen
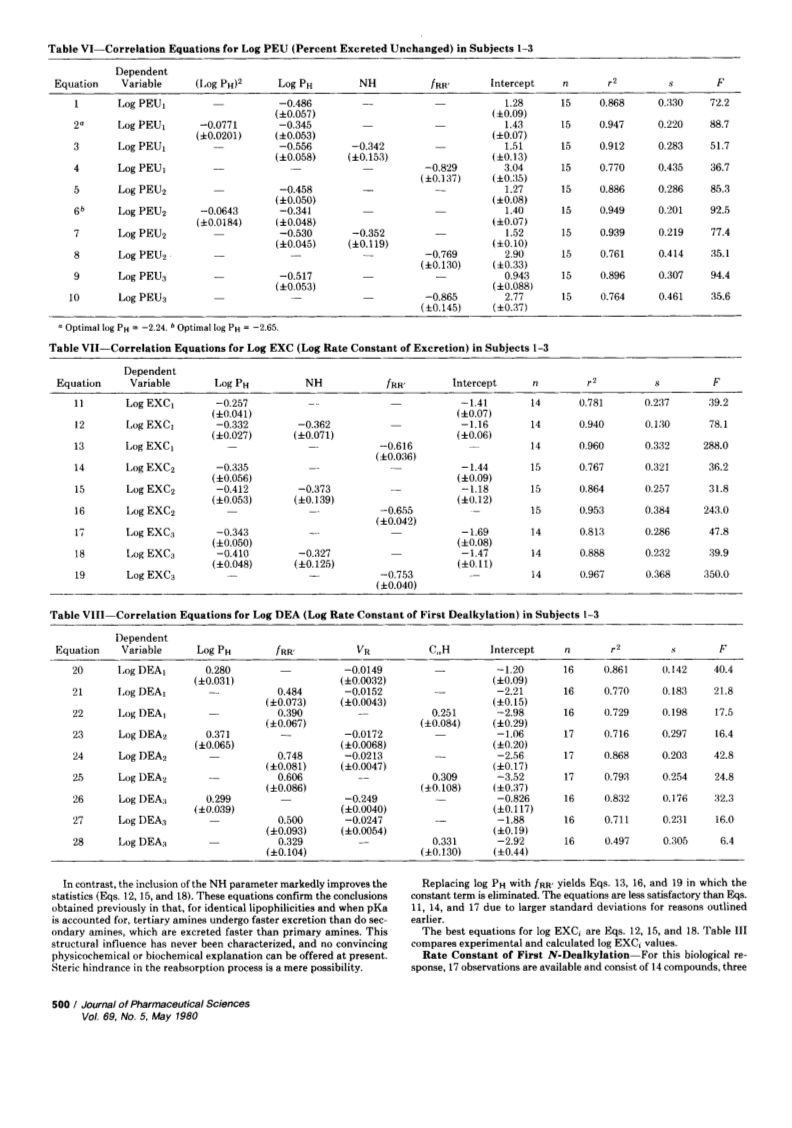
Pharmacokinetic data of 15 N-alkyl-substituted amphetamines in humans have been the object of a retrospective quantitative structure-activity relationship study. The urinary excretion of amphetamines was shown to decrease with increasing lipophilicity; the correlation equations revealed that, for identical lipophilicities, tertiary amines are excreted faster than secondary amines, which are secreted faster than primary amines. The apparent n-heptane-pH 7.4 buffer partition coefficient correlates better with urinary excretion than does the true n-octanol-water partition coefficient, probably because it includes a pKa term that accounts for the fraction of the drug present in the tubules as nonionic species. The N-dealkylation rate increases with increasing lipophilicity of the substrates (enhanced enzyme affinity) but decreases with increasing bulk of the N-substituent that is split off (steric hindrance of initial C(α)-hydroxylation.
View More



Doi:10.1016/S0040-4039(98)02436-8
(1999)Doi:10.1002/anie.200905115
(2009)Doi:10.1002/jps.2600740709
(1985)Doi:10.1002/jhet.5570440642
(2007)Doi:10.1021/om00115a025
(1990)Doi:10.1246/bcsj.20190105
(2019)