ORGANIC PREPARATIONS AND PROCEDURES INTERNATIONAL
EXPERIMENTAL PAPER
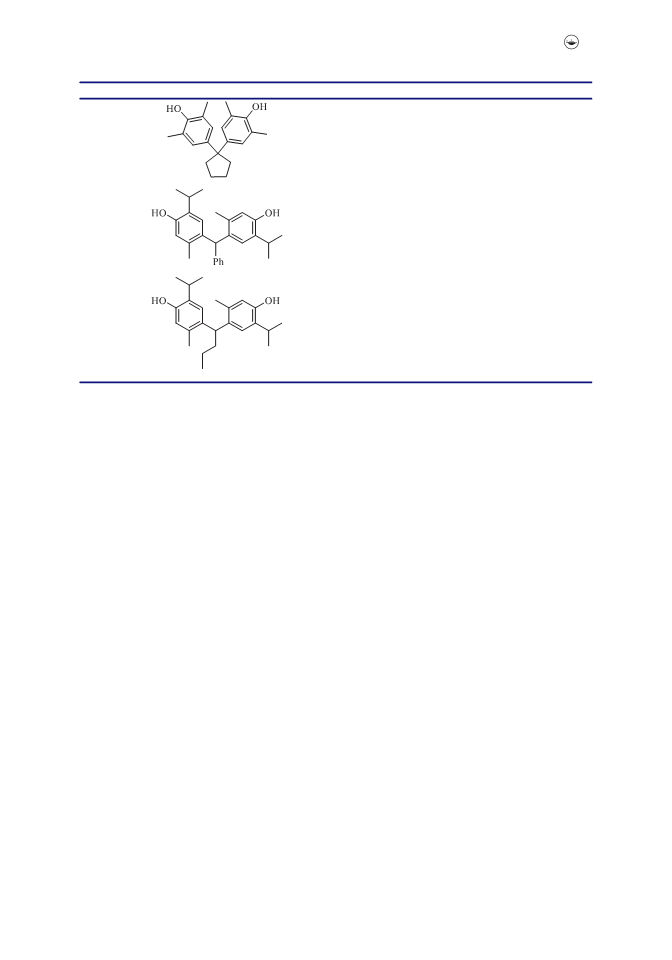
Eco-friendly Solvent-free Route to Alkyl- and Aryl-Bisphenols
Catalyzed by Perchloric Acid-Silica
Deepak Singha
and Pradeep T. Deotab
b
aHankuk University of Foreign Studies, Seoul, South Korea; Applied Chemistry Department, Faculty of
Technology and Engineering, The Maharaja Sayajirao University of Baroda, Vadodara, India
ARTICLE HISTORY Received 15 July 2019; Accepted 8 January 2020
Bisphenols, generally formed by condensation of phenols with carbonyl compounds,
have found several useful applications.1–7 The literature records a number of methods
for synthesis of bisphenols using a variety of catalysts such as mineral acids, bases, mes-
oporous silica, zeolites, and montmorillonite clays.8–13 As useful as they have been,
however, some of these methods have their limitations, such as the corrosive nature of
some of the reagents, the need of an inert atmosphere, harsh reaction conditions or
cumbersome procedures. Despite extensive work on the synthesis of bisphenols, there is
still a need for improvement.
In last few decades, solid acid catalysts have gained much importance in synthetic
chemistry as they are eco-friendly, inexpensive and safe. They offer high efficiency of
conversion under mild conditions.14 In particular, silica-supported perchloric acid
(HClO4-SiO2) is one such system which is employed in a variety of organic transforma-
tions15 such as acylation,16 esterification,17 rearrangements,18 and Michael addition19
among many others.20–31
Herein we describe a convenient and inexpensive method for the preparation of
bisphenols via condensation of phenols with aldehydes or ketones under solvent-free
conditions (Scheme 1). The moisture-stable catalyst (50 mg, 0.025 mol Hþ)5 was easily
prepared and used for our synthesis of bisphenols. To examine the catalytic activity of
this supported reagent over aqueous HClO4, we carried out a model study with 2,6-
dimethylphenol (1) and formaldehyde (2) using 1 mol % of the catalyst at room tem-
perature under solvent-free conditions.
In order to find the optimum quantity of the silica supported perchloric acid, the
reaction of 2,6-dimethylphenol and formaldehyde was carried out under varying
amounts of the catalyst. Silica supported perchloric acid at 2 mol % gave an excellent
yield in 4 h as shown in Table 2. The results shown in Table 3 suggest that silica sup-
ported perchloric acid catalyst is an efficient catalyst in terms of both time and yield,
under solvent free conditions.
For optimizing the temperature of the reaction, we carried out a model study with
2,6-dimethylphenol and formaldehyde with 2 mol % of the catalyst at three different
temperatures under solvent free conditions (Table 4).
CONTACT Pradeep T. Deota
Applied Chemistry Department, Faculty of Technology and
Engineering, The Maharaja Sayajirao University of Baroda, Vadodara, 390001, India
ß 2020 Taylor & Francis Group, LLC

 Deota, Pradeep T.
Deota, Pradeep T.
 Singh, Deepak
Singh, Deepak