Bioorganic & Medicinal Chemistry Letters
Synthesis, in vitro antibacterial activities of a series
of 3-N-substituted canthin-6-ones
y
y
⇑
Jiang-Kun Dai , Wen-Jia Dan , Na Li, Hong-Tao Du, Ji-Wen Zhang, Jun-Ru Wang
College of Science, Northwest A&F University, Yangling 712100, Shaanxi, China
a r t i c l e i n f o
a b s t r a c t
Article history:
An improved synthetic route of canthin-6-one was accomplished. To further enhance the antibacterial
potency and improve water solubility, a series of 3-N-alkylated and 3-N-benzylated canthin-6-ones were
designed and synthesized, and their in vitro antibacterial activities were evaluated. A clear structure–
Received 16 June 2015
Revised 2 November 2015
Accepted 20 November 2015
Available online xxxx
activity relationship with peak minimal inhibitory concentration (MIC) values of 0.98 (
l
gꢀmLꢁ1) was
investigated. Particularly, compounds 6i–r and 6t were found to be the most potent compounds with
minimal inhibitory concentration (MIC) values lower than 1.95 (
l
gꢀmLꢁ1) against Staphylococcus aureus.
Keywords:
Synthesis
Ó 2015 Elsevier Ltd. All rights reserved.
Canthin-6-one
3-N-alkylation
3-N-benzylation
Antibacterial
Staphylococcus aureus
Structure–activity relationship
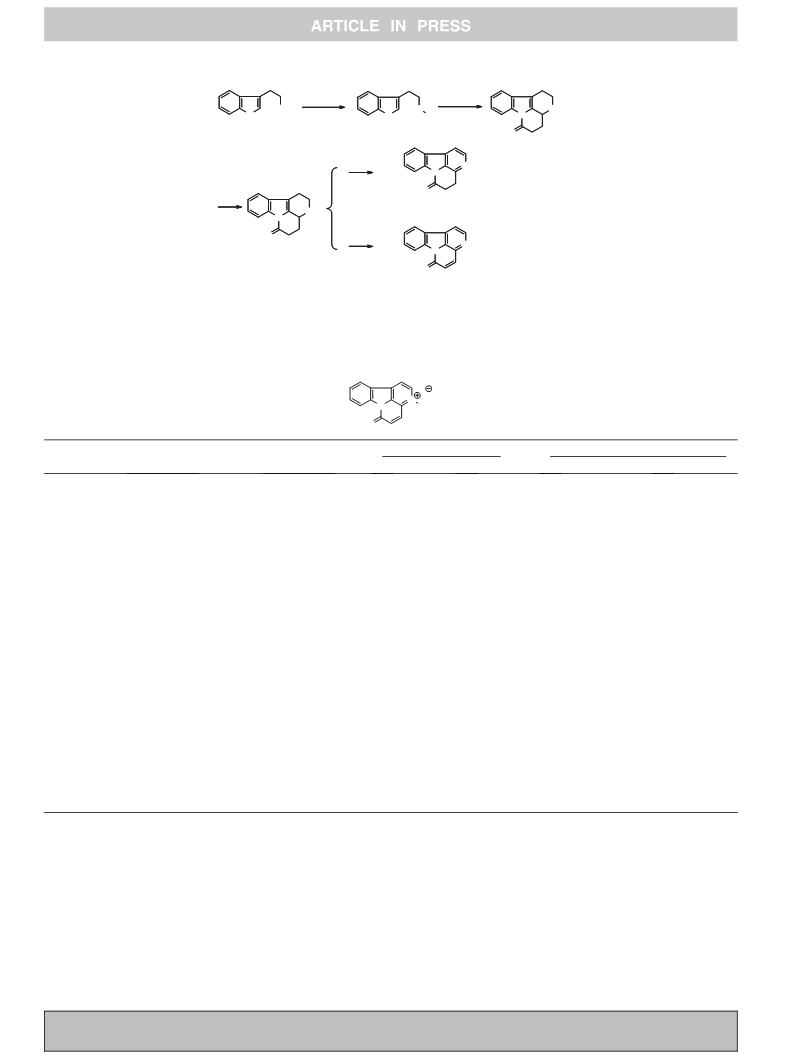
The canthin-6-one is a subclass of b-carboline alkaloids with an
additional D-ring, which have been isolated from various sources,
principally including Rutaceae, Simaroubaceae families and the
fungi.1,2 Simultaneously, canthin-6-one alkaloids are pharmacolog-
ically active natural products, which have shown to possess a
wide-range biological activities consisting of cytotoxic, antiviral,
antimicrobial, anti-inflammatory, anti-parasitic, anticancer, and
enzyme inhibitory.3–9 Based on its potentiality in drug develop-
ment, a growing number of scientists have focused on it. Specially,
its total synthesis continues to the present day.10–12 Gollner et al.
reported a ‘non-classical’ high yield (95%) route which relies on a
concomitant Pd-catalyzed Suzuki–Miyaura C–C coupling followed
by a Cu-catalyzed C–N coupling in 2010.13 Their route is the most
easy and low-cost strategy to date. However, most of these routes
are tedious and harsh, which are not suitable for the preparation of
canthin-6-one. In order to obtain more substrate for drug develop-
ment under mild conditions, our group has explored a relatively
simple method through improved the previous approach.14
Pseudomonas solanacearum are major components of plant
pathogens.17 We could see that all of these diseases caused by bac-
teria constitute a major threat to humans’ life and property.
Although penicillin is currently the first choice of antibiotics to
treat bacterial infections, the overuse of penicillin has resulted in
a situation that higher dose is required to successfully treat the
bacterial infection.18 Many studies have showed that compounds
with large planar surface areas could function at the DNA level
via intercalation between base pairs.19 Moreover, it has been
proved that DNA intercalation of compound could disrupt the
function of DNA topoisomerases and lead to cell death ulti-
mately.20 First described in 2014 by Dejos et al., canthin-6-one
could decrease the synthesis of DNA and possess the properties
of antiproliferative.21 In 2007, O’Donnell et al. reported the
antibacterial activity of canthin-6-one alkaloids which displayed
minimum inhibitory concentrations (MICs) in the range of
8–82
l
gꢀmLꢁ1 against a panel of fast-growing Mycobacterium spe-
cies and 8–64
l
gꢀmLꢁ1 against multidrug-resistant (MDR) and
Staphylococcus aureus is one of the leading causes of bacterial
infections in humans with symptoms ranging from simple skin
infections to severe necrotizing fasciitis and pneumonia.15 Bacillus
cereus could cause food poisoning.16 Ralstonia solanacearum and
methicillin-resistant (MRSA) strains of Staphylococcus aureus.22 At
the same time, Ostrov et al. used structure-based molecular dock-
ing to identify novel drug-like small molecules. They found that
canthin-6-one could not effectively accumulate inside of the
Escherichia coli might be due to drug instability or metabolic inac-
tivation by bacterial enzymes, while it could inhibit the DNA
supercoiling activity of purified E. coli DNA gyrase.8 Based on the
above studies, canthin-6-one which has a highly conjugated large
⇑
Corresponding author. Tel./fax: +86 29 8709 2829.
First two authors contributed equally to this work.
y
0960-894X/Ó 2015 Elsevier Ltd. All rights reserved.

 Dai, Jiang-Kun
Dai, Jiang-Kun
 Dan, Wen-Jia
Dan, Wen-Jia
 Li, Na
Li, Na
 Du, Hong-Tao
Du, Hong-Tao
 Zhang, Ji-Wen
Zhang, Ji-Wen
 Wang, Jun-Ru
Wang, Jun-Ru