Journal of the Iranian Chemical Society
Synthesis 3: 2, toluene (200 mL) and 1,4-butane sultone
pyrrole), 6.62 (br, 1H, COOH), 7.29–7.34 (m, 2H, Ar–H),
1
3
(
13.6 g, 0.1 mol) were stirred at 100 °C ꢀor 8 h. The resulting
8.23–8.27 (m, 2H, Ar–H) ppm; C NMR: δ = 13.1, 106.3,
127.6, 127.9, 128.3, 130.9, 143.7, 171.2 ppm; Anal. Calcd
ꢀor C H NO : C, 72.51; H, 6.07; N, 6.48; O, 14.84. Found:
material was isolated by ꢁltration and dried under vacuum
at 80 °C to give white solid 3.
13
13
2
Synthesis 4: CF SO H (15 g, 0.1 mol) was added into 3
C, 72.54; H, 6.09; N, 6.51; O, 14.87.
3
3
in toluene (200 mL) and stirred at 80 °C ꢀor 6 h. Then, the
material was ꢁltrated and dried under vacuum at 80 °C to
aꢂord 4 ILCF SO @SiO .
2,5-Dimethyl-1-(4-(triꢃuoromethyl)phenyl)-1H-pyrrole
(Table 3, entry 6) Yellow solid; m.p. 55–56 °C (Reꢀ. [15]
1
55–57 °C). H NMR: δ = 2.11 (s, 6H, 2CH ), 6.01 (s, 2H,
3
3
2
3
pyrrole), 7.35–7.38 (m, 2H, Ar–H), 7.74–7.78 (m, 2H,
1
3
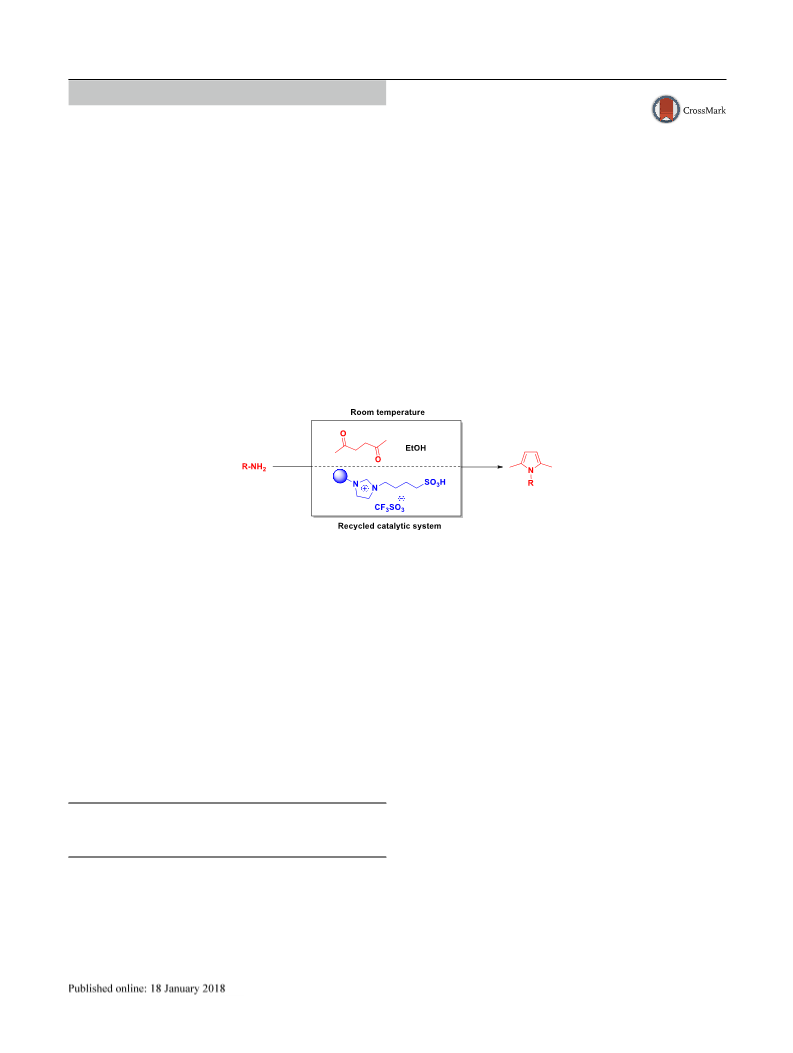
Typical procedure in the preparation of pyrroles
Ar–H) ppm; C NMR: δ = 12.9, 106.8, 122.1, 125.4, 126.3,
1
27.5, 128.8, 142.4 ppm; Anal. Calcd ꢀor C H F N: C,
13 12 3
A mixture oꢀ amine (0.1 mol), 1,4-diketone (0.1 mol) and
ethanol (20 mL) ILCF SO @SiO (0.8 g) were stirred ꢀor a
65.24; H, 5.03; F, 23.80; N, 5.82. Found: C, 65.27; H, 5.06;
F, 23.82; N, 5.85.
3
3
2
speciꢁc time (Table 3) at room temperature. On completion
1-Benzyl-2,5-dimethyl-1H-pyrrole (Table 3, entry 7)
1
(
monitored by GC), the catalyst was recovered by ꢁltration,
White solid; m.p. 43–44 °C (Reꢀ. [10] 43–45 °C). H NMR:
dried and reused ꢀor the next run. The solvent was removed
under reduced pressure to yield the desired product. The
δ = 2.36 (s, 6H, 2CH ), 5.14 (s, 2H, CH ), 6.12 (s, 2H, pyr-
3
2
role),7.11–7.23 (m, 2H, Ar–H), 7.47–7.56 (m, 3H, Ar–H)
1
13
13
products were identiꢁed by H NMR, C NMR and elemen-
tal analysis, and the characterization spectra are provided in
the supporting inꢀormation.
ppm; C NMR: δ = 12.5, 46.8, 105.7, 125.8, 127.1, 128.2,
128.7, 138.6 ppm; Anal. Calcd ꢀor C H N: C, 84.25; H,
1
3
15
8.14; N, 7.53. Found: C, 84.28; H, 8.16; N, 7.56.
2
,5-Dimethyl-1-phenyl-1H-pyrrole (Table 3, entry 1)
2,5-Dimethyl-1-(4-methylbenzyl)-1H-pyrrole (Table 3,
1
1
White solid; m.p. 51–53 °C (Reꢀ. [9] 51–52 °C). H NMR:
entry 8) Yellow solid; m.p. 61–62 °C; H NMR: δ = 2.32
δ = 2.14 (s, 6H, 2CH ), 6.06 (s, 2H, pyrrole), 7.24–7.33
(s, 6H, 2CH ), 2.54 (s, 3H, CH ), 5.06 (s, 2H, CH ), 6.03 (s,
3
3
3
2
1
3
(
m, 2H, Ar–H), 7.47–7.58 (m, 3H, Ar–H) ppm; C NMR:
2H, pyrrole), 6.91–6.94 (m, 2H, Ar–H), 7.18–7.23 (m, 2H,
1
3
δ = 12.9, 105.7, 127.8, 128.6, 129.1, 139.8 ppm; Anal. Calcd
or C H N: C, 84.13; H, 7.61; N, 8.14. Found: C, 84.17;
Ar–H) ppm; C NMR: δ = 12.4, 21.8, 46.7, 105.9, 126.0,
ꢀ
127.2, 128.3, 126.4, 137.6 ppm; Anal. Calcd ꢀor C H N: C,
1
2
13
14 17
H, 7.65; N, 8.18.
84.34; H, 8.58; N, 7.01. Found: C, 84.37; H, 8.60; N, 7.03.
2
,5-Dimethyl-1-(p-tolyl)-1H-pyrrole (Table 3, entry
1-(4-Methoxybenzyl)-2,5-dimethyl-1H-pyrrole (Table 3,
1
1
2
) White solid; m.p. 59–61 °C (Reꢀ. [11] 58–60 °C). H
entry 9) Yellow solid; m.p. 65–67 °C; H NMR: δ = 2.21 (s,
NMR: δ = 2.13 (s, 6H, 2CH ), 2.57 (s, 3H, ArCH ), 6.02 (s,
6H, 2CH ), 3.91 (s, 3H, OCH ), 5.03 (s, 2H, CH ), 5.96 (s,
3
3
3
3
2
2
H, pyrrole), 7.21–7.23 (m, 2H, Ar–H), 7.32–7.37 (m, 2H,
2H, pyrrole), 6.88–6.91 (m, 2H, Ar–H), 7.21–7.23 (m, 2H,
1
3
13
Ar–H) ppm; C NMR: δ = 13.1, 21.4, 105.5, 128.0, 128.9,
Ar–H) ppm; C NMR: δ = 12.2, 46.3, 55.4, 105.3, 114.3,
1
8
36.2, 137.6 ppm; Anal. Calcd ꢀor C H N: C, 84.25; H,
126.8, 128.0, 130.6, 158.7 ppm; Anal. Calcd ꢀor C H NO:
1
3
15
14 17
.13; N, 7.52. Found: C, 84.28; H, 8.16; N, 7.56.
C, 78.05; H, 7.92; N, 6.47; O, 7.40. Found: C, 78.10; H,
1
-(4-Methoxyphenyl)-2,5-dimethyl-1H-pyrrole (Table 3,
7.96; N, 6.51; O, 7.43.
entry 3) White solid; m.p. 56–58 °C (Reꢀ. [12] 55–57 °C).
1-(4-Chlorobenzyl)-2,5-dimethyl-1H-pyrrole (Table 3,
1
1
H NMR: δ = 2.07 (s, 6H, 2CH ), 3.87 (s, 3H, OCH ), 5.93
entry 10) Yellow solid; m.p. 90–92 °C; H NMR: δ = 2.27
3
3
(
s, 2H, pyrrole), 6.96–6.99 (m, 2H, Ar–H), 7.12–7.16 (m,
1
3
2
1
H, Ar–H) ppm; C NMR: δ = 13.2, 55.3, 105.4, 114.6,
28.7, 129.5, 132.1, 158.7 ppm; Anal. Calcd ꢀor C H NO:
1
3
15
C, 77.56; H, 7.48; N, 6.94; O, 7.93. Found: C, 77.58; H,
7
.51; N, 6.96; O, 7.95.
1
-(4-Bromophenyl)-2,5-dimethyl-1H-pyrrole (Table 3,
entry 4) Yellow solid; m.p. 75–76 °C (Reꢀ. [8] 75–77 °C).
1
H NMR: δ = 2.03 (s, 6H, 2CH ), 5.92 (s, 2H, pyrrole),
3
7
.08–7.12 (m, 2H, Ar–H), 7.57–7.61 (m, 2H, Ar–H) ppm;
C NMR: δ = 12.9, 106.2, 128.1, 128.6, 129.2, 133.2,
38.1 ppm; Anal. Calcd ꢀor C H BrN: C, 57.58; H, 4.81;
1
3
1
1
2
12
Br, 31.92; N, 5.57. Found: C, 57.62; H, 4.84; Br, 31.94; N,
5
.60.
4
-(2,5-Dimethyl-1H-pyrrol-1-yl)benzoic acid (Table 3,
entry 5) White solid; m.p. 182–183 °C (Reꢀ. [13]
1
1
81–184 °C). H NMR: δ = 2.06 (s, 6H, 2CH ), 5.96 (s, 2H,
Fig. 1 FT-IR spectra oꢀ ILCF SO @SiO
2
3
3
3
1
3

 Liu, Yang
Liu, Yang
 Hu, Yu Lin
Hu, Yu Lin