Please do not adjust margins
ChemComm
Page 4 of 4
COMMUNICATION
Journal Name
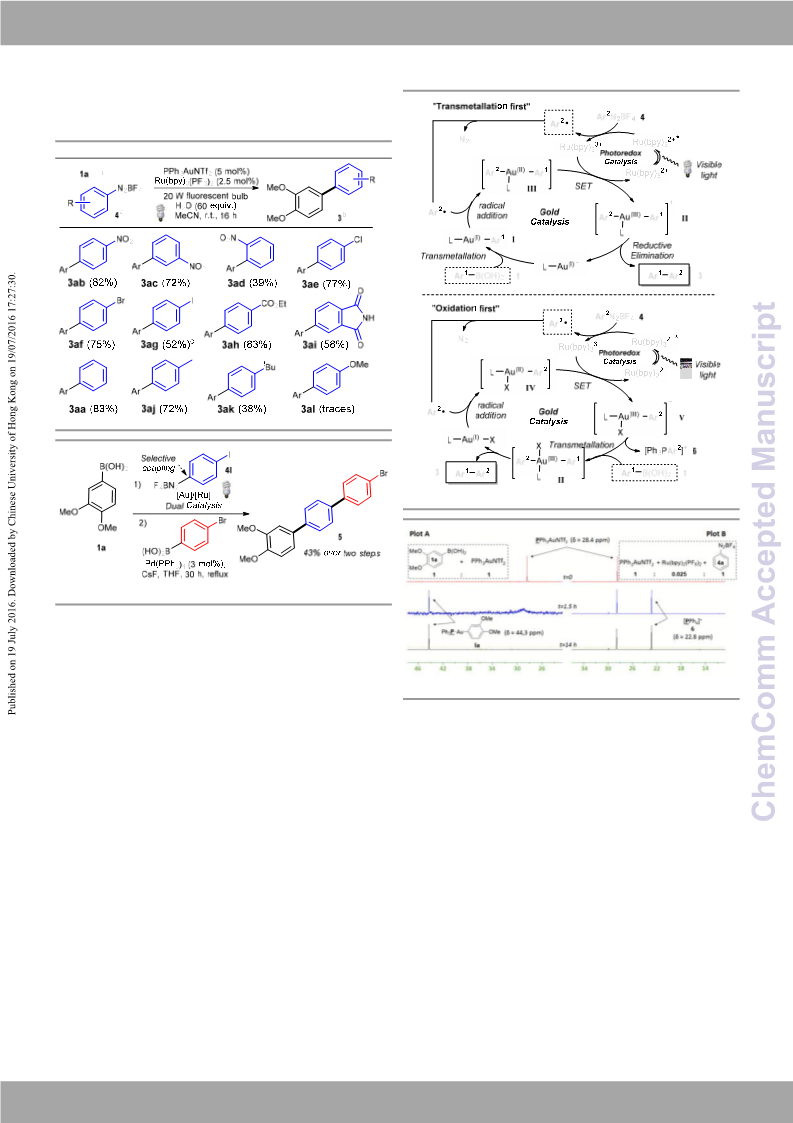
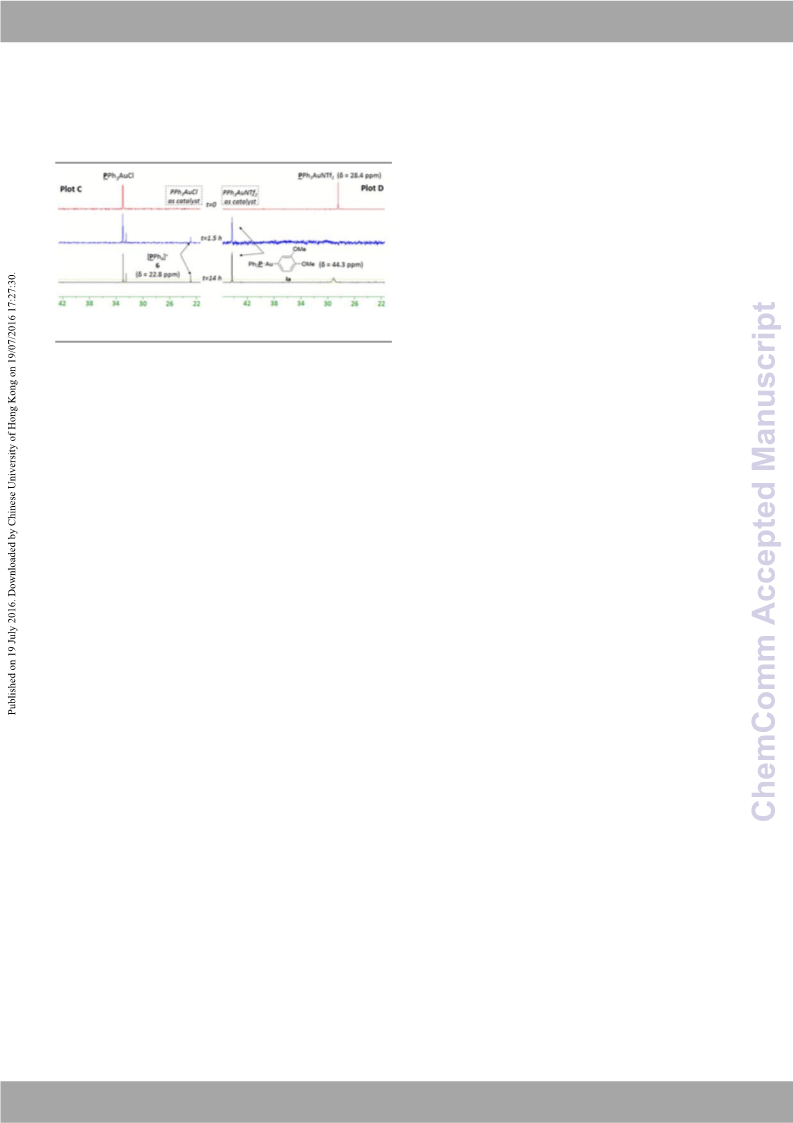
1H NMR analysis clearly showed the formation of coupling product
3aa over the same time period. These results suggest that the
“transmetallation first” catalytic cycle is the most plausible
mechanism when cationic PPh3AuNTf2 is used as catalyst.
7
8
DOI: 10.1039/C6CC05078F
Catal., 2015, 357, 2622.
For selected recent reviews on visible light photoredox
catalysis, see: (a) T. P. Yoon, M. A. Ischay and J. N. Du, Nat.
Chem., 2010, 2, 527; (b) C. K. Prier, D. A. Rankic and D. W.
C. MacMillan, Chem. Rev., 2013, 113, 5322; (c) M. N.
Hopkinson, B. Sahoo, J.‐L. Li and F. Glorius, Chem. Eur. J.,
2014, 20, 3874; (d) D. M. Schultz and T. P. Yoon, Science,
2014, 343; (e) E. Meggers, Chem. Commun., 2015, 51
,
3290; (f) D. A. Nicewicz and T. M. Nguyen, ACS Catalysis,
2013,
2013,
4
, 355; (g) S. Fukuzumi and K. Ohkubo, Chem. Sci.,
4
, 561.
9
For review on cooperative photoredox catalysis, see: X.
Lang, J. Zhao and X. Chen, Chem. Soc. Rev., 2016, 45, 3026.
10 For an elegant alternative; photoredox using only Au
catalysts with blue LEDs by Hashmi and Barriault: (a) L.
Huang, M. Rudolph, F. Rominger and A. S. K. Hashmi,
Angew. Chem. Int. Ed., 2016, 55, 4808; (b) G. Revol, T.
McCallum, M. Morin, F. Gagosz and L. Barriault, Angew.
Chem. Int. Ed., 2013, 52, 13342; (c) L. Huang, F. Rominger,
Figure 2 31P NMR monitoring of PPh3AuCl‐catalysed reaction (plot
PPh3AuNTf2‐catalysed reaction (plot
C) and
D
)
In conclusion, we have developed the first application of
dual gold/photoredox catalysis for C(sp2)‐C(sp2) coupling.
Successful cross‐coupling was found to significantly depend on
the amount of water added. While studies have shown that
M. Rudolph and A. S. K. Hashmi, Chem. Commun., 2016; 52
,
6435; (d) J. Xie, S. Shi, T. Zhang, N. Mehrkens, M. Rudolph
and A. S. K. Hashmi, Angew. Chem. Int. Ed., 2015, 54, 6046.
11 Although it is known with ligand‐ and base‐assisted redox
gold‐catalysis (mechanistically distinct from photoredox):
(a) R. Cai, M. Lu, E. Y. Aguilera, Y. Xi, N. G. Akhmedov, J. L.
Petersen, H. Chen and X. Shi, Angew. Chem. Int. Ed., 2015,
54, 8772. See also Pd‐catalysis review: H. Bonin, E. Fouquet
and F.‐X. Felpin, Adv. Synth. Catal., 2011, 353, 3063.
oxidation of Au(I) to Au(III) species
V is the first step in several
dual gold/photoredox catalysed reactions, our mechanistic
studies reveal that the mechanistic sequence is in fact highly
catalyst dependent. Cationic PPh3AuNTf2 actually undergoes
transmetallation with arylboronic acid prior to oxidation of
Au(I) to Au(III), whereas neutral PPh3AuCl undergoes the
expected “oxidation first” pathway.
We thank the Leverhulme Trust (RPG‐2014‐345) for
funding. Mass spectrometry data was acquired at the EPSRC
UK National Mass Spectrometry Facility at Swansea University.
12 During the preparation of this manuscript, Barriault
disclosed
coupling to alkylate heterocycles: T. McCallum and L.
Barriault, Chem. Sci., 2016, , 4757.
a
photoredox gold catalysed C(sp3)‐C(sp2)
7
13 G. Barker, S. Webster, D. G. Johnson, R. Curley, M.
Andrews, P. C. Young, S. A. Macgregor and A.‐L. Lee, J. Org.
Chem., 2015, 80, 9807.
14 Stoichiometric: (a) D. V. Partyka, M. Zeller, A. D. Hunter
and T. G. Gray, Angew. Chem. Int. Ed., 2006, 45, 8188.
15 See also: M. D. Levin and F. D. Toste, Angew. Chem. Int.
Ed., 2014, 53, 6211.
References
1
Selected reviews: (a) A. Fürstner and P. W. Davies, Angew.
Chem., Int. Ed., 2007, 46, 3410; (b) D. J. Gorin and F. D.
Toste, Nature, 2007, 446, 395; (c) A. S. K. Hashmi and G. J.
Hutchings, Angew. Chem., Int. Ed., 2006, 45, 7896; (d) A. S.
K. Hashmi, Chem. Rev., 2007, 107, 3180; (e) D. J. Gorin, B.
D. Sherry and F. D. Toste, Chem. Rev., 2008, 108, 3351; (f)
C. Obradors and A. M. Echavarren, Chem. Commun., 2014,
50, 16; (g) D. Pflasterer and A. S. K. Hashmi, Chem. Soc.
Rev., 2016, 45, 1331.
16 Reductive elimination steps involving Au(III) can be
extremely fast, see: W. J. Wolf, M. S. Winston and F. D.
Toste, Nat. Chem., 2014, 6, 159.
17 N. Mezailles, L. Ricard and F. Gagosz, Org. Lett., 2005, 7,
4133.
18 This is the same trend observed previously with
aryldiazonium salts, see ref 5.
2
For review, see: M. N. Hopkinson, A. D. Gee and V.
Gouverneur, Chem. Eur. J., 2011, 17, 8248.
S. G. Bratsch, J. Phys. Chem. Ref. Data, 1989, 18, 1.
19 (a) M. Livendahl, C. Goehry, F. Maseras and A. M.
Echavarren, Chem. Commun., 2014, 50, 1533; (b) M. Joost,
A. Zeineddine, L. Estévez, S. Mallet−Ladeira, K. Miqueu, A.
3
4
For selected examples of gold‐catalysed oxidative coupling
of arenes, see: (a)L. T. Ball, G. C. Lloyd‐Jones and C. A.
Russell, Science, 2012, 337, 1644; (b) X. C. Cambeiro, N.
Ahlsten and I. Larrosa, J. Am. Chem. Soc., 2015, 137, 15636;
(c) Q. Wu, C. Du, Y. Huang, X. Liu, Z. Long, F. Song and J.
Amgoune and D. Bourissou, J. Am. Chem. Soc., 2014, 136
,
14654; (c) A. S. K. Hashmi, C. Lothschütz, R. Döpp, M.
Ackermann, J. De Buck Becker, M. Rudolph, C. Scholz and F.
Rominger, Adv. Synth. Catal., 2012, 354, 133.
20 M. Schiek, K. Al‐Shamery and A. Lutzen, Synthesis, 2007,
613.
You, Chem. Sci., 2015, 6, 288; (d) A.Kar, N. Mangu, H. M.
Kaiser, M. Beller and M. K. Tse, Chem. Commun., 2008,
386.
(a) M. N. Hopkinson, B. Sahoo and F. Glorius, Adv. Synth.
Catal., 2014, 356, 2794; (b) B. Sahoo, M. N. Hopkinson and
F. Glorius, J. Am. Chem. Soc., 2013, 135, 5505; (c) A.
Tlahuext‐Aca, M. N. Hopkinson, B. Sahoo and F. Glorius,
21 Q. Zhang, Z.‐Q. Zhang, Y. Fu and H.‐Z. Yu, ACS Catalysis,
2016, 6, 798.
5
6
22 E. Tkatchouk, N. P. Mankad, D. Benitez, W. A. Goddard and
F. D. Toste, J. Am. Chem. Soc., 2011, 133, 14293.
23 D. Weber, T. D. Jones, L. L. Adduci and M. R. Gagne, Angew.
Chem., Int. Ed., 2012, 51, 2452.
24 D. Marcoux and A. B. Charette, J. Org Chem., 2008, 73, 590.
25 Neutral Au(III)‐boron transmetallation: M. Hofer, E.
Chem. Sci., 2016,
(a) Y. He, H. Wu and F. D. Toste, Chem. Sci., 2015,
(b) S. Kim, J. Rojas‐Martin and F. D. Toste, Chem. Sci., 2016,
, 85; (c) X.‐z. Shu, M. Zhang, Y. He, H. Frei and F. D. Toste,
J. Am. Chem. Soc., 2014, 136, 5844.
7, 89.
6
, 1194;
Gomez‐Bengoa and C. Nevado, Organometallics, 2014, 33
,
7
1328.
4 | J. Name., 2012, 00, 1‐3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Gauchot, Vincent
Gauchot, Vincent
 Lee, Ai-Lan
Lee, Ai-Lan