Chemistry of Heterocyclic Compounds 2017, 53(11), 1248–1253
7. (a) Hall, R. G.; Leff, R. D.; Gumbo, T. Pharmacotherapy
(D2O), δ, ppm (J, Hz): 3.84 (2H, dd, J = 10.8, J = 3.2,
CH2); 3.98 (2H, dd, J = 10.8, J = 4.8, CH2); 4.24 (2H, t,
J = 3.6, 2CHCH2). 13C NMR spectrum (D2O), δ, ppm: 54.3
(2C); 75.6 (2C); 167.8 (2C). Mass spectrum, m/z (Irel,%):
205 [M+H]+ (100). Found, %: C 35.30; H 5.91; N 27.45.
C6H12N4O4. Calculated, %: C 35.29; H 5.92; N 27.44.
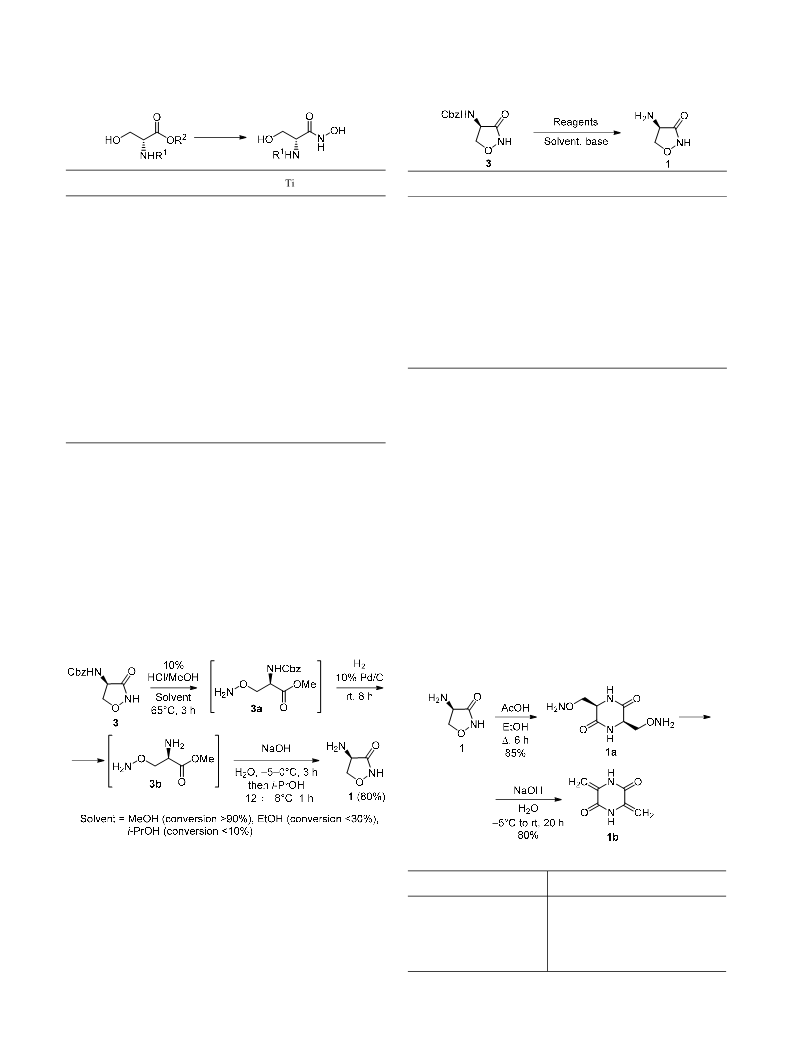
3,6-Dimethylidenepiperazine-2,5-dione (1b). A stirring
solution of compound 1a (10 g, 0.05 mol) in water (250 ml)
was cooled to –5–0°C on an ice bath. A solution of NaOH
(160 mg, 0.004 mol) in water (50 ml) was added dropwise
at –5 –0°C. The reaction mixture was warmed up to 25–30°C,
and product starts to precipitate. The reaction mixture was
stirred at 25–30°C for 20 h. Pure product was isolated by
filtration, washed with cold ethanol (5×3 ml), and dried
under reduced pressure at 50°C to afford product 1b. Yield
5.5 g (80%), white solid, mp 380–390°C (decomp.)
(mp >300°C30). IR spectrum, ν, cm–1: 1687 (C=O), 3182
(NH). 1H NMR spectrum (DMSO-d6), δ, ppm: 4.91 (2H, s,
CH2); 5.26 (2H, s, CH2); 10.89 (2H, s, 2NH). 13C NMR
spectrum (DMSO-d6), δ, ppm: 100.9 (2C); 135.1 (2C);
156.6 (2C). Mass spectrum, m/z (Irel,%): 137 [M–H]– (100).
Found, %: C 52.15; H 4.41; N 20.29. C6H6N2O2.
Calculated, %: C 52.17; H 4.38; N 20.28.
2009, 29, 1468. (b) Keshavjee, S.; Farmer, P. E. N. Engl. J.
Med. 2012, 367, 931.
8. (a) Schade, S.; Paulus, W. Int. J. Neuropsychopharmacol.
2016, 19, 102. (b) Myers, K. M.; Carlezon, W. A., Jr. Biol.
Psychiatry 2012, 71, 947.
9. Hoffman, S. G. Depression Anxiety 2014, 31, 175.
10. Connors, K. A.; Amidon, G. L.; Stella, V. J. Chemical
Stability of Pharmaceuticals: A Handbook for Pharmacists;
John Wiley & Sons: New York, 1986, 2nd ed., p. 394.
11. Kartseva ,V. D.; Bruns ,B. P.; Kruzhkova, N. G.; Bannova, N. A.
Pharm. Chem. J. 1972, 6, 615.
12. (a) Kim, H. K.; Park, K. J. J. Tetrahedron Lett. 2012, 53,
4090. (b) Plattner, P. A.; Boller, A.; Frick, H.; Furst, A.;
Hegedus, B.; Kirchensteiner, H.; Majnoni, St.; Schlapfer, R.;
Spiegelberg, H. Helv. Chim. Acta 1957, 40, 1531. (c) Ruuo, Z.;
Weitan, L.; Binjie, G. CN Patent 105646385, 2016.
13. Kim, H. K.; Park, K. J. Tetrahedron Lett. 2012, 53, 1668.
14. El-Obeid, H. A.; Al-Badr, A. A. Analytical Profiles of Drug
Substances; Elsevier, 1990, vol. 18, p. 567.
15. Li, X.; Meng, X.; Duan, H.; Wang, L.; Wang, S.; Zhang, Y.;
Qin, D. Arch. Pharm. Pharm. Med. Chem. 2010, 8, 473.
16. Isidro-Llobet, A.; Alvarez, M.; Albericio, F. Chem. Rev. 2009,
109, 2455.
17. (a) Awasthi, A. K.; Kumar, B.; Aga, M. A.; Basha, S. T.;
Reddy, C. S.; Kumar, P. ChemistrySelect 2017, 2, 9722.
(b) Awasthi, A. K.; Kumar, L.; Tripathi, P.; Golla, M.;
Aga, M. A.; Reddy, C. S.; Kumar, P. ACS Omega 2017, 2,
5460. (c) Kumar, B.; Aga, M. A.; Rouf, A.; Shah, B. A.;
Taneja, S. C. RSC Adv. 2014, 4, 17206. (d) Aga, M. A.;
Kumar, B.; Rouf, A.; Shah, B. A.; Andotra, S. S.; Taneja, S. C.
Helv. Chem. Acta 2013, 96, 969.
1
A Supplementary information file containing H and
13C NMR spectra and HPLC analysis of the synthesized
compounds is available at the journal website at http://
link.springer.com/journal/10593.
18. Greene, T. W.; Wuts, P. G. M. Protective Groups in Organic
Synthesis; John Wiley & Sons: New York, 2007, 4th еd.
19. Reddy, S.; Kumar, S.; Reddy, G. R. Tetrahedron Lett. 2000,
41, 6285.
The authors acknowledge management of Micro Labs Ltd.,
Active Pharmaceutical Ingredients Research and Develop-
ment Centre for providing research and analytical facilities.
20. Stammer, C. H.; Kartha, C. C.; Chaturvedi, N. C.;
McKinney, J. D. J. Med. Chem. 1970, 13, 1013.
References
1. Global Tuberculosis Report 2014; World Health
Organization, 2015. ISBN 978 92 4 156480 9.
2. Suthar, A. B.; Lawn, S. D.; Amo, J. D.; Getahun, H.; Dye, C.;
Sculier, D.; Sterling, T. R.; Chaisson, R. E.; Williams, B. G.;
Harries, A. D.; Granich, R. M. PLoS Med. 2012, 9, e1001270.
DOI 10.1371/journal.pmed.1001270.
21. Riva, E.; Gagliardi, S.; Mazzoni, C.; Passarella, D.;
Rencurosi, A.; Vigo, D.; Martinelli, M. J. Org. Chem. 2009,
74, 3540.
22. Giacomelli, G.; Porcheddu, A.; Salaris, M. Org. Lett. 2003, 5,
2715.
23. (a) Floyd, C. D.; Lewis, C. N.; Patel, S. R.; Whittaker, M.
Tetrahedron Lett. 1996, 37, 8045. (b) Richter, L. S.;
Desai, M. C. Tetrahedron Lett. 1997, 38, 321. (c) Chen, J. J.;
Spatola, A. F. Tetrahedron Lett. 1997, 38, 1511. (d) Mellor, S. L.;
McGuire, C.; Chan, W. C. Tetrahedron Lett. 1997, 38, 3311.
(e) Bauer, U.; Ho, W.; Koskinen, A. M. P. Tetrahedron Lett.
1997, 38, 7233. (f) Ngu, K.; Patel, D. V. J. J. Org. Chem.
1997, 62, 7088. (g) Robinson, D. E.; Holladay, M. W. Org.
Lett. 2000, 2, 2777. (h) Zhang, W.; Zhang, L.; Li, X.; Weigel, J. A.;
Hall, S. E.; Mayer, J. P. J. Comb. Chem. 2001, 3, 151.
24. United States Pharmacopoeia; 2000, 25th ed., p. 2049.
25. Ahuja, S.; Alsante K. M. Handbook of Isolation and
Characterization of Impurities in Pharmaceuticals; Academic
Press, 2003, p. 6.
3. (a) Addressing the Threat of Drug-Resistant Tuberculosis: A
Realistic Assessment of the Challenge: Workshop Summary;
Giffin, R.; Robinson, S., Eds.; National Academies Press:
Washington, 2009. (b) Rendon, A.; Tiberi, S.; Scardigli, A.;
D'Ambrosio, L.; Centis, R.; Caminero, J. A.; Migliori, G. B.
J. Thorac. Dis. 2016, 8, 2666.
4. Keshavjee, S. Background on the Green Light Committee and
the Second Line Tuberculosis Drug Supply Chain; National
Academies Press: Washington, 2012.
5. Kurosawa, H. J. Antibiot., Ser. B 1952, 5, 682.
6. (a) Stammer, C. H.; Wilson, A. N.; Holly, F. W.; Folkers, K.
J. Am. Chem. Soc. 1955, 77, 2346. (b) El-Obeid, H. A.;
Al-Badr, A. A. Analytical Profile of D-cycloserine in Analytical
Profiles of Drug Substances; Academic Press: New York, 1989,
Vol. 18, p. 567. (c) Hardman, J. G. Goodman and Gilman's. The
Pharmacological Basis of Therapeutics; McGraw-Hill: New
York, 1995, 9th ed., p. 1164. (d) Kuehl, F. A., Jr.; Wolf, F. J.;
Trenner, N. R.; Peck, R. L.; Buhs, R. P.; Howe, E.; Putter, I.;
Hunnewell, B. D.; Ormond, R.; Downing, G.; Lyons, J. E.;
Newstead, E.; Chaiet, L. Folkers, K. J. Am. Chem. Soc. 1955,
77, 2344.
26. Kasture, A. V.; Wadodkar, S. G.; Mahadik, K. R.; More, H. V.
Pharmaceutical Analysis; Pune: Nirali Prakashan, 1997,
Vol. 1, p. 12.
27. Baer, E. J. Biol. Chem. 1955, 212, 25.
28. Stammer, C. H. J. Org. Chem. 1962, 27, 2957.
29. Ongania, K. H. Arch. Pharm (Weinheim, Ger.) 1979, 312,
958.
30. Hidy, P. H. J. Am. Chem. Soc. 1955, 77, 2345.
1253

 Awasthi, Arun Kumar
Awasthi, Arun Kumar
 Kumar, Brijesh
Kumar, Brijesh
 Aga, Mushtaq A
Aga, Mushtaq A
 Tripathi, Punit
Tripathi, Punit
 Reddy, Cirandur Suresh
Reddy, Cirandur Suresh
 Kumar, Pramod
Kumar, Pramod