
Journal of Photochemistry and Photobiology A: Chemistry (2021)
Update date:2022-08-29
Topics:
 Yan, Mao
Yan, Mao
 Wang, Qun-Hui
Wang, Qun-Hui
 Zhu, Yi-Zhou
Zhu, Yi-Zhou
 Han, Ming-Liang
Han, Ming-Liang
 Yan, Yi-Qiao
Yan, Yi-Qiao
 Zheng, Jian-Yu
Zheng, Jian-Yu
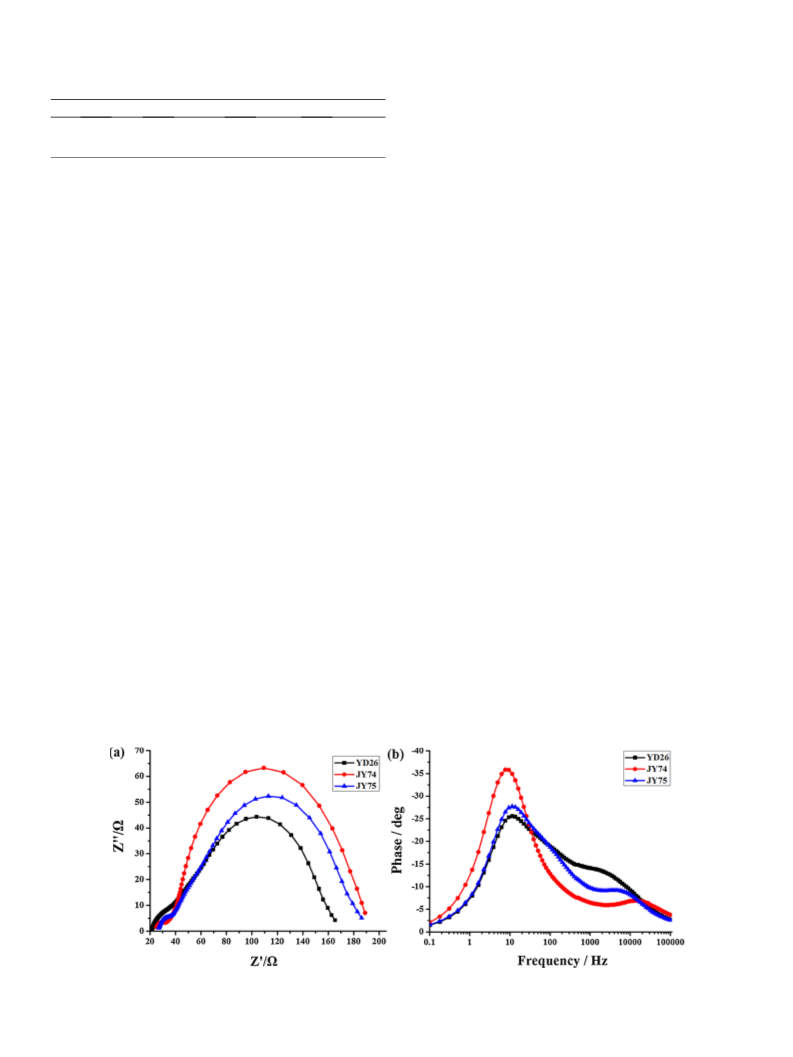
Triptycene has been first introduced into porphyrin sensitizers (JY74 and JY75) for dye sensitized solar cells. Compared to reference dye YD26, the homoaromatic electron delocalization of triptycene makes the designed dyes a broadened absorption. Meanwhile, the rigid shape-persistent character of triptycene endows JY74 and JY75 an improved ability to diminish the charge recombination between electrolyte and semiconductor TiO2. As a result, after structural modification, larger short-circuit current and higher open-circuit voltage are achieved for dyes JY74 and JY75. The power conversion efficiency of JY75 is increased by 26.8 % compared to that of YD26. Consequently, triptycene may be a promising bulky steric hindrance group for decorating photosensitizer to get an attractive photovoltaic performance.
View More




Shanghai Pinewood Fine Chemical Co., Ltd.
website:http://www.pinewoodchem.com
Contact:0086-21-62417129,62414096
Address:Suite B, 27F, No.2, Lane 600, Tianshan Road, Shanghai
Shanghai Bocimed Pharmaceutical Co., Ltd.
website:http://www.bocimed.com
Contact:+86-21-68861632
Address:Building 1, Lane 647, Songtao Road, Zhangjiang High-Tech Park, Shanghai
Shanghai AoBo Bio-Pharmaceutical Technology Co., Ltd.
Contact:+86-21-51320130-801, 816
Address:Room 601, No. 1011, Halei Road, Zhangjiang High-Tech Park, Pudong, Shanghai
Hangzhou Showland Technology Co., Ltd.
Contact:86-571-88920516
Address:ROOM2118,NO.553,WENSAN ROAD,HANGZHOU,CHINA
Contact:+86-717-6370352
Address:168 Chengdong Avenue, Yichang, Hubei 443003, P. R.China
Doi:10.1016/0040-4020(72)80153-4
(1972)Doi:10.1039/DT9810001492
(1981)Doi:10.1002/1521-3773(20020916)41:18<3405::AID-ANIE3405>3.0.CO;2-P
(2002)Doi:10.1002/ardp.19813140215
(1981)Doi:10.1021/ja100399m
(2010)Doi:10.1021/ja00878a050
(1962)