Notes and References
C(10′)
C(11′)
†
E-mail: houz@postman.riken.go.jp
C(9′)
C(8′)
O(2′)
O(3′)
‡ A typical procedure for the synthesis of 1 is as follows. In a glove box,
addition of a thf solution (5 ml) of fluorenone (180 mg, 1 mmol) to freshly-
cut sodium chips (48 mg, 2.1 mmol) gave immediately a brown solution,
which after being stirred at room temperature for ca. 1 h, changed to dark
green and yielded gradually thf-insoluble green precipitates. The green
mixture was further stirred at room temperature for 5 h. The thf was pumped
off and DME (5 ml) was added to give a green solution. Evaporation of
DME yielded a green oily residue, which after addition of thf and standing
at room temperature for several days afforded thf-insoluble green blocks of
C(12′)
C(1′)
C(13)
C(2′)
O(1′)
C(3′)
Na(1′)
C(10)
C(9)
Na(2′)
C(12)
C(11)
1
(292 mg, 0.79 mmol, 79% yield). Anal. Calc. for C21
H, 6.53. Found. C, 67.80, H, 6.44%.
Crystal data: for 1: C21 24Na
1
P2 /n (no. 15), a = 12.6600(9), b = 8.5523(5), c = 17.8251(11) Å, b =
2 3
H24Na O : C, 68.10;
C(8)
O(2)
C(13)
§
H
O
2 3
, M = 370.40, monoclinic, space group
C(1)
O(3)
C(2)
C(3)
O(1)
3
23
9
c
8.942(6)°, U = 1906.5(2) Å , Z = 4, D = 1.25 g cm , m(Cu-Ka) =
2
1
1
0.440 cm , 3120 measured reflections, 2892 independent, R = 0.0847
= 0.0830) for 2387 data with I > 3s(I) and 301 variables. For 2 (two
independent molecules in the unit cell): C46 46Ir , M = 1015.32,
orthorhombic, space group Pcab (no. 61), a = 16.476(5), b = 31.468(8), c
Na(1)
(R
w
H
2 2
O
Na(2)
3
23
=
7
(
14.645(4) Å, U = 7593(3) Å , Z = 8, D
c
= 1.78 g cm , m(Mo-Ka) =
0.131 cm , 9831 measured reflections, 7404 independent, R = 0.0580
w
R = 0.0638) for 4071 data with I > 3s(I) and 451 variables. CCDC
2
1
Fig. 1 Extended structure of 1. Selected bond lengths (Å) angles (°) (A = 1.5
x, 0.5 + y, 1.5 2 z): Na(1)–O(1) 2.168(6), Na(1)–O(2) 2.371(6), Na(1)–
O(3) 2.391(7), Na(2)–O(1) 2.348(6), Na(2)–C(1) 2.644(7), Na(2)–C(2)
.921(7), Na(2A)–O(1) 2.340(6), Na(2A)–C(1) 2.775(7), Na(2A)–C(13)
.083(7), Na(2A)–O(2) 2.440(6), Na(2A)–O(3) 2.606(6), Na(1A)–C(8)
.733(7), Na(1A)–C(9) 2.822(8), Na(1A)–C(10) 2.888(8), Na(1A)–C(11)
.881(8), Na(1A)–C(12) 2.805(8), Na(1A)–C(13) 2.725(7), O(1)–C(1)
.379(8); O(1)–Na(1)–O(2) 91.2(2), O(1)–Na(1)–O(3) 86.2(2),
O(2)–Na(1)–O(3) 84.5(3), Na(1)–O(1)–C(1) 164.1(5), Na(2)–O(1)–C(1)
6.3(4), Na(1)–O(1)–Na(2) 98.9(2), Na(1)–O(1)–Na(2A) 83.6(2), Na(2A)–
O(1)–C(1) 92.9(4), Na(2)–O(1)–Na(2A) 173.4(3), Na(1A)–C(13)–Na(2A)
2.2(2).
2
1
¶
C
82/761.
1H NMR for 2 (CD
Cl
7.26, J
), 6.90 (d of t, J
). C NMR (CD Cl , 22 °C): d 169.3, 154.5, 141.1, 137.2,
25.8, 123.6, 120.3, 99.1, 8.8. IR (in thf): n(CO) 1996.9 cm . Anal. Calc.
23IrO: C, 54.42; H, 4.57. Found. C, 54.24, H, 4.49%.
, 22 °C): d 7.47 (d of d, J
1.32 Hz, 2 H, C12
7.26, J 1.32 Hz, 2 H, C12
7.26, J
), 7.06 (d of t, J
1.32 Hz, 2 H,
7.26,
), 1.87
2
2
1
2
2
3
2
2
1
H
12 8
), 7.46 (d of d, J
1
2
H
8
1
J
2
1.32 Hz, 2 H, C12
H
8
1
2
H
8
13
(s, 15 H, C
5
Me
5
2
2
2
1
1
for C23
H
8
7
1 W. Schlenk and T. Weichel, Ber., 1911, 44, 1182; H. Schlubach, Ber.,
915, 48, 12; C. B. Wooster, J. Am. Chem. Soc., 1928, 50, 1388; W. E.
1
Bachmann, J. Am. Chem. Soc., 1933, 55, 1179; P. J. Hamrick, Jr. and
C. R. Hauser, J. Am. Chem. Soc., 1959, 81, 493; S. Selman and J. F.
Easthan, J. Org. Chem., 1965, 30, 3804; E. L. Anderson and J. E. Casey,
Jr., J. Org. Chem., 1965, 30, 3959; W. S. Murphy and D. J. Buckley,
Tetrahedron Lett., 1969, 2975; J. W. Huffman, in Comprehensive
Organic Synthesis, ed. B. M. Trost and I. Fleming, Pergamon, New
York, 1991, vol. 8, ch. 1.4; G. M. Robertson, in Comprehensive Organic
Synthesis, ed. B. M. Trost and I. Fleming, Pergamon, New York, 1991,
vol. 3, ch. 2.6.
J. E. McMurry and L. R. Krepiski, J. Org. Chem., 1976, 41, 3929; J. E.
McMurry, M. P. Fleming, K. L. Kees and L. R. Krepiski, J. Org. Chem.,
1978, 43, 3255; J. M. Pons and M. Santelli, Tetrahedron Lett., 1982, 23,
4937; J. E. McMurry, Acc. Chem. Res., 1983, 16, 405; 1974, 7, 281;
Chem. Rev., 1989, 89, 1513; A. F u¨ rstner and B. Bogdanovic, Angew.
Chem., Int. Ed. Engl., 1996, 35, 2442.
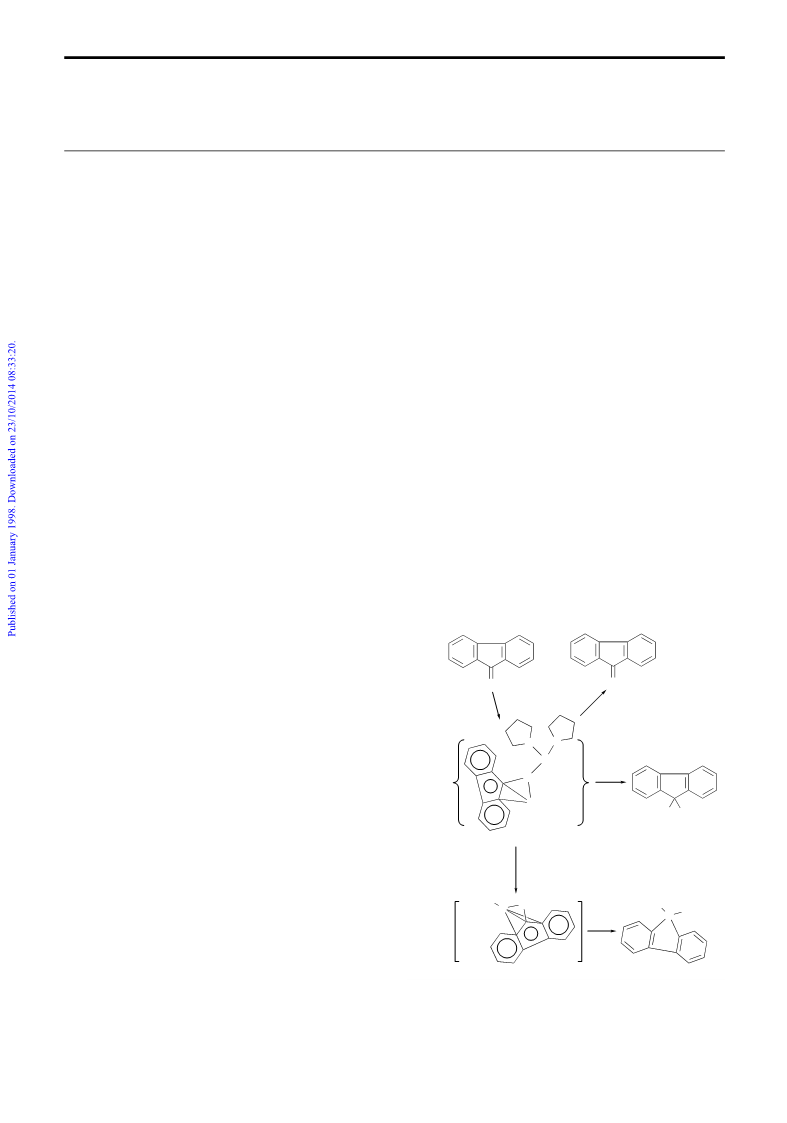
Reflecting the reactivity of a fluorenone dianion species,1–3
hydrolysis of 1 yielded fluorenol, while air oxidation of 1
afforded fluorenone almost quantitatively. Moreover, when 1
was allowed to react with 0.5 equiv. of [(C
5
Me
5
2
)IrCl(m-Cl)] at
room temp. in DME, the decarbonylation product 2 was
obtained as colorless crystals in 68% isolated yield (Scheme 1,
Fig. 2).§¶ In contrast, the similar reaction of sodium benzo-
phenone dianion with [(C
5
Me
5
2
)IrCl(m-Cl)] did not give any
2
3
C–C bond cleavage product, but instead afforded benzophenone
and several unidentified iridium hydride species. These results
again demonstrated the difference in behavior between fluor-
enone dianion and benzophenone dianion. Further studies on
the decarbonylation reaction are in progress.
Z. Hou, K. Takamine, Y. Fujiwara and H. Taniguchi, Chem. Lett., 1987,
This work was partly supported by a grant-in-aid from the
Ministry of Education, Science, Sports, and Culture of Japan.
2
061; Z. Hou, K. Takamine, O. Aoki, H. Shiraishi, Y. Fujiwara and H.
Taniguchi, J. Chem. Soc., Chem. Commun., 1988, 668; J. Org. Chem.,
1
1
988, 53, 6077; H. Olivier, Y. Chauvin and L. Saussine, Tetrahedron,
989, 45, 165; Z. Hou, T. Yoshimura and Y. Wakatsuki, J. Am. Chem.
Soc., 1994, 116, 11169; T. Yoshimura, Z. Hou and Y. Wakatsuki,
Organometallics, 1995, 14, 5382.
C(16)
C(15)
4
5
B. Bogdanovic, C. Kruger and B. Wermeckes, Angew. Chem., Int. Ed.
Engl., 1980, 19, 817.
Z. Hou, H. Yamazaki, K. Kobayashi, Y. Fujiwara and H. Taniguchi,
J. Chem. Soc., Chem. Commun., 1992, 722; Z. Hou, H. Yamazaki, Y.
Fujiwara and H. Taniguchi, Organometallics, 1992, 11, 2711.
J. E. Davies, J. Kopf and E. Weiss, Acta Crystallogr., Sect. B, 1982, 38,
C(14)
C(17)
C(13)
Ir(1)
6
7
C(23)
2
251.
S. Brooker, F. T. Edelmann, T. Kottke, H. W. Roesky, G. M. Scheldrick,
D. Stalke and K. H. Whitmire, J. Chem. Soc., Chem. Commun., 1991,
144.
O(1)
C(2)
C(11)
C(1)
8
9
Z. Hou, A. Fujita, H. Yamazaki and Y. Wakatsuki, J. Am. Chem. Soc.,
C(12)
1
996, 118, 2503.
S. Corbelin, J. Kopf and E. Weiss, Chem. Ber., 1991, 124, 2417.
C(6)
C(7)
1
1
0 Q. Shen, M. Qi and Y. Lin, J.Organomet. Chem., 1990, 399, 247.
1 H. R. Luss and D. L. Smith, Acta Crystallogr., Sect. B, 1972, 28,
8
84.
1
2 Z. Hou, T. Miyano, H. Yamazaki and Y. Wakatsuki, J. Am. Chem. Soc.,
1995, 117, 4421; Z. Hou, A. Fujita, H. Yamazaki and Y. Wakatsuki,
J. Am. Chem. Soc., 1996, 118, 7843.
Fig. 2 ORTEP drawing of 2 (only one of the two independent molecules is
shown for clarity). Selected bond lengths (Å) and angles (°): Ir(1)–C(1)
2
.07(2), Ir(1)–C(12) 2.05(2), Ir(1)–C(13) 2.25(3), Ir(1)–C(14) 2.27(3),
Ir(1)–C(15) 2.27(3), Ir(1)–C(16) 2.27(2), Ir(1)–C(17) 2.28(2), Ir(1)–C(23)
.83(1), O(1)–C(23) 1.14(3); Ir(1)–C(23)–O(1) 179(2).
1
Received in Cambridge, UK, 12th January 1998; 8/00291F
670
Chem. Commun., 1998

 Hou, Zhaomin
Hou, Zhaomin
 Fujita, Akira
Fujita, Akira
 Yamazaki, Hiroshi
Yamazaki, Hiroshi
 Wakatsuki, Yasuo
Wakatsuki, Yasuo