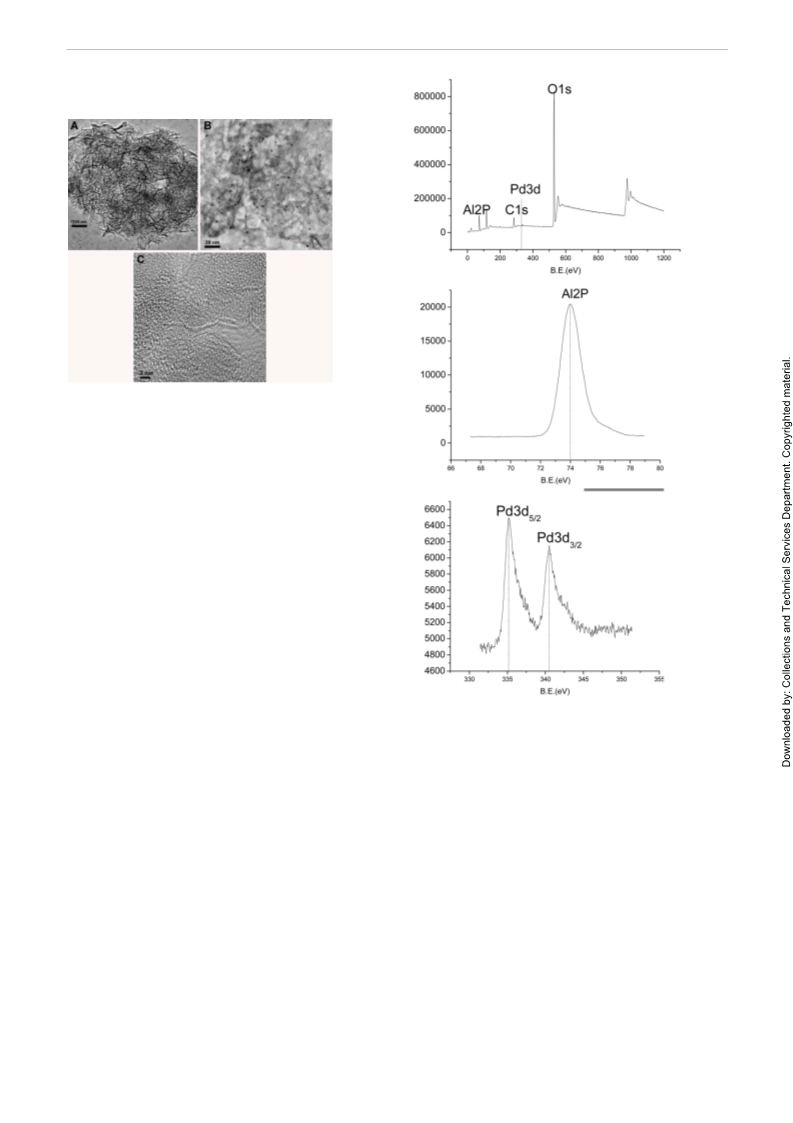
PAPER
Pd/Al(OH) Nanoparticle Catalyst for Suzuki Coupling Reactions
1601
3
provided the coupling product after purification by column chroma- Acknowledgment
tography (eluent: PE) of the crude product (Table 2, entry 21); yield:
We appreciate gratefully the Natural Science Foundation of Shanxi
Province (No. 2012021007-2 and No. 2011011010-2) and Scienti-
fic and Technological Innovation Programs of Higher Education In-
stitutions in Shanxi (No. 20120006) for financial support.
1
05.5 mg (91%); white solid; mp 89–90 °C; R = 0.7 (PE).
f
1
H NMR (600 MHz, CDCl ): δ = 7.39–7.37 (t, J = 7.8 Hz, 1 H),
3
7.48–7.42 (m, 4 H), 7.59–7.56 (m, 4 H).
3,5-Dimethyl-1,1′-biphenyl
Following the general procedure, bromobenzene (52 μL, 0.5 mmol)
and 3,5-dimethylphenylboronic acid (82.5 mg, 0.55 mmol, 1.1
equiv) provided the coupling product after purification by column
chromatography (eluent: PE) of the crude product (Table 2, entry
Supporting Information for this article is available online at
http://www.thieme-connect.com/ejournals/toc/synthesis.SnoIufproi
m
tgioSrantnugIifoop
r
itmnatr
2
2); yield: 91.0 mg (99%); colorless liquid; R = 0.6 (PE).
f
References
1
H NMR (600 MHz, CDCl ): δ = 7.35 (t, J = 7.2 Hz, 1 H), 7.42–7.40
3
(1) (a) Suzuki, A. J. Organomet. Chem. 2002, 653, 83. (b) Phan,
N. T. S.; Sluys, M. V. D.; Jones, C. W. Adv. Synth. Catal.
(t, J = 7.2 Hz, 1 H), 7.48–7.46 (m, 4 H), 7.55–7.54 (d, J = 7.2 Hz, 2
H).
2
006, 348, 609. (c) Yin, L. X.; Jürgen, L. Chem. Rev. 2007,
3,5-Dichloro-1,1′-biphenyl
107, 133. (d) Shen, X. Q.; Jones, G. O.; Watson, D. A.;
Bhayana, B.; Buchwald, S. L. J. Am. Chem. Soc. 2010, 132,
Following the general procedure, bromobenzene (52 μL, 0.5 mmol)
and 3,5-dichlorophenylboronic acid (105.0 mg, 0.55 mmol, 1.1
equiv) provided the coupling product after purification by column
chromatography (eluent: PE) of the crude product (Table 2, entry
1
1278.
2) (a) Bhayana, B.; Fors, B. P.; Buchwald, S. L. Org. Lett.
009, 11, 3954. (b) Jin, M. J.; Lee, D. H. Angew. Chem. Int.
(
2
23); yield: 108.3 mg (97%); colorless liquid; R = 0.6 (PE).
f
Ed. 2010, 49, 1119. (c) Fujihara, T.; Yoshida, S.; Terao, J.;
Tsuji, Y. Org. Lett. 2009, 11, 2121. (d) Ackermann, L.;
Potukuchi, H. K.; Althammer, A.; Born, R.; Mayer, P. Org.
Lett. 2010, 12, 1004. (e) Bellina, F.; Carpita, A.; Rossi, R.
Synthesis 2004, 2419.
1
H NMR (600 MHz, CDCl ): δ = 2.40 (s, 6 H), 7.01 (s, 1 H), 7.23
3
(s, 2 H), 7.35–7.33 (t, J = 1.8 Hz, 1 H), 7.45–7.42 (t, J = 7.2 Hz, 2
H), 7.60–7.58 (q, J = 7.2 Hz, 2 H).
4
-Methyl-4′-nitro-1,1′-biphenyl
(
3) (a) Littke, A. F.; Dai, C. Y.; Fu, G. C. J. Am. Chem. Soc.
Following the general procedure, 4-nitrochlorobenzene (78.5 mg,
2
2
000, 122, 4020. (b) Leadbeater, N. E.; Marco, M. Org. Lett.
002, 4, 2973. (c) Barder, T. E.; Walker, S. D.; Martinelli, J.
0.5 mmol) and 4-methylphenylboronic acid (102 mg, 0.75 mmol,
1.5 equiv) provided the coupling product after purification by col-
R.; Buchwald, S. L. J. Am. Chem. Soc. 2005, 127, 4685.
d) Navarro, O.; Kelly, R. A. III; Nolan, S. P. J. Am. Chem.
umn chromatography (eluent: PE) of the crude product (Table 4, en-
try 6); yield: 100.7 mg (94%); white solid; mp 140–142 °C; R = 0.1
(
f
Soc. 2003, 125, 16194. (e) Kinzel, T.; Zhang, Y.; Buchwald,
S. L. J. Am. Chem. Soc. 2010, 132, 14073. (f) Kondolff, I.;
Doucet, H.; Santelli, M. Synlett 2005, 2057.
(PE).
1
H NMR (600 MHz, CDCl ): δ = 8.30–8.27 (dt, J = 2.4, 4.8, 9.6 Hz,
3
2
H), 7.74–7.71 (dt, J = 2.4, 4.8, 9.6 Hz, 2 H), 7.55–7.53 (d, J = 8.4
(4) (a) Martin, R.; Buchwald, S. L. Acc. Chem. Res. 2008, 41,
461. (b) Fu, G. C. Acc. Chem. Res. 2008, 41, 1555.
Hz, 2 H), 7.32–7.31 (d, J = 7.8 Hz, 2 H), 2.45 (s, 3 H).
1
13
(c) Billingsley, K. L.; Buchwald, S. L. Angew. Chem. Int.
Ed. 2008, 47, 4695. (d) Tang, W. J.; Capacci, A. G.; Wei, X.
D.; Li, W. J.; White, A.; Patel, N. D.; Savoie, J.; Gao, J. J.;
Rodriguez, S.; Qu, B.; Haddad, N.; Lu, B. Z.;
Krishnamurthy, D.; Yee, N. K.; Senanayake, C. H. Angew.
Chem. Int. Ed. 2010, 49, 5879.
C NMR (150 MHz, CDCl ): δ = 149.4, 137.5, 136.3, 132.3, 132.0,
3
128.7, 128.3, 128.2, 127.9, 124.1, 21.2.
4
-Methyl-2′-nitro-1,1′-biphenyl
Following the general procedure, 2-nitrochlorobenzene (78.5 mg,
0.5 mmol) and 4-methylphenylboronic acid (102 mg, 0.75 mmol,
1.5 equiv) provided the coupling product after purification by col-
(
5) (a) Kim, S. W.; Kim, M.; Lee, W. Y.; Hyeon, T. J. Am.
Chem. Soc. 2002, 124, 7642. (b) Zhang, Z. H.; Wang, Z. Y.
J. Org. Chem. 2006, 71, 7485. (c) Lv, G. H.; Mai, W. P.; Jin,
R. Z.; Gao, L. X. Synlett 2008, 1418. (d) Reetz, M. T.;
Westermann, E. Angew. Chem. Int. Ed. 2000, 39, 165.
(e) Lamblin, M.; Nassar-Hardy, L.; Hierso, J. C.; Fouquet,
E.; Felpin, F. X. Adv. Synth. Catal. 2010, 352, 33. (f) Costa,
N. J. S.; Kiyohara, P. K.; Monteiro, A. L.; Coppel, Y.;
Philippot, K.; Rossi, L. M. J. Catal. 2010, 276, 382.
(g) Scheuermann, G. M.; Rumi, L.; Steurer, P.; Bannwarth,
W.; Mülhaupt, R. J. Am. Chem. Soc. 2009, 131, 8262.
(h) Tandukar, S.; Sen, A. J. Mol. Catal. A: Chem. 2007, 268,
112. (i) Yuan, B. Z.; Pan, Y. Y.; Li, Y. W.; Yin, B. L.; Jiang,
H. F. Angew. Chem. Int. Ed. 2010, 49, 4054. (j) Gallon, B.
J.; Kojima, R. W.; Kaner, R. B.; Diaconescu, P. L. Angew.
Chem. Int. Ed. 2007, 46, 7251. (k) Kogan, V.; Aizenshtat,
Z.; Biro, R. P.; Neumann, R. Org. Lett. 2002, 4, 3529.
umn chromatography (eluent: PE) of the crude product (Table 4, en-
try 7); yield: 70.7 mg (66%); yellow oil; R = 0.6 (PE).
f
1
H NMR (600 MHz, CDCl ): δ = 7.84–7.83 (dd, J = 1.2, 7.8 Hz, 1
3
H), 7.62–7.59 (td, J = 1.2, 7.8, 14.4 Hz, 1 H), 7.48–7.44 (m, 4 H),
2.42 (s, 3 H).
13
C-NMR (100 MHz, CDCl ): δ = 149.2, 138.0, 136.0, 134.3, 132.1,
3
131.8, 129.3, 127.8, 127.6, 123.8, 21.1.
4
-Methyl-3′-nitro-1,1′-biphenyl
Following the general procedure, 3-nitrochlorobenzene (78.5 mg,
0.5 mmol) and 4-methylphenylboronic acid (102 mg, 0.75 mmol,
1
.5 equiv) provided the coupling product after purification by col-
umn chromatography (eluent: PE) of the crude product (Table 4, en-
try 8); yield: 53.6 mg (50%); white solid; mp 75–76 °C; R = 0.6
f
(PE).
1
H NMR (600 MHz, CDCl ): δ = 8.45–8.44 (t, J = 2.4 Hz, 1 H),
3
(l) Wu, L.; Li, B. L.; Huang, Y. Y.; Zhou, H. F.; He, Y. M.;
8.19–8.17 (m, 1 H), 7.91–7.90 (m, 1 H), 7.61–7.58 (t, J = 7.8 Hz, 1
Fan, Q. H. Org. Lett. 2006, 8, 3605. (m) Mandali, P. K.;
Chand, D. K. Catal. Commun. 2013, 31, 16. (n) Senapati, K.
K.; Roy, S.; Borgohain, C.; Phukan, P. J. Mol. Catal. A:
Chem. 2012, 352, 128. (o) Singh, A. S.; Patil, U. B.;
Nagarkar, J. M. Catal. Commun. 2013, 35, 11. (p) Das, P.;
Sharma, D.; Shil, A. K.; Kumari, A. Tetrahedron Lett. 2011,
H), 7.54–7.53 (d, J = 7.8 Hz, 2 H), 7.32–7.31 (d, J = 7.8 Hz, 2 H),
2.43 (s, 3 H).
13
C NMR (150 MHz, CDCl ): δ = 148.7, 142.8, 138.6, 135.7, 132.8,
3
129.9, 129.7, 127.0, 121.7, 121.6, 21.2.
5
2, 1176. (q) Samarasimhareddy, M.; Prabhu, G.;
©
Georg Thieme Verlag Stuttgart · New York
Synthesis 2014, 46, 1593–1602

 Li, Xing
Li, Xing
 Chen, Wenwen
Chen, Wenwen
 Chang, Honghong
Chang, Honghong
 Shao, Zhongqi
Shao, Zhongqi
 Wei, Wenlong
Wei, Wenlong