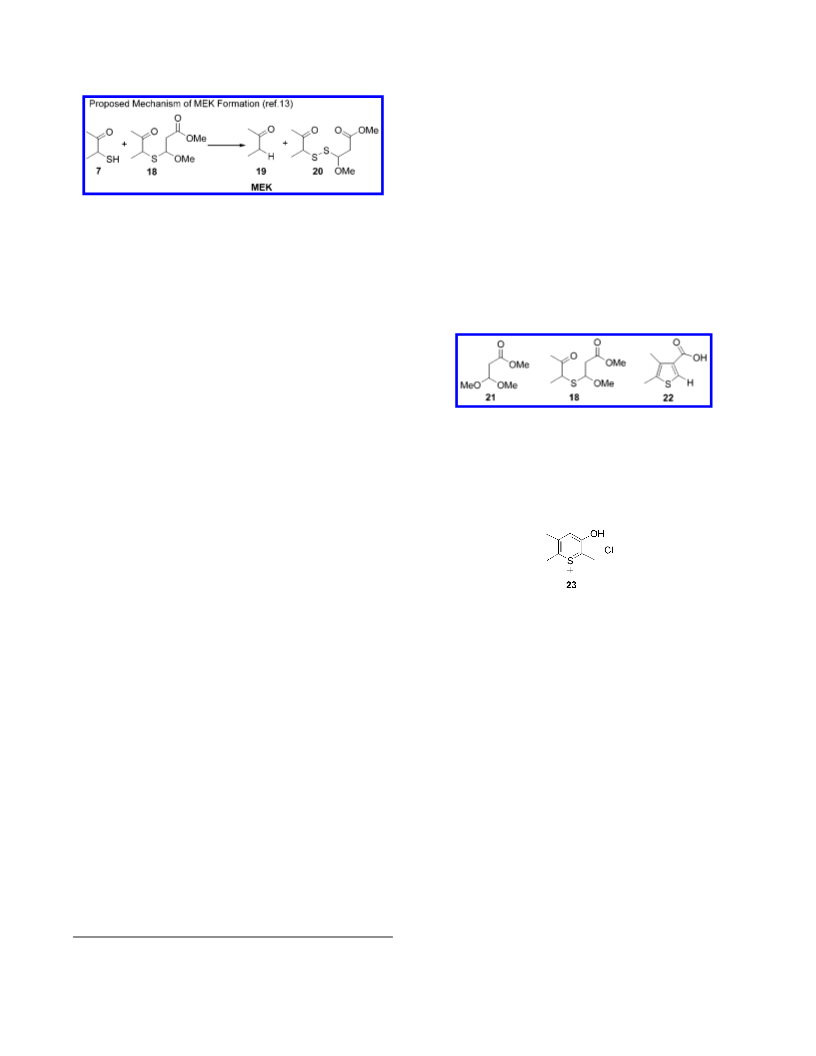
butanone is foul smelling, with a very low detection limit.
The material should be kept in the fume hood. The 3-chloro-
and assayed as 29.8% 13, indicating a chemical yield of 3.16
kg of 13, or 81% yield from 6.
2
-butanone is an irritant and lachrymator, and should be
handled with due care. Allylamine is highly toxic, volatile
bp 53 °C), and is absorbed readily through the skin;
The crude reaction mixture was placed in a 22-L flask
equipped with a mechanical stirrer, a vacuum inlet, and a
distillation head atop a two-tray, Oldershaw-type, vacuum-
jacketed distillation column. Most of the toluene was distilled
through the column at ca. 75 mmHg. The residue (6.04 kg)
was then transferred to a 6-L flask, similarly equipped, but
with a five-tray Oldershaw column. Two early fractions were
collected, the first containing mostly toluene, and the second
consisting of toluene, acrylate 11, methyl 3,3-dimethoxypro-
pionate, and 13. The main fraction of 13 came over at about
(
appropriate personal protective equipment should be worn,
and the material should be kept in the fume hood.
Methyl 4,5-dimethylthiophene-3-carboxylate (13). A
35-L jacketed reactor equipped with an overhead stirrer, a
bottom drain valve, a dropping funnel, a thermocouple and
a nitrogen inlet was charged with toluene (6.5 kg), NaSH
(
43% aqueous solution, 3.43 kg, 26.34 mol), and water (2.72
kg). The mixture was stirred vigorously and cooled to 10
C. The 3-chloro-2-butanone 6 (2.50 kg, 22.91 mol) was
1
09 °C at 11.5 mmHg as a yellow liquid, affording 2.90 kg
of material with >99% purity. The isolated yield was
therefore 74% from chlorobutanone 6. The main loss during
distillation was in the fraction immediately preceding the
main cut. An analytical sample was obtained by washing
distilled material with bleach (to oxidize aliphatic sulfur
°
charged through the addition funnel, the reaction temperature
being maintained at 15-20 °C by adjusting the feed rate
and jacket temperature. The addition required 2 h and 15
min. After the addition was complete, the mixture was stirred
vigorously for 1 h while the jacket temperature was raised
to 25 °C. Stirring was then stopped, the phases were allowed
to separate and settle for 10 min, whereupon the lower,
aqueous phase was drained out of the bottom of the reactor.
The agitator was pulsed a few times to knock water off the
reactor walls and complete the phase separation. Note: The
aqueous phase can be deodorized prior to disposal by
treatment with bleach (caution! exothermic reaction). After
drainage of the aqueous phase, the drain stem was washed
with water and acetone, and dried under a stream of nitrogen.
The homogeneous, light yellow organic phase containing
1
5
compounds ), and redistilling through a Vigreux column
under vacuum. The compound was obtained as a nearly
1
colorless liquid, bp 85 °C (3 mmHg). H NMR (CDCl
3
, 400
MHz) δ 7.83 (s, 1H), 3.80 (s, 3H), 2.33 (s, 3H), 2.31 (s,
1
3
3
1
5
5
H); C NMR (CDCl
3
, 100 MHz) δ 164.0, 134.3, 134.1,
+
32.4, 130.1, 51.5, 13.5, 13.4; MS (GC-MS) m/z 170 (M ,
4%), 155 (17), 139 (100); Anal. Calcd for C
6.45; H, 5.92; S, 18.83. Found: C, 56.37; H, 5.98; S, 18.61.
,5-Dimethyl-2-trimethylsilylthiophene-3-carboxylic Acid
4). To a 22-L three-necked flask equipped with a thermo-
8 10 2
H O S: C,
4
(
couple, condenser, and overhead stirrer under a nitrogen
purge was added 1.19 kg (6.57 mol) of ester 13, and 2.08
kg of a solution of LiOH (6.90 mol) in water. The mixture
was heated to reflux for about 60 min, whereupon LC
analysis confirmed that the hydrolysis reaction was complete.
A Dean-Stark receiver was then placed under the condenser,
heptane was added, and the mixture was distilled to the
absence of visible water in the distillate. After all methanol
and water had been removed, distillation was continued to
remove about half of the heptane over about 90 min.
The resulting slurry of the lithium carboxylate was cooled
to 0 ( 2 °C, and treated with 4.88 kg of anhydrous, BHT
stabilized THF, and 350 g of diisopropylamine (DIPA, 3.42
mol). A solution of n-BuLi (1.898 kg, 24.2 wt % n-BuLi in
heptane, 2.636 L, 7.18 mol) was slowly charged over about
3-mercapto-2-butanone 7 was then drained into a tightly
sealed polypropylene container while the reactor was cleaned
out with water and acetone, and dried under a strong stream
of nitrogen.
The solution of 7 (8.75 kg) was then returned to the clean,
dry reactor along with a toluene rinse (280 g). A portion of
the methyl 3-methoxyacrylate (11, 250 g, 2.29 mol) was
added to the solution through the dropping funnel, and the
mixture was stirred vigorously with the jacket temperature
at 15 °C. Solid NaOMe (128 g, 2.29 mol) was added all at
once, causing an immediate temperature rise of ca. 3 °C
followed by slow cooling of the mixture. After about 30 min,
the reaction temperature started rising against the cooling
jacket, signaling the initiation of the cyclization. The
remaining acrylate (2.28 kg, 19.13 mol) was then added in
over 2 h to maintain the reaction temperature below 30 °C.
After the addition was completed, the mixture was stirred
overnight at room temperature.
The mixture was cooled to 10 °C with good stirring, and
concentrated HCl (1.67 kg, 16.93 mol) was added in over
about 30 min, producing an exotherm (to 28 °C) and a
precipitate of NaCl. The mixture was stirred at room
temperature for 1.5 h, and then water (1.39 kg) was added
to dissolve the salt. After stirring for 15 min, the phases were
allowed to separate and settle for 15 min. The lower, aqueous
phase was drained out, and the organic phase containing 13
3
h, the long addition time serving solely to maintain the
reaction temperature at 0 ( 2 °C. After the addition funnel
was rinsed with 50 g of heptane, 797 g (7.26 mol) of
chlorotrimethylsilane was added in over 2 h while maintain-
ing the reaction temperature at 0 ( 2 °C. The mixture was
stirred for another 30 min, and then assayed by LC to confirm
reaction completion.
The reaction mixture was then heated at atmospheric
pressure, and the THF (for recycle) was distilled through an
efficient fractionating column. The heat-up was performed
slowly in order to expel most of the byproduct butane in a
controlled manner. After THF distillation, the resulting slurry
was cooled to 60 °C, and 1.2 kg of heptane was added.
Heating was resumed to distill off 1.2 kg of solvent. The
3
was washed similarly with 1.98 kg of 5% aqueous NaHCO .
The aqueous phase was drained, and the organic phase was
weighed into a small drum. The material weighed 10.59 kg
(15) We thank Dr. James A. Miles for suggesting this procedure.
Vol. 6, No. 4, 2002 / Organic Process Research & Development
•
365

 Phillips, Gary
Phillips, Gary
 Fevig, Thomas L.
Fevig, Thomas L.
 Lau, Patrick H.
Lau, Patrick H.
 Klemm, George H.
Klemm, George H.
 Mao, Michael K.
Mao, Michael K.
 Ma, Chun
Ma, Chun
 Gloeckner, James A.
Gloeckner, James A.
 Clark, Art S.
Clark, Art S.