Novel Fluoronaphthyridone Antibacterial Agents
J ournal of Medicinal Chemistry, 1997, Vol. 40, No. 22 3591
NMR (CDCl3, ppm) δ 4.5 (1H, m), 4.05 (1H, m), 3.9-3.5 (3H,
m), 1.5 (9H, s); MS (FAB, m/e) 211 (M + H). Anal. (C10H14N2O3)
C, H, N.
(1H, bs), 3.41 (1H, bs), 3.28 (1H, m), 3.00 (1H, m), 1.40 (18H,
s); MS (FAB, m/e) 344 (M + H). Anal. (C16H29N3O5) C, H, N.
Syn th esis of 4-((N-(ter t-bu toxycar bon yl)am in o)m eth yl)-
1-N-(ter t-bu toxyca r bon yl)p yr r olid in -3-on e Oxim e (11a ).
To a solution of the ketone 10 (300 mg, 0.95 mmol) in a mixture
of 5 mL of 95% EtOH and 2.5 mL of THF was added
hydroxylamine hydrochloride (232 mg, 3.5 molar equiv) and
NaHCO3 (281 mg, 3.5 molar equiv) dissolved in 2.5 mL of
distilled water. The reaction mixture was stirred for 1 h at
40 °C, and concentrated under reduced pressure. The residue
was diluted with EtOAc (50 mL), washed successively with
water and saturated saline solution, dried over anhydrous
MgSO4, and filtered. The filtrate was concentrated in vacuo,
and the residue was purified by column chromatography (silica
gel, hexane/EtOAc ) 2:1) to give the oxime 11a (292 mg, yield
93%) as a colorless oil: 1H NMR (CDCl3, ppm) δ 9.70 (1H, bs),
5.05 (1H, bs), 3.83-2.79 (7H, m), 1.42 (18H, s). MS (FAB, m/e)
330 (M + H). Anal. (C15H27N3O5) C, H, N.
Syn th esis of 4-Cya n o-1-N-(ter t-bu toxyca r bon yl)p yr -
r olid in -3-ol (7). The ketone compound 6 (10 g, 0.047 mol) in
500 mL of EtOH was cooled to 0 °C (ice bath), and to this
solution was added portionwise 3.6 g (0.094 mol) of NaBH4
over a period of 20 min. The reaction mixture was stirred for
30 min at the same temperature and concentrated under
reduced pressure. The residue was diluted with EtOAc (400
mL), washed with water, dried over anhydrous MgSO4, and
filtered. The filtrate was concentrated under reduced pressure
to give a residue which was purified by column chromatogra-
phy (silica gel, hexane/EtOAc ) 4:1), affording the alcohol 7
(10.1g, 0.047 mol) as a colorless oil in quantitative yield: 1H
NMR (CDCl3, ppm) δ 4.5 (1H, m), 3.8 (3H, m), 3.3 (1H, m), 3.0
(1H, m), 1.4 (9H, s); MS (FAB, m/e) 213 (M + H). Anal.
(C10H16N2O3) C, H, N.
Syn th esis of 4-((N-(ter t-bu toxycar bon yl)am in o)m eth yl)-
1-N-(ter t-bu toxyca r bon yl)p yr r olid in -3-on e O-Isop r op yl-
oxim e (11e). To a solution of the free oxime 11a (292 mg,
0.88 mmol) and n-Bu4NBr (85 mg, 0.26 mmol) in 15 mL of
CH2Cl2 were added isopropyl bromide (216 mg, 1.76 mmol) and
5 mL of a 15% solution of NaOH. The reaction mixture was
stirred for 5 h at room temperature and diluted with CH2Cl2
(20 mL). The organic layer was washed successively with
water and saturated saline solution, dried over anhydrous
MgSO4, and filtered. The filtrate was concentrated in vacuo,
and the residue was purified with column chromatography
(silica gel, hexane/EtOAc ) 3:1) to give the desired compound
11d (265 mg, yield 81%) as a colorless oil: 1H NMR (CDCl3,
ppm) δ 5.05 (1H, bs), 4,11 (2H, s), 4.01 (1H, m), 3.55-3.25 (3H,
m), 3.40 (1H, m), 3.02 (1H, m), 1.55 (18H, s), 1.05 (1H, d); MS
(FAB, m/e) 372 (M + H). Anal. (C18H33N3O5) C, H, N.
Syn th esis of 3,3a ,4,6-Tetr a h yd r op yr r o[3,4-c]p yr a zole-
2,5-d ica r boxylic Acid Di-ter t-bu tyl Ester . A solution of the
free oxime 11a (30 mg, 0.09 mmol) in 3 mL of CH2Cl2 was
cooled to 0 °C. To this solution were added Et3N (25 µL, 0.18
mmol) and p-TsCl (20 mg, 0.108 mmol), and the reaction
mixture was stirred for 10 min at the same temperature. After
3 h at room temperature, the reaction mixture was concen-
trated and the residue was purified by column chromatography
(silica gel, hexane/EtOAc ) 3:1) to give the cyclized compound
(29.8 mg, yield 95%) as a colorless oil: 1H NMR (CDCl3, ppm)
δ 4.10 (4H, m), 3.65 (1H, m), 3.51 (1H, m), 3.05 (1H, m), 1.53
(9H, s), 1.50 (9H, s); MS (FAB, m/e) 312 (M + H). Anal.
(C15H24N4O3) C, H, N.
Syn th esis of 4-(Am in om eth yl)pyr r olidin -3-on e-O-Meth -
yloxim e Dih yd r och lor id e (12b). Acetyl chloride (10 mL)
was added slowly to 20 mL of MeOH at 0 °C. The reaction
mixture was stirred for 30 min at the same temperature,
followed by addition of the oxime 11b (990 mg, 2.97 mmol) in
10 mL of methanol. The reaction mixture was stirred for 50
min at room temperature and concentrated under reduced
pressure. The residue was suspended with EtOAc and filtered.
The solid obtained was washed twice with EtOAc and dried
in vacuo to give 648 mg (yield 94%) of the dihydrochloride 12b
as a white solid: 1H NMR (DMSO-d6, ppm) δ 10.0 (3H, bs),
8.35 (2H, bs), 3.90 (2H, m), 3.80 (3H, s), 3.69 (1H, m), 3.40
(2H, m), 3.12 (2H, m); MS (FAB, m/e) 144 (M + H). Anal.
(C6H15N3OCl2) C, H, N.
Syn th esis of 4-((N-(ter t-Bu toxycar bon yl)am in o)m eth yl)-
1-N-(ter t-bu toxyca r bon yl)p yr r olid in -3-ol (9). The alcohol
7 was dissolved in 200 mL of dry THF, and this solution was
cooled to -5 °C using an ice-salt bath. LiAlH4 (2.6 g, 0.066
mol) was then added over a period of 20 min. The reaction
mixture was stirred for 30 min at the same temperature and
quenched with 2.6 mL of water, 2.6 mL of 15% NaOH solution,
and finally with 7.8 mL of water. This mixture was stirred
for 1 h at room temperature. After 6 g of anhydrous MgSO4
was added the mixture was stirred for a further 30 min and
filtered. The filtrate was concentrated to yield the crude amine
product 8. The latter was diluted with 200 mL of dioxane-
water (2:1 by volume) followed by portionwise addition of di-
tert-butoxycarbonyl dicarbonate (12.3 g, 0.056 mol). The
mixture was stirred for 30 min and concentrated in vacuo, and
the residue was diluted with EtOAc (400 mL). This solution
was washed with water and saturated saline, dried over
anhydrous MgSO4, and filtered. The filtrate was concentrated
in vacuo, and the residue was purified by column chromatog-
raphy (silica gel, hexane/EtOAc ) 3:1) to give 12.3 g (yield,
83%) of the title compound 9 as a colorless oil: 1H NMR
(CDCl3, ppm) δ 4.95 (1H, m), 4.1 (1H, m), 3.5 (2H, m), 3.3-3.0
(4H, m), 2.1 (1H, m), 1.45 (18H, s); MS (FAB, m/e) 317 (M +
H). Anal. (C15H28N2O5) C, H, N.
Syn th esis of 4-((N-(ter t-bu toxycar bon yl)am in o)m eth yl)-
1-N-(ter t-bu toxyca r bon yl)p yr r olid in -3-on e (10). To a so-
lution of the alcohol 9 (14 g, 0.044 mol) in DMSO (64 mL) was
added Et3N (18.5 mL, 3 molar equiv), and the mixture was
cooled to 5 °C (ice bath). When the mixture by the wall of the
reaction flask begins to freeze, pyridine-sulfur trioxide (Py-
SO3, 12.7 g, 1.8 molar equiv) oxidant was added portionwise
thereto. After the addition was completed, the ice bath was
removed and the reaction solution was stirred for 3 h at room
temperature. The reaction mixture was diluted with water
and extracted three times with EtOAc (300 mL). The com-
bined extracts were dried over anhydrous MgSO4, filtered, and
concentrated in vacuo. The residue thus obtained was purified
by column chromatography (silica gel, hexane/EtOAc ) 3:1)
to give 11.8 g (yield, 85%) of the ketone 10 as a colorless oil:
1H NMR (CDCl3, ppm) δ 4.95 (1H, bs), 4.11 (1H, m), 3.90 (1H,
m), 3.69 (1H, m), 3.40 (3H, m), 2.78 (1H, m), 1.45 (9H, s), 1.40
(9H, s); MS (FAB, m/e) 315 (M + H). Anal. (C15H26N2O5) C,
H, N.
Syn th esis of 4-((N-(ter t-bu toxycar bon yl)am in o)m eth yl)-
1-N-(ter t-bu toxycar bon yl)pyr r olidin -3-on e O-Meth yloxim e
(11b). To a solution of the ketone 10 (260 mg, 0.83 mmol) in
a mixture of 5 mL of 95% EtOH and 2.5 mL of THF were added
methoxylamine hydrochloride (256 mg, 3.7 molar equiv) and
NaHCO3 (257 mg, 3.7 molar equiv) dissolved in 2.5 mL of
distilled water. The reaction mixture was stirred for 1 h at
40 °C and concentrated under reduced pressure. The residue
was diluted with EtOAc (50 mL), washed successively with
water and saturated saline solution, dried over anhydrous
MgSO4, and filtered. The filtrate was concentrated in vacuo,
and the residue was purified by column chromatography (silica
gel, hexane/EtOAc ) 3:1), affording the oxime compound 11b
(250 mg, yield 88%) as a colorless oil: 1H NMR (CDCl3, ppm)
δ 4.98 (1H, bs), 4.08 (2H, bs), 3.81 (3H, s), 3.75 (1H, m), 3.50
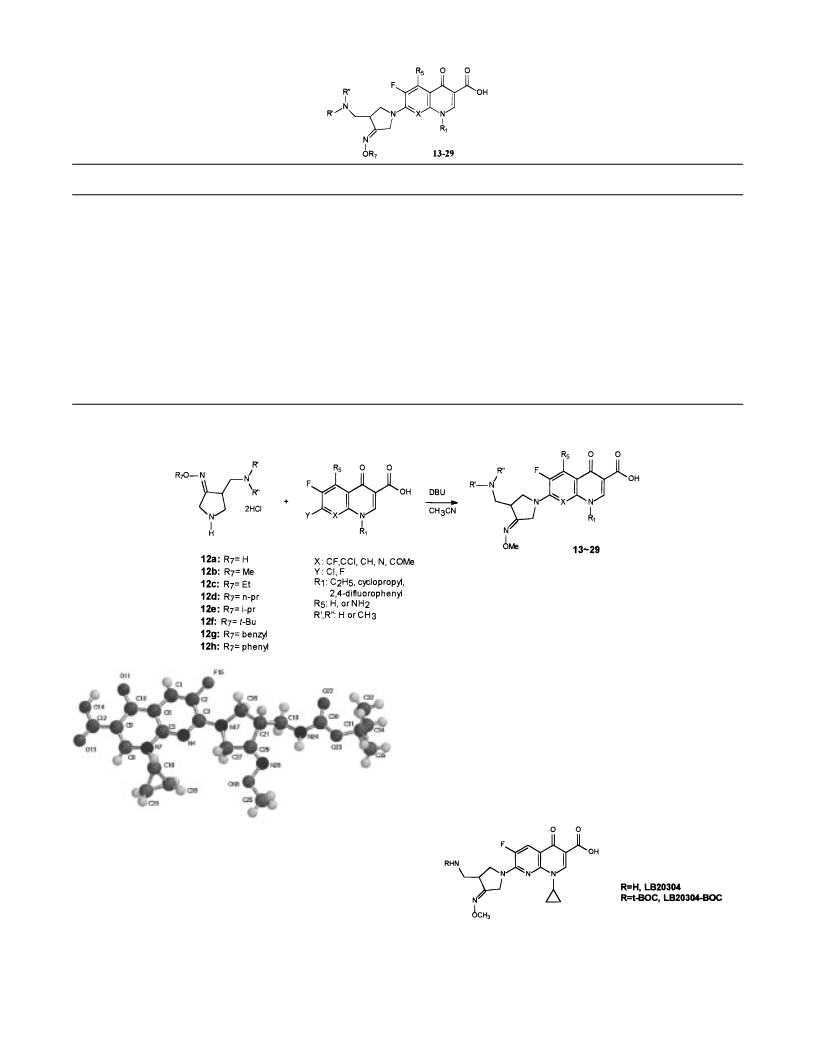
Syn th esis of 7-(4-(Am in om eth yl)-3-(m eth oxyim in o)-
pyr r olidin -1-yl)-1-cyclopr opyl-6-flu or o-4-oxo-1,4-dih ydr o-
[1,8]n a p h th yr id in e-3-ca r boxylic Acid (20). 7-Chloro-1-
cyclopropyl-6-fluoro-4-oxo-1,4-dihydro-1,8-naphthyridine-3-
carboxylic acid (141 mg, 0.5 mmol) and 4-(aminomethyl)-
pyrrolidin-3-one O-methyloxime dihydrochloride (110 mg, 0.5
mmol) were added to 15 mL of dry CH3CN. Then, 1,8-
diazabicyclo[5.4.0]undec-7-ene (DBU, 4.6 g, 30 mmol) was
added at 0 °C, and the reaction mixture was stirred for 30
min at room temperature. Distilled water (15 mL) was added
to the reaction mixture. The precipitated solids were filtered,
washed with CH3CN and Et2O, and dried in vacuo to yield
167 mg (yield 85%) of coupling product 20. The product was
recrystallized from CHCl3-EtOH to give an off-white amor-
phous solid: 1H NMR (DMSO-d6, ppm) δ 8.61 (1H, s), 8.05

 Hong, Chang Yong
Hong, Chang Yong
 Kim, Young Kwan
Kim, Young Kwan
 Chang, Jay Hyok
Chang, Jay Hyok
 Kim, Se Ho
Kim, Se Ho
 Choi, Hoon
Choi, Hoon
 Nam, Do Hyun
Nam, Do Hyun
 Kim, Yong Zu
Kim, Yong Zu
 Kwak, Jin Hwan
Kwak, Jin Hwan