Communications
1
H), 7.14 (pseudo t (dd), Jffi9.5 Hz, 1H), 7.30 (d, J = 4.2 Hz, 1H), 7.53
(
pseudo t (dd), Jffi10.3 Hz, 1H), 7.67 (d, J = 4.2, 1H), 8.40 ppm (d, J =
1
3
9
1
3
7
.5 Hz, 1H); C NMR (75.5 MHz, CDCl ): d = 24.1, 113.4, 117.1,
3
22.0, 127.2, 133.3, 133.5, 134.8, 136.7, 137.8, 148.0 ppm; FTIR: n˜ =
085, 3021, 2957, 2923, 2853, 1591, 1560, 1487, 1419, 1389, 1358, 906,
À1
+
72, 743 cm ; MS (DCI): m/z: 177 [M+H] ; HRMS calcd for
+
[
C H Cl+H] : 177.0471; found: 177.0484; elemental analysis (%)
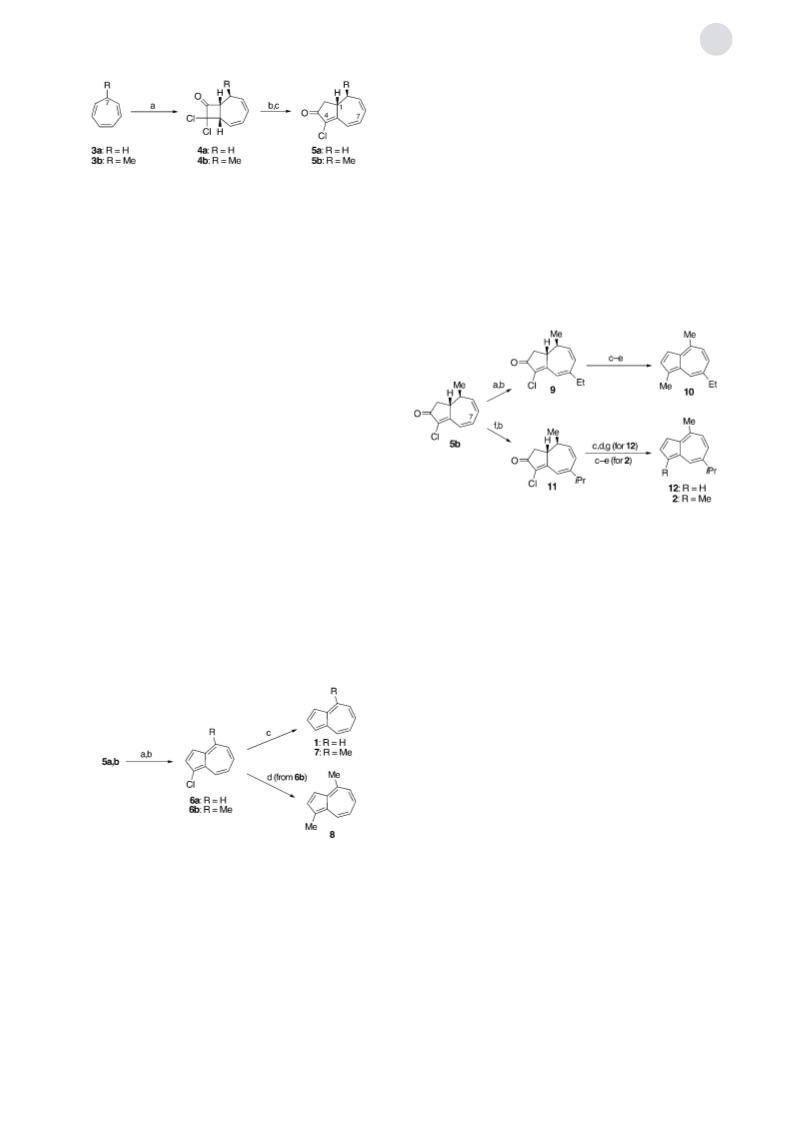
Scheme 4. Preparation of (ꢀ)-chamazulenecarboxylic acid (14):
a) 1. MeCH=C(OMe)OTBDMS, LiClO , CH Cl , 208C, 12 h; 2. PhSeCl,
11 9
calcd for C H Cl: C 74.79, H5.14; found: C 74.84, H5.18.
11
9
4
2
2
1
Data for selected compounds: 2: M.p. 31–328C; HNMR
À50!08C, 2 h; b) H O , pyridine, 08C, 1 h (46%, 2 steps); c) NaBH ,
2
2
4
(
300 MHz, CDCl ): d = 1.36 (d, J = 6.9 Hz, 6H), 2.66 (s, 3H), 2.83
MeOH/CeCl , 08C, 1.5 h (92%); d) 1. Burgess reagent, THF, 08C, 1 h;
3
3
(s, 3H), 3.08 (sept, J = 6.9 Hz, 1H), 7.01 (d, J = 10.7 Hz, 1H), 7.22 (d,
2
. p-chloranil, 208C, 24 h (55%); e) MeB(OH) , Pd(OAc) , dpdb,
2
2
J = 3.7 Hz, 1H), 7.42 (dd, J = 10.7, 1.8 Hz, 1H), 7.62 (d, J = 3.7 Hz,
K PO , PhMe, 1008C, 24 h (99%); f) LiOH, THF, H O, 208C, 12 h
3
4
2
1
3
1
1
1
1
H), 8.19 ppm (d, J = 1.8 Hz, 1H); C NMR (75.5 MHz, CDCl ): d =
(
94%).
3
3.0, 24.2, 24.9, 38.4, 112.9, 125.2, 125.3, 133.5, 135.0, 136.2, 136.4,
37.4, 140.0, 144.4 ppm; FTIR: n˜ = 3095, 3064, 2958, 2924, 2854, 1554,
527, 1462, 1420, 1387, 1367, 772 cm ; MS (DCI): m/z: 199 [M+H] ;
HRMS calcd for [C H +H] : 199.1487; found: 199.1482.
0: HNMR (300 MHz, CDCl 3): d = 1.34 (t, J = 7.6 Hz, 3H), 2.65
s, 3H), 2.82 (s, 3H), 2.83 (q, J = 7.6 Hz, 2H), 6.98 (d, J = 10.5 Hz,
H), 7.21 (brs, 1H), 7.38 (dd, J = 10.5, 1.9 Hz, 1H), 7.61 (br s, 1H),
.15 ppm (d, J = 1.9 Hz, 1H); C NMR (75.5 MHz, CDCl ): d = 13.0,
7.5, 24.2, 34.0, 112.9, 125.1, 125.2, 134.8, 135.9, 136.3, 136.4, 136.5,
À1
+
+
1
5
19
1
1
(
1
8
1
1
1
1
3
3
37.5, 144.4 ppm; FTIR: n˜ = 3098, 3063, 2960, 2926, 2866, 1555, 1561,
526, 1452, 1422, 1364, 772 cm ; MS (DCI): m/z: 185 [M+H] ;
HRMS calcd for [C H +H] : 185.1330; found: 185.1334; elemental
À1
+
+
1
4
16
analysis (%) calcd for C H : C 91.25, H8.75; found: C 91.44, H8.86.
1
4
16
1
1
2: HNMR (300 MHz, CDCl 3): d = 1.35 (d, J = 6.8 Hz, 6H), 2.88
s, 3H), 3.07 (sept, J = 6.8 Hz, 1H), 7.11 (d, J = 10.6 Hz, 1H), 7.29 (d,
J = 3.8 Hz, 1H), 7.31 (d, J = 3.8 Hz, 1H), 7.46 (dd, J = 10.6, 2.0 Hz,
also been efficiently prepared, thereby demonstrating four
distinctly different substitution options as well. Finally,
(
[
18e]
[18f]
acetoxy and aza derivatives 17a
and 17b
are examples
1
H), 7.80 (pseudo t (dd), Jffi3.8 Hz, 1H), 8.31 ppm (d, J = 2.0 Hz,
1
3
of novel azulenes that are readily prepared with this method-
ology.
1H); C NMR (75.5 MHz, CDCl ): d = 24.3, 24.7, 38.2, 114.7, 118.0,
3
126.0, 135.3, 135.4, 136.6, 137.4, 140.2, 141.8, 145.2 ppm; FTIR: n˜ =
093, 3064, 2958, 2924, 1556, 1529, 1461, 1422, 1389, 1362, 749 cm
MS (DCI): m/z: 185 [M+H] ; HRMS calcd for [C H ] : 184.1252;
À1
3
;
In conclusion, modern chemistry has been applied to the
long-standing problem of azulene synthesis and an efficient,
highly flexible approach has resulted. Our approach allows
controlled access to a wide variety of substituents and
substitution arrays and should prove to be among the most
broadly useful methods to prepare these important com-
pounds.
+
+
1
4
16
found: 184.1259.
4: M.p. 86–878C; HNMR (300 MHz, CDCl 3): d = 1.61 (d, J =
.2 Hz, 3H), 2.65 (s, 3H), 2.83 (s, 3H), 3.88 (q, J = 7.2 Hz, 1H), 7.00
1
1
7
(d, J = 10.7 Hz, 1H), 7.28 (d, J = 3.8 Hz, 1H), 7.44 (dd, J = 10.7,
1.9 Hz, 1H), 7.64 (d, J = 3.8 Hz, 1H), 8.22 ppm (d, J = 1.9 Hz, 1H);
C NMR (75.5 MHz, CDCl ): d = 13.0, 19.2, 24.2, 48.8, 114.3, 125.1,
27.1, 130.9, 133.9, 135.7, 135.8, 136.9, 137.6, 145.6 ppm; FTIR: n˜ =
585, 3103, 3065, 2961, 2923, 2853, 1704, 1556, 1454, 1261, 1023,
1
3
3
1
3
7
À1
+
74 cm ; MS (DCI): m/z: 229 [M+H] .
Experimental Section
Experimental procedure for the preparation of 6b from 5b: NaBH
4
Received: April 12, 2005
Published online: July 13, 2005
(43 mg, 1.13 mmol) was added to a stirred solution of 5b (200 mg,
1
.03 mmol) in MeOH/CeCl (2.80 mL, 0.40m, 1.12 mmol) at 08C. The
3
reaction mixture was stirred for 1.5 h at 08C and then treated with a
Keywords: aromatic compounds · azulenes · cycloaddition ·
natural products · synthesis design
saturated solution of aqueous NaH PO . The crude product was
2
4
.
isolated with EtOAc/pentane (1:1) in the usual manner and purified
by column chromatography on dry silica gel with diethyl ether/
pentane (40:60) to afford 198 mg (98%) of the trans alcohol as a
1
white solid. M.p. 67–688C; HNMR (300 MHz, CDCl 3): d = 1.17 (d,
J = 7.1 Hz, 3H), 1.38–1.48 (m, 1H), 2.16–2.24 (m, 2H), 2.33 (pseudo q,
Jffi7.3 Hz, 1H), 2.68–2.77 (m, 1H), 4.57 (pseudo t, Jffi7.2 Hz, 1H), 5.66
[1] S. Piesse, C. R. Hebd. Seances Acad. Sci. 1863, 57, 1016.
[2] P. A. Plattner, A. S. Pfau, Helv. Chim. Acta 1937, 20, 224 – 232;
see, also: H.-J. Hansen, Chimia 1996, 50, 489 – 496.
(
dd, J = 11.4, 3.3 Hz, 1H), 5.85–5.94 (m, 1H), 6.01 (dd, J = 11.5,
1
3
6
.9 Hz, 1H), 6.51 ppm (d, J = 11.5 Hz, 1H); C NMR (75.5 MHz,
[3] For reviews on the azulenes, see: a) M. Gordon, Chem. Rev.
1952, 52, 127 – 200; b) V. B. Mochalin, Y. N. Porshnev, Russ.
Chem. Rev. 1977, 46, 530 – 547; c) K.-P. Zeller in Methoden der
Organischen Chemie (Houben-Weyl), Vol. V/2c (Ed.: H. Kropf),
Georg Thieme, Stuttgart, 1985, pp. 127 – 418; for a recent
synthetic approach, see: d) A. L. Crombie, J. L. Kane, Jr.,
K. M. Shea, R. L. Danheiser, J. Org. Chem. 2004, 69, 8652 – 8667.
[4] a) Y. Coquerel, PhD Thesis, University of Grenoble, 2001; b) Y.
Coquerel, A. Blanc, J.-P. Deprꢀs, A. E. Greene, M.-T. Averbuch-
Pouchot, C. Philouze, A. Durif, Acta Crystallogr. Sect. C 2000, 56,
1480 – 1481; c) Y. Coquerel, A. E. Greene, J.-P. Deprꢀs, Org.
Lett. 2003, 5, 4453 – 4455; see, also: d) R. Yokoyama, S. Ito, M.
CDCl ): d = 19.2, 38.9, 40.3, 46.6, 74.6, 123.7, 125.3, 127.7, 133.6, 139.6,
3
À1
1
41.5 ppm; FTIR: n˜ = 3360, 3016, 1700 cm . A solution of the
Burgess reagent (347 mg, 1.46 mmol) in dry THF (5 mL) was added to
a stirred solution of the above alcohol (198 mg, 1.01 mmol) in dry
THF (6.0 mL) at 08C. The reaction mixture was allowed to warm to
2
08C and stirred for 1 h, whereupon p-chloranil (745 mg, 3.03 mmol)
was added and the resulting mixture was stirred for a further 24 h. The
crude product was isolated with pentane in the usual way and purified
by column chromatography on dry silica gel with pentane to give
9
2 mg (52%) of chloroazulene 6b as a blue solid. M.p. 368C;
1
HNMR (300 MHz, CDCl 3): d = 2.86 (s, 3H), 7.13 (d, J = 10.3 Hz,
5
132
ꢀ 2005 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Angew. Chem. Int. Ed. 2005, 44, 5130 –5133

 Carret, Sebastien
Carret, Sebastien
 Blanc, Aurelien
Blanc, Aurelien
 Coquerel, Yoann
Coquerel, Yoann
 Berthod, Mikael
Berthod, Mikael
 Greene, Andrew E.
Greene, Andrew E.
 Depres, Jean-Pierre
Depres, Jean-Pierre