Inorganic Chemistry Communications
journal homepage: www.elsevier.com/locate/inoche
Cyclopentasilane Si H : First single crystal X-ray structure of an
5
10
oligosilane Si H and thermal analysis with TG/MS
x
y
a
a
b
a,
Dana Schmidt , Uwe Böhme , Jürgen Seidel , Edwin Kroke ⁎
a
TU Bergakademie Freiberg, Institut für Anorganische Chemie, Leipziger Straße 29, 09596 Freiberg, Germany
TU Bergakademie Freiberg, Institut für Physikalische Chemie, Leipziger Straße 29, 09596 Freiberg, Germany
b
a r t i c l e i n f o
a b s t r a c t
Article history:
Cyclopentasilane Si H (CPS) – a liquid silicon precursor – was crystallized in situ at −100 °C which allowed
5
10
Received 10 April 2013
Accepted 16 May 2013
Available online 25 May 2013
determination of the single crystal X-ray structure, showing short intermolecular distances below the sum of
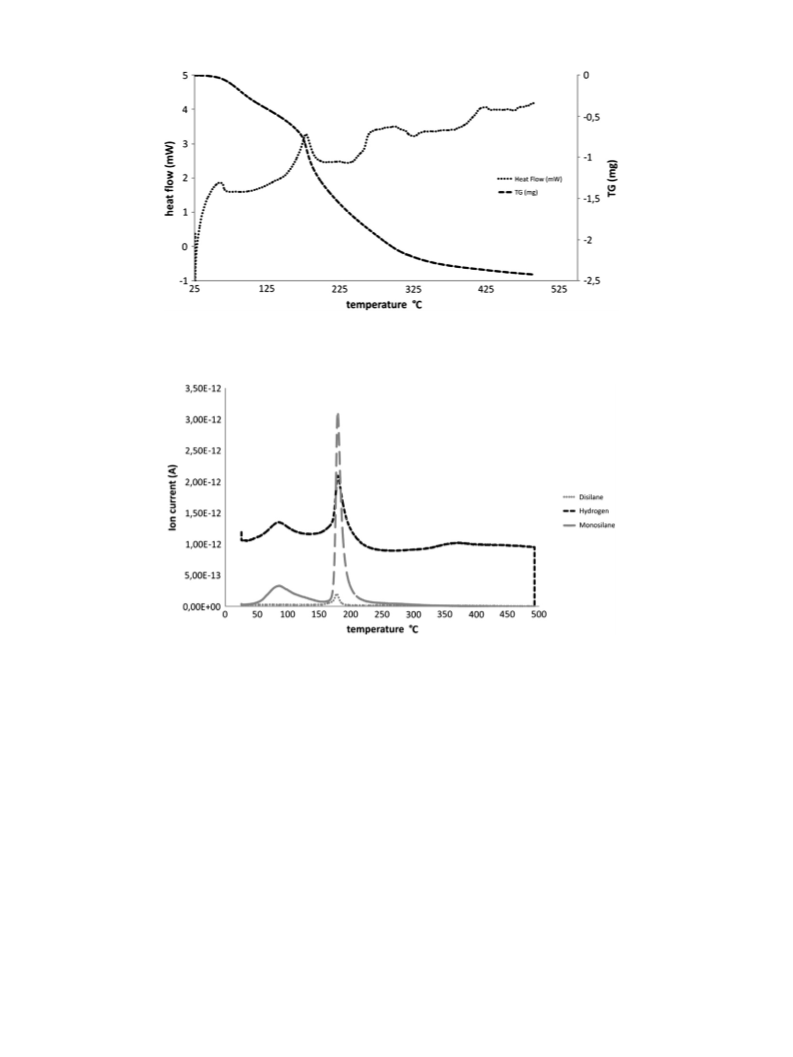
the v.d.Waals radii of the corresponding atoms. Further characterization included a thermal analysis with
mass spectrometry (TG/MS) of the segregated species hydrogen, mono- and disilane.
©
2013 Elsevier B.V. All rights reserved.
Keywords:
Silicon
Silanes
Precursors
Molecular structure
Thermal transformation
Recently, attention has been paid to (liquid) oligosilanes Si
1–7]. This renewed interest stems from the concept to prepare sili-
x
H
y
by 1H and 29Si NMR spectra in benzene solution, showing singlets at
3.5 ppm and -106.9 ppm, respectively. These values correspond well
with the literature data. Similarly, Raman spectra obtained from sam-
ples in sealed capillaries correspond well with the data published by
Hengge et al. [9].
For determination of a single crystal X-ray structure CPS was filled
in a thin-walled glass capillary which was sealed and mounted on the
goniometer head of the diffractometer. Freezing the liquid on the dif-
fractometer gave polycrystalline material which was unsuitable for an
X-ray structure analysis. Repeated melting and refreezing between
263 and 153 K under slow movement along the z-axis of the goniom-
eter head, gave at last a monocrystalline block in the capillary which
was suitable for a crystal structure determination. The data collection
was performed at 173 K in order to reduce ice formation outside the
[
con films for transistors, solar cells and other devices via printing
and deposition techniques, which are simpler and more cost efficient
than traditional semiconductor processing [1–5]. Oligosilanes are
convertible to a-Si:H at low temperatures (>250 °C). Suitable com-
pounds are restricted to oxygen- and carbon-free liquid oligosilanes
x y
Si H , which was shown in a pioneering paper by Shimoda et al. [1].
The principal syntheses of CPS and other oligosilanes are long known
since the early works of Stock [8] and Hengge [9]. However, several
efforts have been undertaken in recent years to characterize the highly
sensitive and pyrophoric CPS and to utilize it for “liquid silicon inks”
[
1–5]. Further interest arises from rocket technology, since oligosilanes
might be very good combustion enhancing fuel additives [6], and useful
for the synthesis of high capacity graphite–silicon composite anode ma-
terials for lithium-ion batteries [7]. Besides, oligosilanes are known to
show the phenomenon of σ-delocalization, which refers to a σ-orbital
interaction similar to the conjugation and delocalization of π-electrons
in unsaturated carbon compounds [10]. There are few examples of
crystal structure studies on hydrosilanes. These are restricted to
glass capillary. CPS crystallizes in the monoclinic space group P 2
(Fig. 1).
1
/m
The structure is not isomorph with the X-ray structure of
cyclopentane C 10 which crystallizes in a hexagonal unit cell [15].
5
H
Silicon atom Si1 of CPS is situated on special position 2e, site symme-
try m. Thereby Si2A and Si3A are generated by the crystallographic
mirror plane. Thus, the CPS molecule has an envelope conformation
powder-XRD of layered polysilane (Si
a high-pressure phase of monosilane (SiH
v.d.Waals adduct SiH (H [13]. Thus, to the best of our knowledge
6
H
6
)
n
[11], the crystal structure of
4
) [12], and the high-pressure
s
with perfect C symmetry. Atom Si1 lies 0.54(1) Å outside the plane
4
2
)
2
formed by the atoms Si2, Si3, Si3A, and Si2A. The angle between
the plane formed by the atoms Si2, Si1, Si2A and the plane formed
by the atoms Si2, Si3, Si3A and Si2A is 23.05(6)°. The Si\Si bond
lengths are between 2.3353(1) and 2.3377(7) Å (Table S1). These
no crystal structure analysis has been reported for any linear, cyclic or
branched hydrosilane so far.
CPS was prepared according to known procedures with some mod-
ifications [9,14], according to Scheme 1. Formation of CPS was indicated
5 3 5 6 5
bonds are shorter than in Si (CH )10 [16] (2.349 Å), Si (C H )10 [17]
(
2.396 Å) or Si Cl10 [18] (2.347 and 2.363 Å). Also permethylated linear
5
silanes have longer Si\Si bond lengths of 2.35 Å [19]. The structure of
⁎
CPS was also investigated by gas-phase electron diffraction. Here

 Schmidt, Dana
Schmidt, Dana
 B?hme, Uwe
B?hme, Uwe
 Seidel, Jürgen
Seidel, Jürgen
 Kroke, Edwin
Kroke, Edwin