W. Buckel, B. T. Golding et al.
FULL PAPER
(
(
2
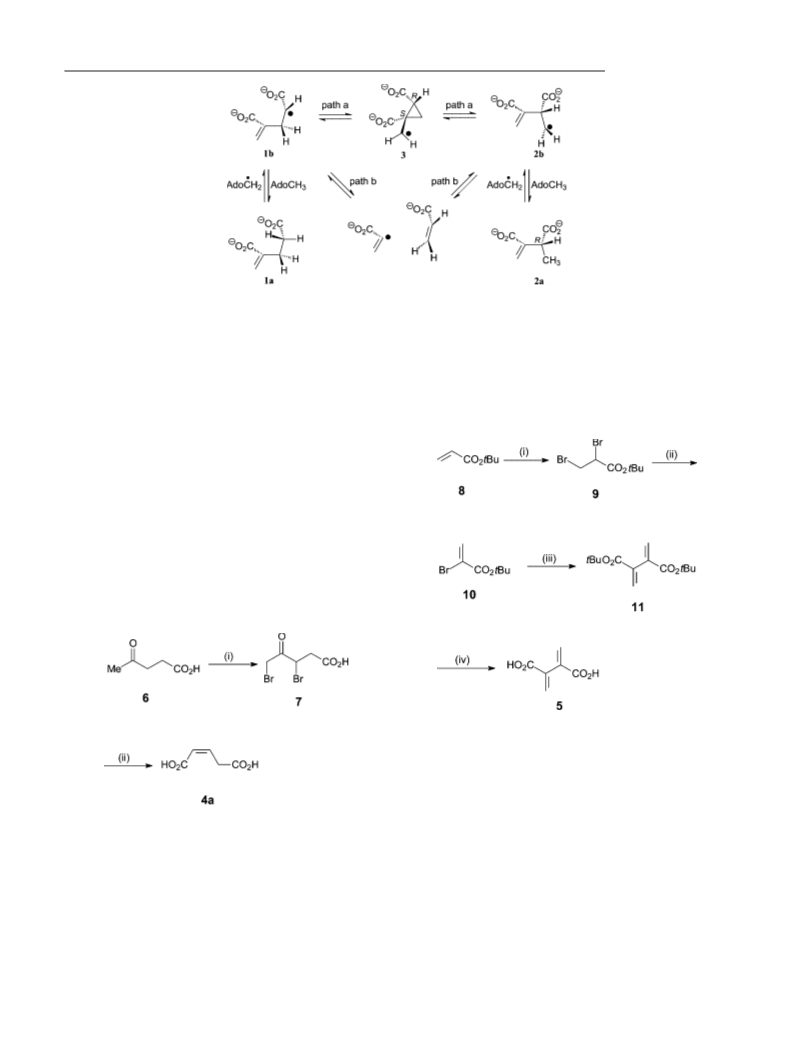
Z)-Glutaconic Acid (4a): KHCO
210 mL) was added to 3,5-dibromolaevulinic acid (5.5 g,
1 mmol). The mixture was vigorously stirred for 19 h. When con-
3
(17.0 g, 169 mmol) in water
taining fractions (R
collected and the solvent was removed to afford the title compound
as a colourless oil (0.48 g, 47% yield). H NMR (300 MHz,
f
= 0.36 on silica, same solvent system) were
1
stant titration values against methyl orange were obtained, the re-
action was quenched by acidification (aq. HCl) and extracted with
diethyl ether (10×30 mL). After drying the combined organic ex-
CDCl
CHH), 6.06 (d, J = 1.7 Hz, 2 H, CHH) ppm. C NMR (75 MHz,
CDCl ): δ = 28.4 [C(CH ], 81.3 [C(CH ], 116.6 (CH ), 126.0 (C-
2, C-3), 165.3 (C-1) ppm.
3 3 3
): δ = 1.42 [s, 18 H, C(CH ) ], 5.60 (d, J = 1.7 Hz, 2 H,
1
3
3
3
)
3
3
)
3
2
2 4
tracts (Na SO ) the solvent was removed. The resulting yellow solid
was recrystallised from acetonitrile (decolourisation by charcoal)
to give the title compound as a white solid (0.21 g, 8% yield). M.p.
Buta-1,3-diene-2,3-dicarboxylic Acid (5): Trifluoroacetic acid (TFA)
1.0 mL) was added to di-tert-butyl buta-1,3-diene-2,3-dicarboxyl-
(
[
7]
[12]
136–136.5). 1H NMR
1
(
30–132 °C (lit. m.p. 136–136.5, lit.
300 MHz, CD OD): δ = 3.71 (dd, J = 3.0 and 7.0 Hz, 2 H,
H), 5.93 (dt, J = 2.0 and 11.5 Hz, 1 H, CH), 6.52 (dt, J =
ate (0.16 g, 0.63 mmol) in DCM (2 mL). The resulting solution was
stirred at room temperature for 20 min. The solvent was removed
and the title compound was obtained as a white solid (0.08 g, 90%
yield). Recrystallisation from acetonitrile gave an analytically pure
3
2 2
CH CO
13
7
.0 and 11.6 Hz, 1 H, CH) ppm. C NMR [δ
δ = 35.2 (C-4), 123.5 (C-3), 142.2 (C-2), 169.5 (C-1), 174.7 (C-5)
ppm. C : calcd. C 46.16, H 4.65; found C 45.99, H 4.62. IR
KBr disc): ν˜ = 3066 (m), 1686 (s), 1649 (s) cm .
C 3
(75 MHz, CD OD)]:
sample. M.p. 184–186 °C (lit.[ m.p. 186–188, lit. 192–192.5). H
13]
[8]
1
5
6 4
H O
NMR (300 MHz, CD
.22 (d, J = 1.5 Hz, 2 H, CHH) ppm. 13C NMR (75 MHz,
CD OD): δ = 128.1 (CH ), 140.1 (C-2, C-3), 167.1 (C-1) ppm. IR
3
OD): δ = 5.85 (d, J = 1.5 Hz, 2 H, CHH),
–1
(
6
tert-Butyl 2,3-Dibromopropionate (9): Bromine (2.9 ) in DCM
10 mL) was added dropwise to tert-butyl acrylate (2.0 g,
5.6 mmol) in DCM (20 mL), cooled below 10 °C over 20 min. The
3
2
–
1
(
1
(KBr disc): ν˜ = 3066 (m), 1675 (s), 1614 (s) cm .
mixture was stirred at ca. 5 °C for 2 h at which stage an orange
Acknowledgments
colour persisted. The solvent was removed to afford the title com-
1
pound as a colourless oil (4.41 g, 90% yield). H NMR (300 MHz,
We are grateful to the European Commission, Deutsche For-
schungsgemeinschaft, and Fonds der Chemischen Industrie for
funding this work.
CDCl
H, CHHBr), 3.81 (dd, J = 9.8 and 11.3 Hz, 1 H, CHHBr), 4.27
dd, J = 4.4 and 11.3 Hz, 1 H, CHBr
) ppm. 13C NMR (75 MHz,
CDCl ): 28.1 [C(CH ], 41.0 (C-3), 43.3 (C-2), 83.9
C(CH ], 166.8 (C-1) ppm.
3 3 3
): δ = 1.43 [s, 9 H, C(CH ) ], 3.57 (dd, J = 4.4 and 9.8 Hz 1
(
2
3
δ
=
3 3
)
[
3 3
)
[1] R. Banerjee, Chemistry and Biochemistry of B12, John Wiley &
Sons Inc., New York, 1999.
tert-Butyl 2-Bromoacrylate (10): Triethylamine (0.77 g, 0.80 mL,
[
[
2] K. L. Brown, Chem. Rev. 2005, 105, 2075–2149.
3] W. Buckel, C. Kratky, B. T. Golding, Chem. Eur. J. 2006, 12,
7
2
.7 mmol) in diethyl ether (5 mL) was added dropwise to tert-butyl
,3-dibromopropionate (3.0 g, 7.0 mmol) in dry diethyl ether
352–362.
(
15 mL), cooled to 0 °C . The mixture was stirred overnight at room
temperature and then filtered. The filtrate was washed with water
3×10 mL), dried (Na SO ) and the solvent was removed. The title
[
4] W. Buckel, G. Bröker, H. Bothe, A. J. Pierik, B. T. Golding, in
Chemistry and Biochemistry of B12, John Wiley & Sons, Inc.,
New York, 1999, p. 757.
[5] A. J. Pierik, D. Ciceri, R. F. Lopez, F. Kroll, G. Bröker, B. Be-
atrix, W. Buckel, B. T. Golding, Biochemistry 2005, 44, 10541–
10551.
(
2
4
compound was obtained as a pale yellow oil for which no further
1
purification was needed (1.72 g, 80% yield). H NMR (300 MHz,
CDCl
.77 (d, J = 1.5 Hz, 1 H, CHH) ppm. C NMR (75 MHz, CDCl
δ = 28.3 [C(CH ], 83.4 [C(CH ], 123.8 (C-3), 129.2 (C-2), 161.1
C-1) ppm.
3 3 3
): δ = 1.45 [s, 9 H, C(CH ) ], 6.13 (d, J = 1.5 Hz, 1 H, CHH),
13
[6] M. Newcomb, N. Miranda, J. Am. Chem. Soc. 2003, 125, 4080–
6
3
):
4086.
3
)
3
3 3
)
[
[
7] C. Rappe, R. Adeström, Acta Chem. Scand. 1965, 19, 383–389.
8] M. F. Semmelhack, P. Helquist, L. D. Jones, L. Keller, L. Men-
delson, L. S. Ryono, J. G. Smith, R. D. Stauffer, J. Am. Chem.
Soc. 1981, 103, 6460–6471.
(
Di-tert-butyl Buta-1,3-diene-2,3-dicarboxylate (11): Bis(1,5-cyclooc-
tadiene)nickel(0) (1.5 g, 5.3 mmol) and triphenylphosphane (2.8 g,
[
9] M. S. Huhta, D. Ciceri, B. T. Golding, E. N. G. Marsh, Bio-
chemistry 2002, 41, 3200–3206.
10.6 mmol) were suspended in dry diethyl ether (20 mL) at 25 °C
under nitrogen in a flame-dried Schlenk tube. Neat tert-butyl 2-
bromoacrylate (1.87 g, 8.9 mmol) was added rapidly via syringe
and the reaction mixture was stirred at 25 °C for 4.5 h. The mixture
was diluted with diethyl ether (20 mL), washed with water
[
10] K. H. Tang, C. H. Chang, P. A. Frey, Biochemistry 2001, 40,
5190–5199.
[
[
[
11] M. D. Vlasie, R. Banerjee, Biochemistry 2004, 43, 8410–8417.
12] R. Malachowski, Ber. Dtsch. Chem. Ges. 1929, 62, 1323–1326.
13] S. G. Deshpande, N. P. Argade, Synthesis 1999, 8, 1306–1308.
Received: May 3, 2006
(
2×25 mL), dried (MgSO
crude product was purified by medium pressure chromatography
silica, elution with 5% ethyl acetate in petrol). The product con-
4
) and the solvent was removed. The
(
Published Online: August 2, 2006
3626
www.eurjic.org
© 2006 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Eur. J. Inorg. Chem. 2006, 3622–3626

 Buckel, Wolfgang
Buckel, Wolfgang
 Pierik, Antonio J.
Pierik, Antonio J.
 Plett, Sandra
Plett, Sandra
 Alhapel, Ashraf
Alhapel, Ashraf
 Suarez, Diana
Suarez, Diana
 Tu, Shang-Min
Tu, Shang-Min
 Golding, Bernard T.
Golding, Bernard T.