Photochemistry and Photobiology, 2009, 85 691
0.1) · 107
M
)1s)1 is obtained for clomazone
2. Hodgson, E. and P. E. Levi (1996) Pesticides: An important but
underused model for the environmental health sciences. Environ.
Health Persp. 104, 97–106.
3. Frimmel, F. H., H. Bauer, J. Putzlen, P. Murasecco and
A. M. Braun (1987) Laser flash photolysis of dissolved aquatic
humic material and the sensitized production of singlet oxygen.
Environ. Sci. Technol. 21, 541–545.
4. Paul, A., S. Hackbarth, R. D. Vogt, B. Roder, B. K. Burnison and
C. E. W. Steinberg (2004) Photogeneration of singlet oxygen by
humic substances: Comparison of humic substances of aquatic
and terrestrial origin. Photochem. Photobiol. Sci. 3, 273–280.
kr = (5.4
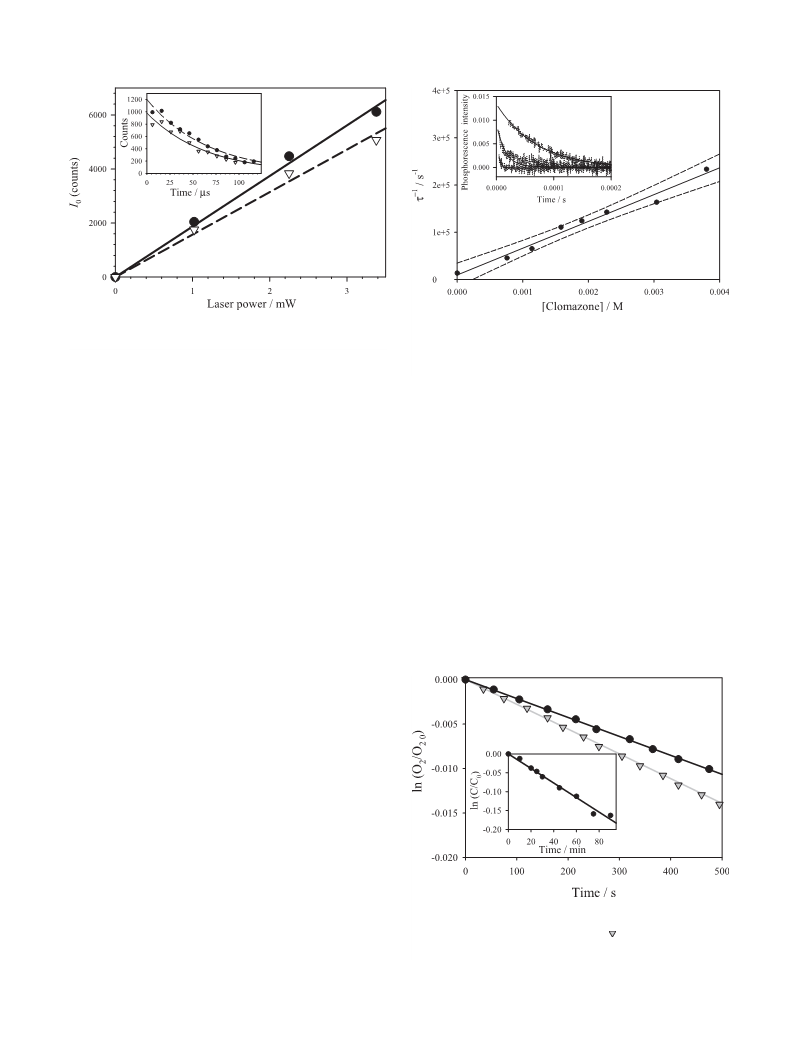
(Fig. 7). The coincidence, within the experimental error, of kt
and kr means that the quenching of O2(a1Dg) by clomazone is
mainly of chemical nature.
Determination of reaction products in steady state experiments
(k > 300 nm). The reaction products of the AHA photosen-
sitized (k > 300 nm) degradation of clomazone in experiments
performed in either air- or O2-saturated solutions are 2-chlo-
robenzylic alcohol and 2-chlorobenzaldehyde (products A and
B in Table 1).
Singlet oxygen by virtue of its electrophilic character reacts
efficiently at electron pair-bearing heteroatom centers (43). The
interaction leads to breaking of the heterocycle, in agreement
with the lack of this ring in the products detected (44).
5. Haag, W. R. and J. Hoigne (1986) Singlet oxygen in surface
´
waters. 3. Photochemical formation and steady state concentra-
tions in various types of water. Environ. Sci. Technol. 20, 341–
348.
6. Goldstone, J. V. and B. M. Voelker (2000) Chemistry of super-
oxide radical in seawater: CDOM associated sink of superoxide in
coastal waters. Environ. Sci. Technol. 34, 1043–1048.
7. Scully, N. M., D. R. S. Lean, W. J. Cooper and D. J. McQueen
(1996) Hydrogen peroxide formation interaction of UV radiation
and dissolved organic carbon in lake waters along a 43–75ꢁN
gradient. Limnol. Oceanogr. 41, 540–548.
CONCLUSIONS
8. Herat, B., E. Shoham-Frider, N. Kress, U. Fiedler and
D. L. Angel (1998) Hydrogen peroxide production rates in clean
and polluted coastal marine waters of the Mediterranean, Red and
Baltic Seas. Mar. Pollut. Bull. 36, 994–1003.
9. Vaughan, P. and N. V. Blough (1998) Photochemical formation of
hydroxyl radical by constituents of natural waters. Environ. Sci.
Technol. 32, 2947–2953.
10. Cooper, W. J., R. G. Zika, R. G. Petasne and A. M. Fischer
(1989) Sunlight-induced photochemistry of humic substances in
natural waters: Major reactive species. In Aquatic Humic Sub-
stances: Influence on Fate and Treatment of Pollutants (Edited by I.
H. Suffet and P. MacCarthy), pp. 333–362. American Chemical
Society, Washington, DC.
11. Richard, C., D. Vialaton, J. P. Aguer and F. Andreux (1997)
Transformation of monuron photosensitized by soil extracted
humic substances: Energy or hydrogen transfer mechanism?
J. Photochem. Photobiol. A: Chem. 111, 265–271.
The results of this study suggest that ISCO treatments with
Na2S2O8, which involve the reaction of SO4•) radicals with the
contaminant, should be efficient for degrading the herbicide
clomazone.
As with other herbicides or pesticides (45), direct sunlight-
mediated photodegradation of clomazone is a minor event (see
Fig. 3 for photolysis with k > 300 nm), which leads to the
problem of their long-term presence in the environment.
Instead, clomazone is more likely to be effectively degraded
in sunlit irradiated natural waters containing appreciable
amounts of HS mainly through a O2(a1Dg)-mediated oxida-
tion. Despite its short lifetime of about 4 ls in water, O2(a1Dg)
may present steady state concentrations which can exceed the
value of 10)14 M (34).
12. Liang C., C. J. Bruell., M. C. Marley and K. L. Sperry (2004)
ISCO persulfate oxidation for in situ remediation of TCE.
II. Activated by chelated ferrous ion. Chemosphere 55, 1225–
1233.
13. Anipsitakis, G. P. and D. D. Dionysiou (2003) Degradation of
organic contaminants in water with sulfate radicals generated by
the conjunction of peroxymonosulfate with cobalt. Environ. Sci.
Technol. 37, 4790–4797.
Acknowledgements—This research was supported by ANPCyT, Argen-
tina (PICT 2003 06-14508), CONICET and CICPBA. The work at the
University of Aarhus was supported by the Danish National Research
Foundation through the Center for Oxygen Microscopy and Imaging.
The authors thank the CAPES-SECYT cooperation program and the
DAAD Alumni Program for an equipment grant. P.D.G. thanks
14. House, D. A. (1962) Kinetics and mechanism of oxidations by
peroxydisulfate. Chem. Rev. 62, 185–203.
Fundacion YPF for a graduate studentship. M.C.G. and D.O.M. are
´
research members of CONICET and CICPBA, respectively.
15. Noldin, J. A., D. S. Ederhadt, F. C. Deschamps and L. C. Hermes
(2001) Strategies for Water Sampling for Monitoring the Rice
Enviromental Impact. Proceedings of the 2nd Brazilian Congress of
the Irrigated Rice, Instituto Riograndense do Arroz, Porto Alegre,
Brazil, August 20–23, 2001.
16. Mervosh, T. L., G. K. Sims and E. W. Stollert (1995) Clomazone
fate in soil as affected by microbial activity, temperature, and soil
moisture. J. Agr. Food Chem. 43, 537–543.
17. Marchesan, E., R. Zanella, L. A. de Avila, E. Rabaioli Camargo,
S. L. de Oliveira Machado and V. R. Mussoi Macedo (2007) Rice
herbicide monitoring in two Brazilian rivers during the rice
growing season. Sci. Agric. 64, 131–137.
SUPPORTING INFORMATION
Additional Supporting Information may be found in the online
version of this article:
Figure S1. (A) FTIR spectra of AHA (upper trace) and the
VCE (lower trace); (B) UV–visible spectra of the 20 ppm AHA
solution (upper trace) and 20 ppm VCE solution (lower trace);
(C) EEM of the VCE in solution.
18. David Gara, P. M., G. N. Bosio, M. C. Gonzalez and D. O. Martire
´
(2008) Kinetics of the sulfate radical-mediated photooxidation of
Please note: Wiley-Blackwell are not responsible for the
content or functionality of any supporting materials supplied
by the authors. Any queries (other than missing material)
should be directed to the corresponding author for the
article.
humic substances. Int. J. Chem. Kinet. 40, 19–24.
19. Bosio, G. N., P. M. David Gara, F. S. Garcı
´
M. C. Gonzalez, M. T. del Panno and D. O. Ma
a Einschlag,
rtire (2008)
´
Photodegradation of soil organic matter and its effect on
Gram-negative bacterial growth. Photochem. Photobiol. 84, 1126–
1132.
20. Zanella, R., E. G. Primel, F. F. Gonc¸ alves, M. L. Martins, M. B.
REFERENCES
Adaime, E. Marchesan and S. L. O. Machado (2008) Study of the
degradation of the herbicide clomazone in distilled and in irrigated
rice field waters using HPLC-DAD and GC-MS. J. Braz. Chem.
Soc. 19, 987–995.
1. Cabras, P. and A. Angioni (2000) Pesticide residues in grapes,
wine, and their processing products. J. Agric. Food. Chem. 48,
967–973.

 David Gara, Pedro M.
David Gara, Pedro M.
 Bosio, Gabriela N.
Bosio, Gabriela N.
 Arce, Valeria B.
Arce, Valeria B.
 Poulsen, Lars
Poulsen, Lars
 Ogilby, Peter R.
Ogilby, Peter R.
 Giudici, Reinaldo
Giudici, Reinaldo
 Gonzalez, Monica C.
Gonzalez, Monica C.
 Martire, Daniel O.
Martire, Daniel O.