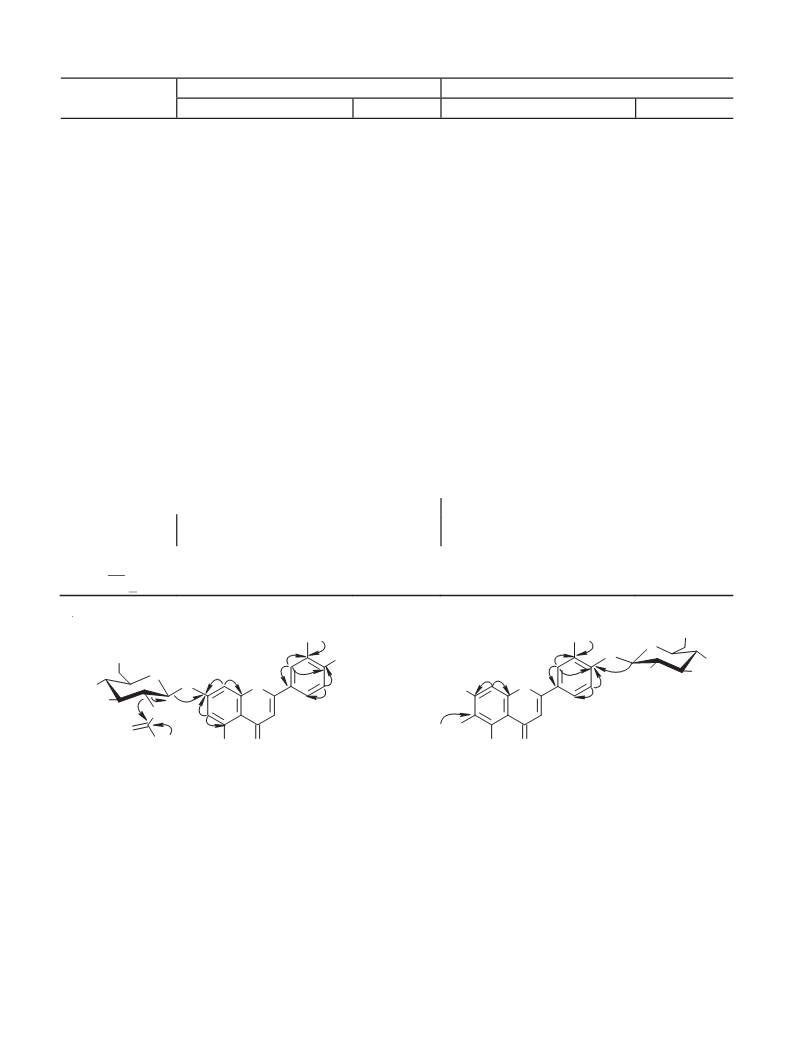
2
6 mg, 14, prep. HPLC t = 11.0–12.0 min) [14], and 6-hydroxyluteolin-7-O-glucoside (14 mg, 15, prep. HPLC
t = 10.0–10.5 min) [15].
Chrysoeriol-7-O-(2″-O-acetyl)-β-D-glucopyranoside (1). C H O . UV spectrum (70% EtOH, λmax, nm): 270,
2
4 24 12
–
1
–
3
42. IR spectrum (ν, cm ): 1642, 1725. HR-ESI-MS, m/z 503.2471 [M – H] (calcd 503.3978); (+)ESI-MS, m/z: 505
M + H] , 527 [M + Na] ; (–)ESI-MS, m/z: 503 [M – H] , 461 [(M – H) – C H O] , 299 [(M – H) – C H O – C H O ] .
Í NMR spectrum (500 MHz, DMSO-d , 298 K, δ, ppm) and Ñ NMR spectrum (125 MHz, DMSO-d , 298 K, δ, ppm) are
+
+
–
–
–
[
2
2
2
2
6 10 5
1
13
6
6
given in Table 1.
Acid Hydrolysis of 1. Compound 1 (2 mg) and TFA (5%, 2 mL) were heated at 110°C (2 h). The hydrolysate was
concentrated under vacuum with MeOH and chromatographed over polyamide (CC, 5 g) with elution sequentially by H O
2
(
50 mL, eluate I) and MeOH (70%, 50 mL, eluate II). The eluates were analyzed by anal. HPLC (conditions 1, monosaccharides
as derivatives with 3-methyl-1-phenyl-2-pyrazolin-5-one [25]; conditions 2, flavonoids). Eluate I was also analyzed to determine
D- and L-type monosaccharides after derivatization with L-tryptophan [26]. After hydrolysis of 1 and 2, D-glucose was
detected in eluates I (tR = 12.51–12.53 min); chrysoeriol, in eluate II from 1 (tR = 13.10 min); jaceosidin, from 2
(
tR = 14.67 min).
Alkaline Hydrolysis of 1. Compound 1 (2 mg) was dissolved in MeOH (1 mL), treated with NaHCO solution (3%.
3
2
50 μL), incubated at 40°C for 2 h, diluted with H O (1:2), neutralized with AcOH, and extracted with Et O (2 × 5 mL).
2
2
The Et O layer was concentrated to dryness. The dry residue was dissolved in MeOH (50%, 2 mL). The resulting solution
2
was separated over polyamide (10 g) with elution by H O (50 mL, eluate I) and MeOH (60%, 80 mL, eluate II). Eluate II was
2
concentrated to produce a compound (0.8 mg) that was identified by UV and NMR spectroscopy and mass spectrometry as
chrysoeriol-7-O-β-D-glucopyranoside (11).
Chrysoeriol-7-O-β-D-glucopyranoside (11). C H O . UV spectrum (70% EtOH, λmax, nm): 269, 339.
2
2 22 11
–
+
+
HR-ESI-MS, m/z: 461.4913 [M – H] (calcd 461.3640). (+)ESI-MS, m/z: 463 [M + H] , 485 [M + Na] ; (–)ESI-MS, m/z: 461
–
– 1
[
M – H] , 299 [(M – H) – C H O ] . Í NMR spectrum (500 MHz, DMSO-d , 298 K, δ, ppm, J/Hz): chrysoeriol – 3.92 (3Í,
6 10 5 6
s, 3′-ÎÑÍ ), 6.42 (1Í, d, J = 2.2, H-6), 6.79 (1Í, d, J = 2.2, H-8), 6.88 (1Í, s, H-3), 6.94 (1Í, d, J = 8.1, H-5′), 7.53 (1Í, d,
3
J = 2.0, H-2′), 7.85 (1Í, dd, J = 2.0, 8.1, H-6′), 10.06 (1Í, br.s, 4′-OH), 10.63 (1Í, br.s, 7-OH), 13.09 (1Í, br.s, 5-OH);
7
-O-β-D-glucopyranose – 3.02–3.12 (2Í, m, H-4′′, 5′′), 3.16 (1Í, m, H-2′′), 3.20 (1Í, m, H-3′′), 3.46 (1H, m, H-6′′ ), 3.61 (1H,
b
1
3
d, J = 11.2, H-6′′ ), 5.41 (1H, d, J = 7.1, H-1′′). C NMR spectrum (125 MHz, DMSO-d , 298 K, δ, ppm): chrysoeriol – 164.7
a
6
(C-2), 103.0 (C-3), 182.4 (C-4), 160.9 (C-5), 100.3 (C-6), 162.5 (C-7), 95.0 (C-8), 157.1 (C-9), 104.8 (C-10), 122.4 (C-1′),
1
10.2 (C-2′), 148.7 (C-3′), 164.2 (C-4′), 115.2 (C-5′), 120.5 (C-6′), 59.6 (3′-ÎÑÍ ); 7-O-β-D-glucopyranose – 99.6 (C-1′′), 72.9
3
(C-2′′), 77.8 (C-3′′), 69.7 (C-4′′), 76.7 (C-5′′), 60.3 (C-6′′).
Jaceosidin-4′-O-β-D-glucopyranoside (2). C H O . UV spectrum (70% EtOH, λ , nm): 277, 335. HR-ESI-MS,
2
3
24 12
max
–
+
+
–
m/z 491.2611 [M – H] (calcd 491.3882); (+)ESI-MS, m/z: 493 [M + H] , 515 [M + Na] . (–)ESI-MS, m/z: 491 [M – H] , 329
–
1
13
[
(M – H) – C H O ] . Í NMR spectrum (500 MHz, DMSO-d , 298 K, δ, ppm) and C NMR spectrum (125 MHz,
6 10 5 6
DMSO-d , 298 K, δ, ppm) are given in Table 1.
6
–
Jaceosidin. C H O . UV spectrum (70% EtOH, λ , nm): 275, 345. HR-ESI-MS, m/z 329.4107 [M – H]
1
7
14
7
max
+
+
– 1
(
(
calcd 329.2576). (+)ESI-MS, m/z: 331 [M + H] , 353 [M + Na] . (–)ESI-MS, m/z 329 [M – H] . Í NMR spectrum
500 MHz, DMSO-d , 298 K, δ, ppm, J/Hz): 3.76 (3Í, s, 6-ÎÑÍ ), 3.87 (3Í, s, 3′-ÎÑÍ ), 6.63 (1Í, s, H-8), 6.87 (1Í, s,
6
3
3
H-3), 7.12 (1Í, d, J = 8.8, H-5′), 7.51 (1Í, d, J = 2.0, H-2′), 7.69 (1Í, dd, J = 2.0, 8.8, H-6′), 10.11 (1Í, br.s, 4′-OH), 10.69
1
3
(
(
1Í, br.s, 7-OH), 13.09 (1Í, br.s, 5-OH). C NMR spectrum (125 MHz, DMSO-d , 298 K, δ, ppm): 163.4 (C-2), 102.5
6
C-3), 182.4 (C-4), 152.3 (C-5), 131.7 (C-6), 157.0 (C-7), 94.2 (C-8), 152.1 (C-9), 104.0 (C-10), 122.4 (C-1′), 109.7 (C-2′),
1
50.5 (C-3′), 148.2 (C-4′), 116.1 (C-5′), 120.4 (C-6′), 59.6 (6-OCH ), 55.8 (3′-OCH ).
3 3
Analytical HPLC. Conditions 1: mobile phase NH OAc (100 mM, pH 4.5) (A), MeCN (B); gradient mode (%B):
4
0
–20 min, 20–26%; flow rate 150 μL/min; column temperature 35°C, UV detector at λ = 250 nm. Retention times of reference
standards (t , min): mannose, 6.83; glucose, 12.52; galactose, 13.54. Conditions 2: mobile phase LiClO (0.2 M) in HClO
R
4
4
(
0.006 M) (A), MeCN (B); gradient mode (%B): 0–18 min, 25–100%, 18–20 min 100%; flow rate 150 μL/min;
column temperature 35°C; UV detector at λ = 270 nm. Retention times of reference standards (t , min): chrysoeriol, 13.09;
R
jaceosidin, 14.65.
Anti-α-glucosidase activity was determined using α-glucosidase from Saccharomyces cerevisiae (No. G5003,
≥
10 U/mg, Sigma-Aldrich) using a microplate spectrophotometric method as described before [27]. Inhibition efficiency was
calculated as the 50% inhibitory activity of α-glucosidase (IC ). Results were given as means of six independent determinations
5
0
(
± standard deviation, S.D.) in μM. Quercetin (≥95%, No. E2250, Sigma-Aldrich) was used as a reference compound.
626

 Olennikov
Olennikov