1976
V. Cantonetti et al. / Tetrahedron: Asymmetry 15 (2004) 1969–1977
c) Schenning, A. P. H. J.;Hubert, D. H. W.;van Esch,
(
13. For some examples of aggregation of porphyrin deriva-
tives in aqueous solutions see: (a) Pasternack, R. F.;
Huber, P. R.;Boid, P.;Engasser, G.;Francesconi, L.;
Gibbs, E.;Fasella, P.;Cerio Venturo, G.;deC. Hinds, L.
J. Am. Chem. Soc. 1972, 94, 4511–4517;(b) Guilard, R.;
Senglet, N.;Liu, Y. H.;Sazou, D.;Findsen, E.;Faure, D.;
Des Curieres, T.;Kadish, K. M. Inorg. Chem. 1991, 30,
1898–1905;(c) Schenning, A. P. H. J.;Feiters, M. C.;
Nolte, R. J. M. Tetrahedron Lett. 1993, 34, 7077–7078;(d)
J. H.;Feiters, M. C.;Nolte, R. J. M. Angew. Chem., Int.
Ed. Engl. 1994, 33, 2468–2470;(d) van Esch, J. H.;Roks,
M. F. M.;Nolte, R. J. M. J. Am. Chem. Soc. 1986, 108,
6093–6094.
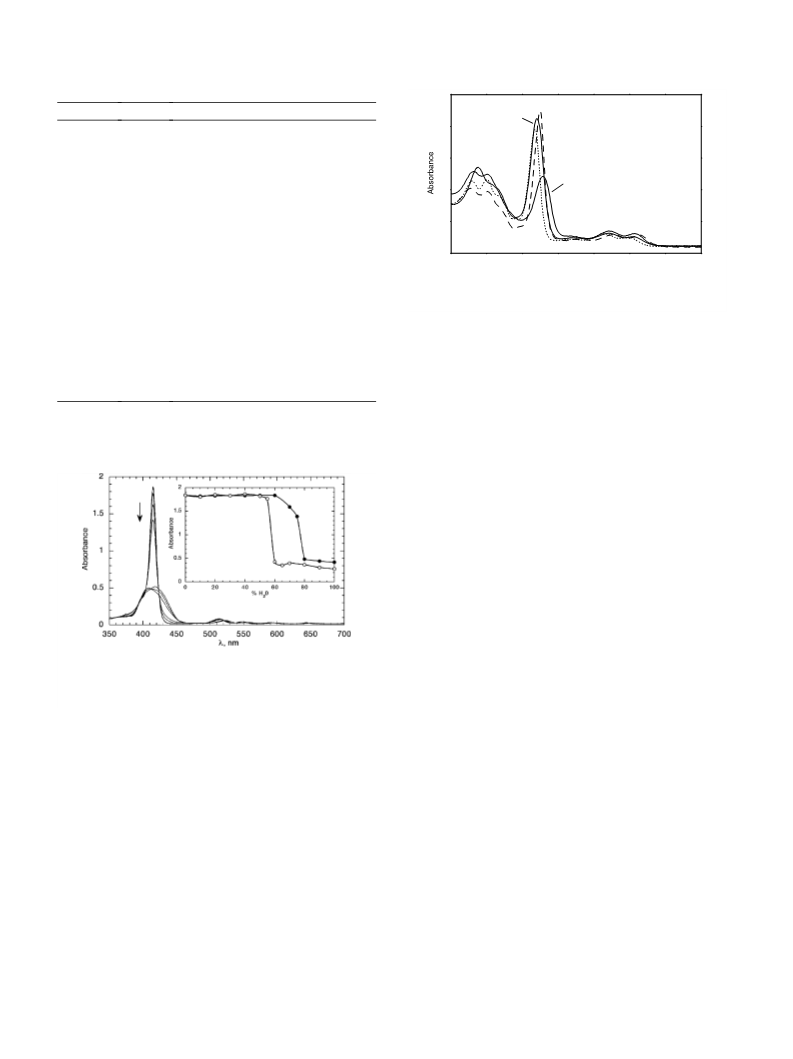
5
6
. (a) Monti, D.;Tagliatesta, P.;Mancini, G.;Boschi, T.
Angew. Chem., Int. Ed. 1998, 37, 1131–1133;(b) Borocci,
S.;Marotti, F.;Mancini, G.;Monti, D.;Pastorini, A.
Langmuir 2001, 17, 7198–7203;(c) Monti, D.;Pastorini,
A.;Mancini, G.;Borocci, S.;Tagliatesta, P. J. Mol. Catal.
A 2002, 179, 125–131.
. (a) Schenning, A. P. H. J.;Hubert, D. H. W.;Feiters, M.
C.;Nolte, R. J. M. Langmuir 1996, 12, 1572–1577;(b)
Schell, C.;Hombrecher, H. K. Chem. Eur. J. 1999, 5, 587–
Kano, K.;Minamizono, H.;Kitae, T.;Negi, S.
J. Phys.
Chem. A 1997, 101, 6118–6124;(e) Rib oꢁ , J. M.;Bofill, J.
M.;Crusats, J.;Rubires, R. Chem. Eur. J. 2001, 7, 2733–
2737.
ꢀ3
14. The cmc of SDS is 8.2 · 10 M. See, for example, (a)
Fendler, J. H.;Fendler, E. J. Catalysis in Micellar and
Macromolecular Systems;Academic: New York, 1975;(b)
598;(c) Vermathen, M.;Luie, E. A.;Chodosh, A. B.;
Ried, S.;Simonis, U. Langmuir 2000, 16, 210–221;(d)
ꢀ3
Zhang, Y.-H.;Guo, L.;Ma, C.;Li, Q.-S.
Chem. Phys. 2001, 3, 583–587.
Phys. Chem.
The cmc of L-SDP has been reported to be 3.0 · 10
(see Ref. 13b). We found, by means of various experi-
M
ꢀ2
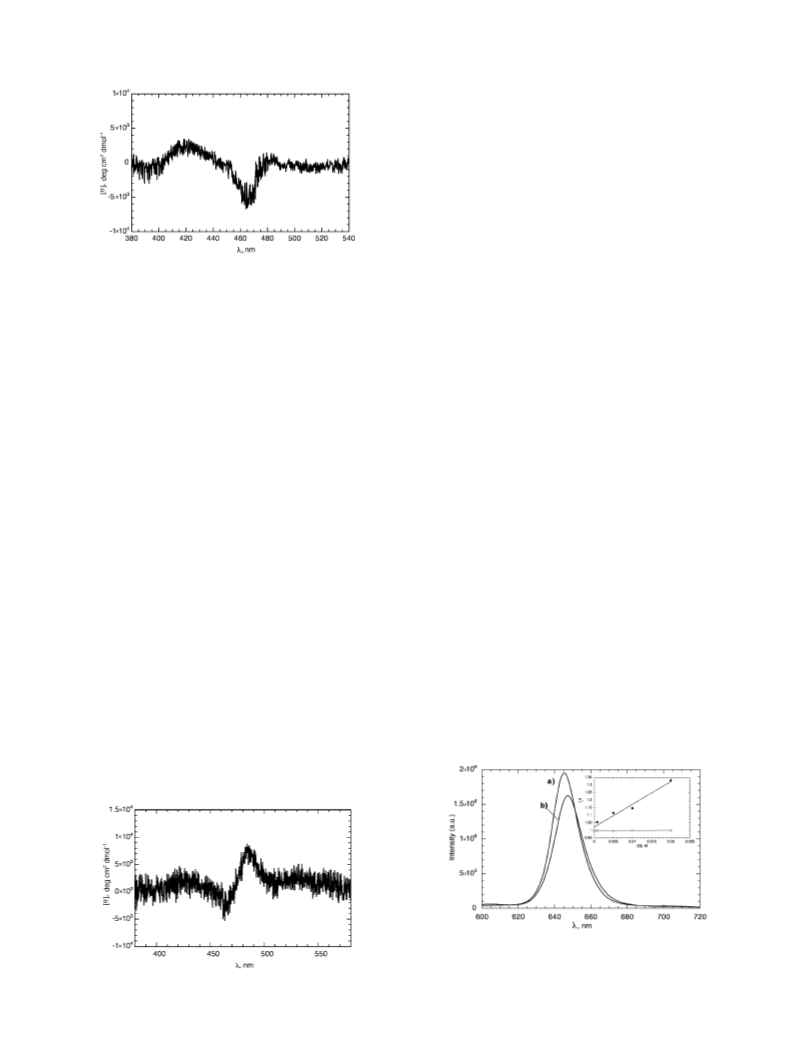
7
8
. (a) Ricchelli, F. J. Photochem. Photobiol. B: Biol. 1995, 29,
09–118;(b) Ricchelli, F.;Gobbo, S. J. Photochem.
mental techniques, a value of 1.0 · 10 M, which has been
consistently used throughout the work.
1
Photobiol. B: Biol. 1995, 29, 65–70, and references cited
therein;(c) Monti, D.;Venanzi, M.;Cantonetti, V.;
Borocci, S.;Mancini, G. J. Chem. Soc., Chem. Commun.
15. It has been reported that the inclusion of porphyrin
derivatives in SDS micelle occurs with significative degree
of aggregation. See, for example: Mons uꢁ Scolaro, L.;
Donato, C.;Castriciano, M.;Romeo, A.;Romeo, R.
Inorg. Chim. Acta 2000, 300–302, 978–986.
16. For a recent survey on Induced Circular Dichroism in
micellar systems see: Allenmark, S. Chirality 2003, 15,
409–422, and references cited therein.
17. The presence of a strong hypochromic effect along with the
presence of coupled CD bands are usually taken as an
evidence for the formation of porphyrin chiral aggregates.
See for example: Fuhrhop, J.-H.;Demoulin, C.;Boettcher,
C.;K o€ ning, J.;Siggel, U. J. Am. Chem. Soc. 1992, 114,
4159–4165.
18. (a) Blatt, E.;Sawyer, W. H. Biochim. Biophys. Acta 1985,
822, 43–62;(b) van Esch, J. H.;Feiters, M. C.;Peters, A.
M.;Nolte, J. M. J. Phys. Chem. 1994, 98, 5541–5551;(c)
Steiner, R. F. In Topics in Fluorescence Spectroscopy Vol.
2: Principles;Lakowicz, J. R., Ed.;Plenum: New York,
1991, Chapter 1;(d) Eftink, M. R. Biophysical and
Biochemical Aspects of Fluorescence Spectroscopy;Ple-
num: New York, 1987.
2002, 774–775.
. (a) Monti, D.;Cantonetti, V.;Venanzi, M.;Ceccacci, F.;
Bombelli, C.;Mancini, G. J. Chem. Soc., Chem. Commun.
2004, 972–973;For some recent, important examples, on
the interaction of porphyrin derivatives with DNAs or
other chiral polymeric matrices see: (b) Pasternack, R. F.;
Gibbs, E. J.;Bruzewicz, D.;Stewart, D.;Engstrom, K. S.
J. Am. Chem. Soc. 2002, 124, 3533–3539;(c) Pasternack,
R. F.;Ewen, S.;Rao, A.;Meyer, A. S.;Freedman, M. A.;
Collings, P. J.;Frey, S. L.;Ranen, M. C.;de Paula, J. C.
Inorg. Chim. Acta 2001, 317, 59–71;(d) Purrello, R.;
Mons uꢁ Scolaro, L.;Bellacchio, E.;Guerrieri, S.;Romeo,
A. Inorg. Chem. 1998, 37, 3647–3648;(e) Bellacchio, E.;
Lauceri, R.;Gurrieri, S.;Mons uꢀ Scolaro, L.;Romeo, A.;
Purrello, R. J. Am. Chem. Soc. 1998, 120, 12353–12354;(f)
Purrello, R.;Bellacchio, E.;Guerrieri, S.;Lauceri, R.;
Raudino, A.;Mons uꢀ Scolaro, L.;Santoro, A. M. J. Phys.
Chem. B 1998, 102, 8852–8857;(g) Pasternack, R. F.;
Gurrieri, S.;Lauceri, R.;Purrello, R. Inorg. Chim. Acta
1
996, 246, 7–12;Polymer-anchored porphyrin nanorods
19. In previous papers, the notation trans- or cis-epoxides, has
been used, referring to the relative stereochemistry of
1-methylene and the 4-isopropenyl substituents. See, for
example, Battioni, P.;Renaud, J. P.;Bartoli, J. F.;Reina-
with helical structure have been recently reported: (h) de
Witte, P. A. J.;Castriciano, M.;Cornelissen, J. J. L. M.;
Mons uꢀ Scolaro, L.;Nolte, R. J. M.;Rowan, A. E. Chem.
Eur. J. 2003, 9, 1775–1781;For recent reports on the
chiral-symmetry-breaking in the self-association of achiral
porphyrin derivatives see: (i) Rubires, R.;Ferrera, J.-A.;
Rib oꢁ , J. M. Chem. Eur. J. 2001, 7, 436–446;(j) Borovkov,
V. V.;Harada, T.;Hembury, G. A.;Inoue, Y.;Kuroda, R.
Angew. Chem., Int. Ed. 2003, 42, 1746–1749;For an
example of chiral memory in templated-imprinted chiral
porphyrin aggregates: (k) Lauceri, R.;Raudino, A.;
Mons uꢀ Scolaro, L.;Micali, R.;Purrello, R. J. Am. Chem.
Soc. 2002, 124, 894–895.
Artiles, M.;Fort, M.;Mansuy, D.
J. Am. Chem. Soc.
1988, 110, 8462–8470. We prefer to use the notation syn
and anti referring to the relative stereochemistry of the 1,2-
epoxides and the 4-isopropenyl group.
20. Grassert, I.;Schinkowski, K.;Vollhardt, D.;Oehme, G.
Chirality 1998, 10, 754–759.
21. The extent of chiral induction is well known to largely
depend on the steric hindrance at the metal centre. For
recent discussions on this important aspect of chiral
metalloporphyrin-based catalysts see: (a) Gross, Z.;Ini, S.
J. Org. Chem. 1997, 62, 5514–5521;(b) Tagliatesta, P.;
Bernini, R.;Crestini, C.;Monti, D.;Boschi, T.;Mincione,
E.;Saladino, R. J. Org. Chem. 1999, 64, 5361–5365;(c)
Reginato, G.;Di Bari, L.;Salvadori, P.;Guilard, R. Eur.
J. Org. Chem. 2000, 1165–1171;(d) Boitrel, B.;Bavex-
Chambeno ^ı t, V.;Richard, P. Eur. J. Inorg. Chem. 2002,
1666–1672.
9
. (a) Jungermann, E.;Gerecht, J. F.;Krems, I. J. J. Am.
Chem. Soc. 1956, 78, 172–174;(b) Grassert, I.;Schinkowski,
K.;Vollhardt, D.;Oehme, G. Chirality 1998, 10, 754–759.
0. Bodanski, M.;Bodanski, A. The Practice of Peptide
Synthesis. In Reaction and Structure Concepts in Organic
Chemistry;Springer: Berlin, 1984, and references cited
therein.
1
1
1
1. (a) Kobayashi, M.;Ohno, M. J. Org. Chem. 1990, 55,
22. (a) Belogi, G.;Croce, M.;Mancini, G. Langmuir 1997, 13,
2903–2904;(b) Borocci, S.;Erba, M.;Mancini, G.;
Scipioni, A. Langmuir 1998, 14, 1960–1962;(c) Bella, J.;
Borocci, S.;Mancini, G. Langmuir 1999, 15, 8025–8031;
(d) Borocci, S.;Ceccacci, F.;Galantini, L.;Mancini, G.;
Monti, D.;Scipioni, A.;Venanzi, M. Chirality 2003, 15,
1169–1177;(b) Shiozaki, M.;Arai, M. J. Org. Chem. 1989,
54, 3754–3755;(c) Kikugawa, Y. J. Org. Chem. 1994, 59,
929–931.
2. Gouterman, M. In The Porphyrins;Dolphin, D., Ed.;
Academic: New York, 1978;Vol. 3. Chapter 1.

 Cantonetti, Veronica
Cantonetti, Veronica
 Monti, Donato
Monti, Donato
 Venanzi, Mariano
Venanzi, Mariano
 Bombelli, Cecilia
Bombelli, Cecilia
 Ceccacci, Francesca
Ceccacci, Francesca
 Mancini, Giovanna
Mancini, Giovanna