Chemical Evolution of Simple Amino Acids to
Asparagine under Discharge onto the Primitive Hydrosphere:
Simulation Experiments Using Contact Glow Discharge
Toratane Munegumi
Department of Science Education, Naruto University of Education,
748 Nakashima, Takashima, Naruto-cho, Naruto, Tokushima 772-8502
E-mail: tmunegumi@naruto-u.ac.jp
Received: June 11, 2014; Accepted: July 17, 2014; Web Released: August 1, 2014
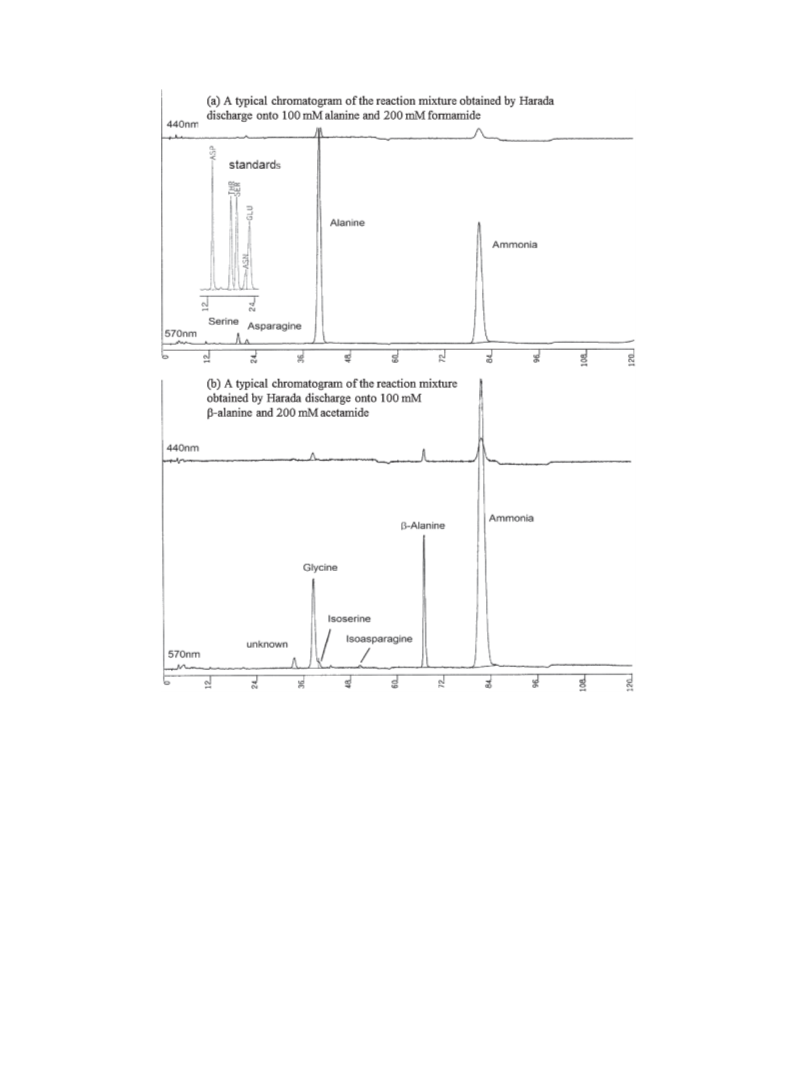
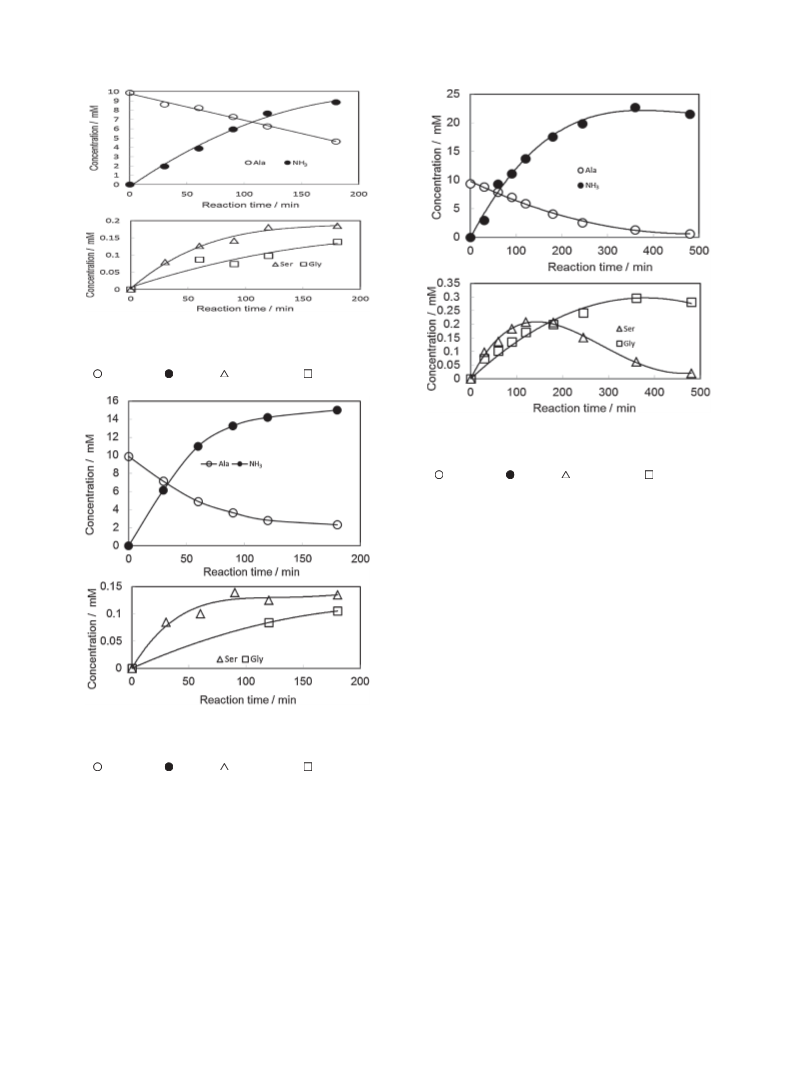
Asparagine is an important amino acid for abiotic polypeptide synthesis. In simulation experiments, it was obtained
in 3.0% yield (based on the amount of consumed alanine) from alanine (100 mM) and formamide (200 mM) by contact
glow discharge (Harada discharge) onto aqueous solutions. The present results suggest that asparagine could be
abiotically synthesized from simple amino acids under possible primitive earth conditions.
Asparagine is a neutral proteinous amino acid that plays a
number of important roles in metabolic pathways, in particular
prebiotic chemistry occurring in the boundary between the
atmosphere and hydrosphere. Harada discharge accelerates
1
3
0,32,33
in the synthesis of oxaloacetic acid via aspartic acid. It is
formed by nucleophilic substitution of 2-aspartyladenosine
monophosphate by ammonia from glutamine in a process
many types of reactions
including amination, carboxy-
lation, reduction, oxidation, hydrogenation, etc. Although
Harada and Iwasaki have reported the amination of succinamic
1
33
catalyzed by asparagine synthetase. Asparagine has a uniquely
acid to asparagine (1.1%), very few examples of asparagine
3
3,34
reactive methylcarboxamide side chain, the amide group of
which serves as a leaving group to form polyaspartic acid
in aqueous solutions2 and to generate polypeptides containing
formation have been reported.
3
4
After the publication of a previous letter, amino acids were
further investigated from the viewpoint of their extraterrestrial
4
5
35
aspartic residues in the solid phase. Asparagine residues of
and terrestrial origins. Glycine, alanine, and serine were
6
10
some proteins cyclize to an imide structure,
which can
found in interstellar medium ice analogs irradiated by ultra-
accelerate epimerization.11 These chemical features of aspar-
violet light.
3639
β-Alanine and γ-aminobutyric acid as well as
agine seem to provide an important pathway of chemically
glycine and alanine were obtained in the hydrolyzates after
high energy proton irradiation to ice mixtures of carbon mon-
consistent evolutionary polypeptide formation2
5,12
under prim-
4
0,41
42
itive earth conditions. However, the pathway for the abiotic
formation of asparagine, as well as glutamine and basic amino
acids, is still unclear, although many simulation experiments
oxide, ammonia, and water.
Bada et al. reported on the
use of the molar ratio between glycine, D-alanine, β-alanine,
and 2-amino-2-methylpropanoic acid as a signature for parent
body processes of carbonaceous chondrites. They applied
Michael addition between ammonia and cyanoacetylene (or
acrylonitrile) as a plausible pathway for explaining β-amino
1
318
have been carried out and discussed.
1
9
Sanchez et al. reported asparagine formation from cyano-
acetylene, which is a typical product in many Miller-type spark
discharge experiments,1
3,14
using gas mixtures containing
acid formation. The significance of asparagine as a chiral-
42
4
3
methane and nitrogen. However, the existence of such a
resolving agent of amino acids was also reported. It was
therefore considered meaningful to investigate the abiotic
formation of asparagine in connection with other amino acids
using simulation experimental methodology.
reducing atmosphere containing methane is disputed.2
026
Non-
reducing conditions in the primitive atmosphere make ammonia
formation difficult, although ammonia, fumaric acid, aspartic
acid, phosphoric acid, and asparagine can exist in equilibrium in
I now report on asparagine formation from simple amino
acids with formamide or acetamide by using Harada dis-
2
7
aqueous solution.
On the other hand, electric discharge (contact glow dis-
2
8,29
charge.
This is a detailed paper that expands on a previ-
2
8,29
34
charge
) into aqueous solutions makes it possible to produce
ously published letter. Asparagine is formed from alanine and
formamide in aqueous solution under Harada discharge as
shown in Figure 1. The three amino acids used for these studies
(alanine, glycine, and β-alanine) are abundant in carbonaceous
30
ammonia from molecular nitrogen, irrespective of whether a
reducing or nonreducing atmosphere is present. Dissociation of
water molecules to hydrogen (H) and hydroxyl (OH) radicals
is a trigger that initiates reducing reactions that yield ammo-
nia and other reducing molecules. Contact glow discharge
hereafter referred to as Harada discharge) was employed in
the study described in this paper for simulative research on
3
1
4
446
meteorites.
The two carboxylic acid amides (formamide
30
32
29
47
and acetamide) are the hydrolysis products of nitriles, which
are thought to be important starting materials for prebiotic
amino acid formation.
(
1
7
© 2014 The Chemical Society of Japan

 Munegumi, Toratane
Munegumi, Toratane