Synthesis and Diels–Alder reactions of a,b-unsaturated g-sultone
Albert W. M. Lee,* W. H. Chan,* L. S. Jiang and K. W. Poon
Department of Chemistry, Hong Kong Baptist University, Kowloon Tong, Kowloon, Hong Kong
An efficient synthesis of prop-1-ene 1,3-sultone 1 and its
Diels–Alder reactions are reported; ring-opening reactions
of the cycloadducts with nucleophiles and transformations
to the sultams were also investigated.
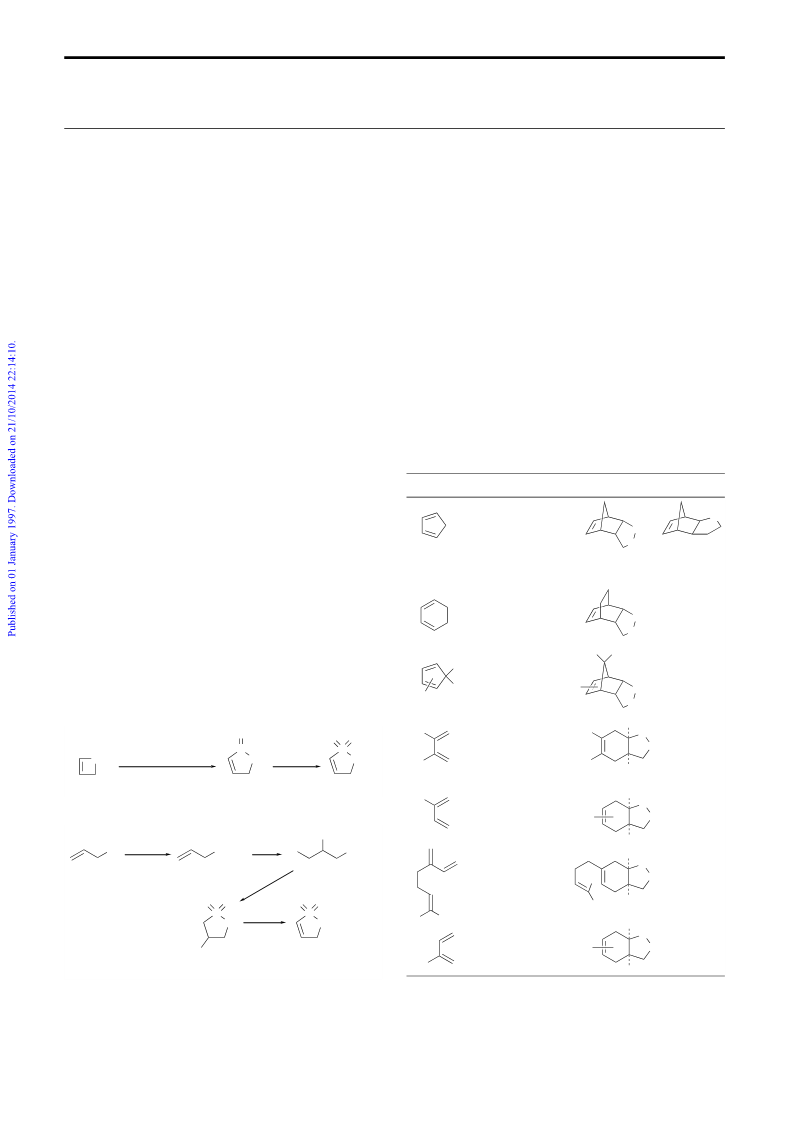
cyclopentadiene, despite the slow reaction rate, reaction with 1
could take place at room temperature with moderate endo:exo
selectivity. At a higher temperature, the reaction could be
completed in 4 h. The endo-6 and exo-7 isomers could be easily
separated by column chromatography. For less reactive dienes,
elevated temperature or sealed tube conditions were used. In the
cases of cyclohexa-1,3-diene and 2,3,4,5-tetrachloro-1,1-dime-
thoxycyclopentadiene, the endo-isomers were the only isolated
products. For unsymmetrical dienes, 1:1 inseparable mixture of
regioisomers resulted.
The sultone cycloadduct could be further manipulated by
ring-opening with various nucleophiles such as alcohols,
amines and thiols at the g-position. Selected examples are
depicted in Schemes 3 and 4. The alkoxide reacted smoothly
with cycloadducts 6 and 8 at room temperature to afford the ring
opening products 9a,b in quantitative yields. Similarly, amine
and thio compounds also reacted with the cycloadducts in
Sultones are cyclic esters of the corresponding hydroxysulfonic
acid, the sulfur analogues of lactones. The preparation and
chemistry of saturated sultones of 4- to 6-membered ring sizes
1
are relatively well studied. However, there is less investigation
of the chemistry of the unsaturated sultones, in particular, the
a,b-unsaturated g-sultone prop-1-ene 1,3-sultone 1. We have
long been interested in the uses of sulfur-containing functional
groups for the activation of acetylenic or vinyl moieties
2
resulting in useful synthons for organic synthesis. We here
report a synthesis of 1, its Diels–Alder reactions and some
further transformations of the cycloadducts.
There are only limited accounts in literature for the synthesis
of 1. Thermolysis of thiete 1,1-dioxide 2 yielded sultine 3 which
3
could be oxidized to 1 (Scheme 1). It had also been reported
Table 1 Diels–Alder reactions of a,b-unsaturated g-sultone
that distillation of 3-hydroxyprop-1-ene-1-sulfonic acid, which
was synthesized from prop-2-ynyl alcohol via free radical
sulfitation, also afforded 1. These reported procedures were not
Dienes
Solvent; T/°C; t/h Adduct (% yield)
4
efficient and practical enough for the laboratory synthesis of the
compound. Our synthesis of 1 is shown in Scheme 2. Sodium
prop-2-enesulfonate 4 could be prepared from allyl bromide and
O2
S
SO2
5
O
sodium sulfite in good yield. Bromination of 4 in water gave
6
7
the dibromide which was not isolated. Vacuum distillative
cyclization of the dibromide under acidic conditions afforded
b-bromo sultone 5 in 45% isolated yield from 4. Treatment with
amine base in benzene solution at room temperature afforded 1†
as a white crystalline solid in good yield.
2 2
CH Cl ; 20; 168
Toluene; 120 ; 4
8
4
:
:
16 (quant)
27 (96%)
a
73
endo only
(96%)
SO2
a
Toluene; 150 ; 18
The dienophilicity of unsaturated sultone 1 in the Diels–
Alder reaction was investigated. Sulfonate has been used as an
activating group for olefinic dienophiles, for example, vinyl
sulfonates were found to be reactive dienophiles in both
O
MeO
OMe
OMe
OMe
endo only
(72%)
6
7
intermolecular and intramolecular Diels–Alder reactions. The
results of Diels–Alder reactions of 1 towards a series of simple
dienes are summarized in Table 1.‡ For a reactive diene such as
Xylene; reflux; 20
Cl4
SO2
Cl4
O
H
O2
S
O
Me
Me
Me
Me
O
O
O
O
a
(96%)
S
S
Toluene; 150 ; 18
SO2
250 °C, benzene
O
H2O2
reflux
H
H
or 450 °C, gas phase
8
2
3
1
O2
S
Me
Scheme 1
a
O
(84%)b
(75%)b
Toluene; 140 ; 13
Me
Br
H
H
Na2SO3
Br2
H O
Br
SO3Na
Br
3
SO Na
O2
S
O
78%
2
4
Me
Me
HCl, heat
Xylene; reflux; 20
4
5%
H
H
O
O
O
O
O
S
3
Et N
S
Me
Me
O
O2
S
98%
a
But
O
(89%)b
Benzene; 110 ; 18
Br
5
1
But
mp 81–83 °C
H
a
Sealed tube. b Mixture of regioisomers.
Scheme 2
Chem. Commun., 1997
611

 Lee, Albert W. M.
Lee, Albert W. M.
 Chan
Chan
 Jiang
Jiang
 Poon
Poon