
Tetrahedron Letters p. 6531 - 6534 (2010)
Update date:2022-08-17
Topics:
 Collinet-Fressancourt, Marion
Collinet-Fressancourt, Marion
 Azaroual, Nathalie
Azaroual, Nathalie
 Aubry, Jean-Marie
Aubry, Jean-Marie
 Nardello-Rataj, Véronique
Nardello-Rataj, Véronique
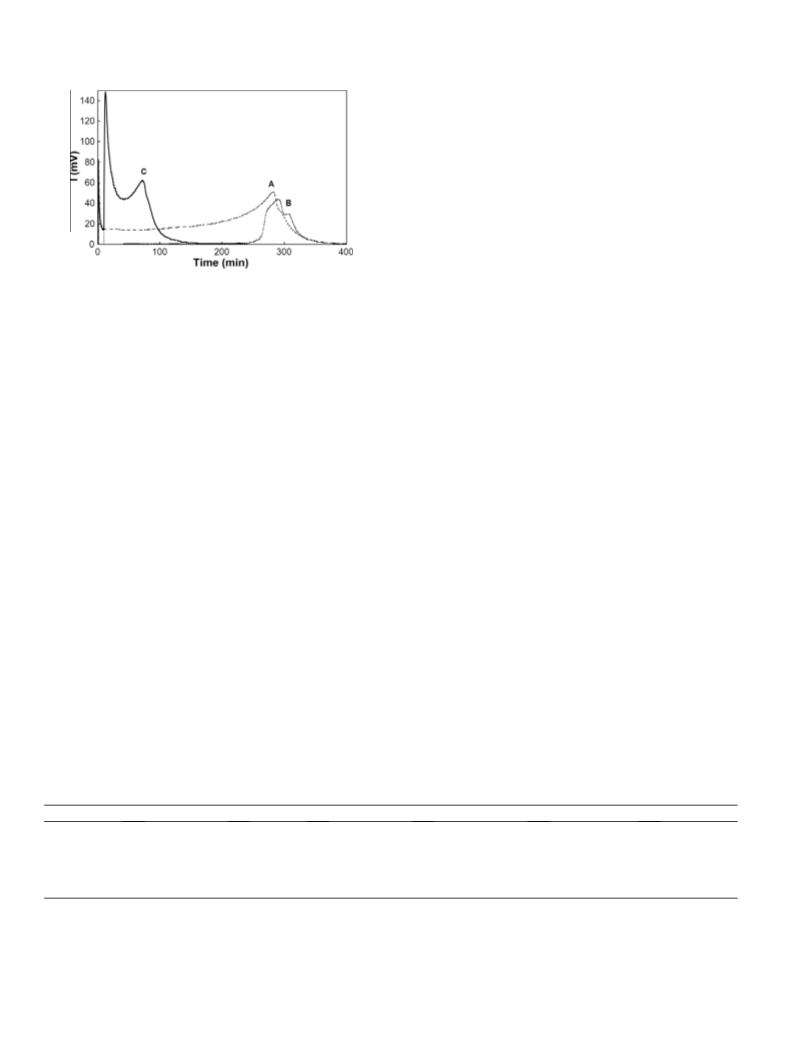
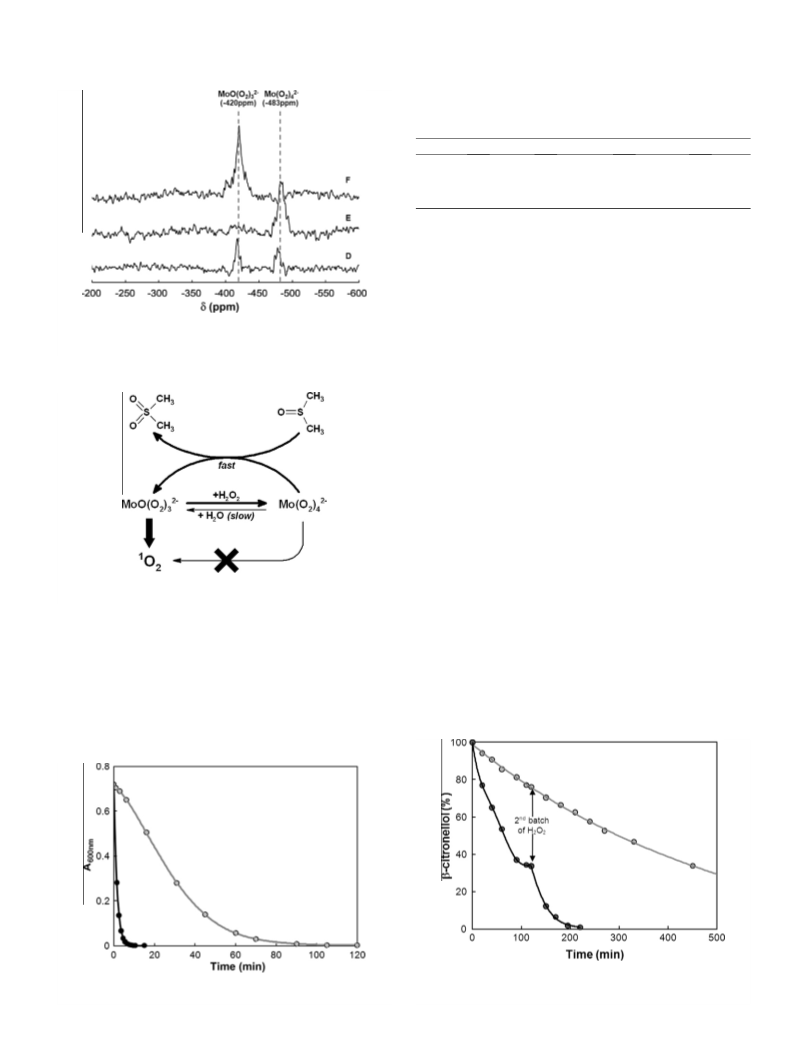
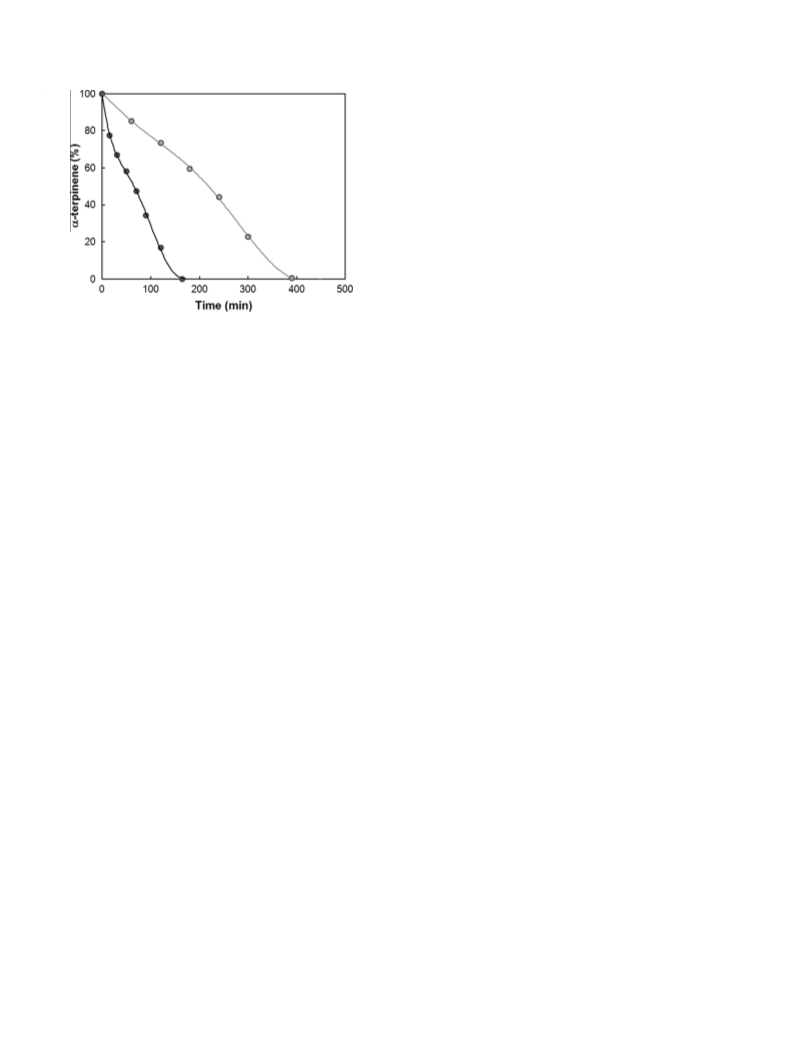
The kinetic booster effect of dimethylsulfoxide on the chemical generation of singlet oxygen, 1O2, from the disproportionation of hydrogen peroxide catalyzed by molybdate ions in methanol has been evidenced by detection of the IR luminescence of 1O2 at 1270 nm and by 95Mo NMR spectroscopy. DMSO interacts rapidly, through a direct oxygen transfer with the stable tetraperoxomolybdate Mo(O2) 42-, leading to DMSO2 and to the unstable triperoxomolybdate Mo(02)32-, which releases 1O2. The procedure was applied to accelerate the dark singlet oxygenation of β-citronellol and α-terpinene.
View More
Xinjiang Fufeng Biotechnologies Co., Ltd.
Contact:+86-539-7287111
Address:GANQUANPU INDUSTRIAL PARK, ECONOMIC AND TECHNOLOGICAL DEVELOPMENT AREA (TOUTUNHE DISTRICT) OF URUMQI
Contact:+33-5-34012600
Address:28 ZA des Pignès
Shanghai Synmedia Chemical Co., Ltd
Contact:+86-21-38681880
Address:6th Floor, 11A Building, No.528 Ruiqing Road, Heqing town, Pudong new district, Shanghai China
Contact:+852-8198 2399
Address:9E, Leapont Industrial Building, 18-28 Wo Liu Hang Road, Shatin, New Territories, Hong Kong
ZHEJIANG JIANYE CHEMICAL CO.,LTD.
Contact:86-571-64149273,64149234
Address:No. 48, Fuxi Road, Meicheng Town
Doi:10.1021/ac980266c
(1998)Doi:10.1080/00958972.2016.1268257
(2017)Doi:10.1002/ejic.201500395
(2015)Doi:10.1039/ft9928800091
(1992)Doi:10.1246/bcsj.60.1789
(1987)Doi:10.1134/S1070427209080047
(2009)