
Molecules (2021)
Update date:2022-08-10
Topics:
 Kim, Ga Yeong
Kim, Ga Yeong
 Song, Chae Won
Song, Chae Won
 Yang, Yo-Sep
Yang, Yo-Sep
 Lee, Na-Rae
Lee, Na-Rae
 Yoo, Hyung-Seok
Yoo, Hyung-Seok
 Son, Seung Hwan
Son, Seung Hwan
 Lee, Soo Jin
Lee, Soo Jin
 Park, Jong Seon
Park, Jong Seon
 Lee, Jong Kil
Lee, Jong Kil
 Inn, Kyung-Soo
Inn, Kyung-Soo
 Kim, Nam-Jung
Kim, Nam-Jung
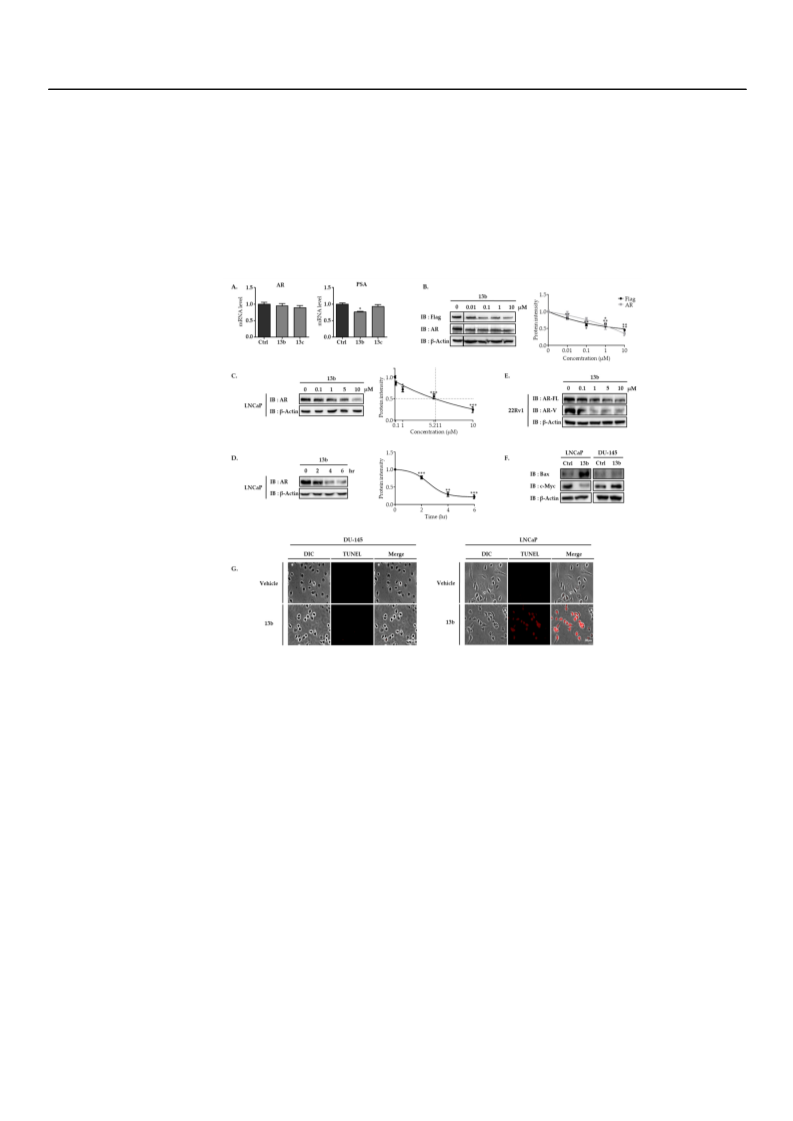
A series of PROTACs (PROteolysis-TArgeting Chimeras) consisting of bicalutamide analogs and thalidomides were designed, synthesized, and biologically evaluated as novel androgen receptor (AR) degraders. In particular, we found that PROTAC compound 13b could successfully demonstrate a targeted degradation of AR in AR-positive cancer cells and might be a useful chemical probe for the investigation of AR-dependent cancer cells, as well as a potential therapeutic candidate for prostate cancers.
View More















Beijing Apis Biotechnology Co., Ltd.
Contact:86-010-67856775-8551
Address:NO.4PUHUANGYU ROAD,FENTAI DISTRICT, BEIJING, CHINA
Contact:+86-512-69561895
Address:No.111, Building A4, 218 Xinghu Street, Suzhou Industrial Park, P. R. China
Tianjin Ingenochem Technology Co.,Ltd
Contact:+86-22-23677060
Address:Hitech Green Industry Park K2-9-602, Nankai district
Nanjing Zelang Medical Technology Co. Ltd
Contact:86-25-83063290/13770714480
Address:Ganjiabian 108# 01 Unit,701-702 room,Yao Hua Street,Qixia District,Nanjing,Jiangsu,China
Shanghai Dynamic Industrial Co.,Ltd.
website:http://www.shdynamic.com
Contact:86-021 3392 6680
Address:Room 805 Information Tower, No.1403 Minsheng Road, Pudong New Area, Shanghai 200135, P. R. China
Doi:10.1021/acsmedchemlett.6b00005
(2016)Doi:10.1039/f19827801937
(1982)Doi:10.1016/S0008-6215(00)85143-5
(1980)Doi:10.1021/ja00453a068
(1977)Doi:10.1246/cl.2000.694
(2000)Doi:10.1016/j.materresbull.2010.06.058
(2010)