E
Synlett
P. Massé et al.
Cluster
2
7 and 4, deprotection, and oxidation proceeded smoothly
grateful to the French Ministry of Research and Development for fi-
nancial support.()
to procure 29. Unfortunately, RCM attempts on 28 gave us
notably a 28-membered macrocycle 30 among a myriad of
isomerized products (adding benzoquinone to prevent
isomerization did not succeed). Compound 29 with the
ketone gave us also a head-to-tail dimer 31 in 37% yield
Acknowledgment
28
We are really grateful to Marc Mauduit who provided us his catalyst.
(Scheme 8).
Supporting Information
OH
OBn
MeO
MeO
MeO
Supporting information for this article is available online at
https://doi.org/10.1055/s-0039-1691523.
c, b
i, d, e
11
S
u
p
p
orit
n
g Inform ati
o
n
S
u
p
p
orit
n
g Inform ati
o
n
MeO
I
BF3K
26
27
References and Notes
OBn
OBn
(1) Begley, M. J.; Campbell, R. V.; Crombie, L.; Whiting, D. A. J. Chem.
MeO
MeO
Soc. 1971, 3634.
f, g
MeO
BnO
h
MeO
BnO
(2) (a) Jahng, Y.; Park, J. G. Molecules 2018, 23, 3107. (b) Alberti, Á.;
Riethmüller, E.; Béni, S. J. Pharm. Biomed. Anal. 2018, 147, 13.
(c) Zhu, J.; Islas-Gonzalez, G.; Bois-Choussy, M. Org. Prep.
Proced. Int. 2000, 32, 505. (d) Sun, C.; Huang, H.; Xu, C.; Li, X.;
Chen, K. Plant Foods Hum. Nutr. 2013, 68, 97.
3) (a) Gulder, T.; Baran, P. S. Nat. Prod. Rep. 2012, 29, 899. (b) Joshi,
B. S.; Pelletier, S. W.; Newton, M. G.; Lee, D.; McGaughey, G. B.;
Puar, M. S. J. Nat. Prod. 1996, 59, 759.
4) (a) Tao, J.; Morikawa, T.; Toguchida, I.; Ando, S.; Matsuda, H.;
Yoshikawa, M. Bioorg. Med. Chem. 2002, 10, 4005. (b) Wang, M.;
Liu, Y.; Pan, R. L.; Wang, R. Y.; Ding, S. L.; Dong, W. R.; Sun, G. B.;
Ye, J. X.; Sun, X. B. Int. J. Mol. Med. 2019, 43, 2133. (c) Kubo, M.;
Yamazaki, M.; Matsuda, H.; Matsuo, K.; Asanuma, Y. Biol.
Pharm. Bull. 2002, 24, 259.
5) (a) Shen, S.; Liao, Q.; Feng, Y.; Liu, J.; Pan, R.; Lee, S. M. Y.; Lin, L.
Food Chem. 2019, 270, 305. (b) Shen, S.; Liao, Q.; Liu, J.; Pan, R.;
Lee, S. M. Y.; Lin, L. J. Cachexia Sarcopenia Muscle 2019, 10, 429.
X
X
2
8 X = H, OH
29 X = O
(
r
j
1
1%
37%
OBn
(
OMe
OMe
OBn
X
BnO
MeO
X
(
MeO
OBn
0 X = H, OH
(c) Cao, Y.; Chen, X.; Deng, S.; Li, Z.; Xu, T.; Yu, G.; Yu, Y.; Yuan,
3
31 X = O
G.; Zhang, M. CN 108434127A, 2018. (d) Shen, S.; Liao, Q.; Pan,
R.; Lin, L. Br. J. Pharmacol 2019, 176, 3983.
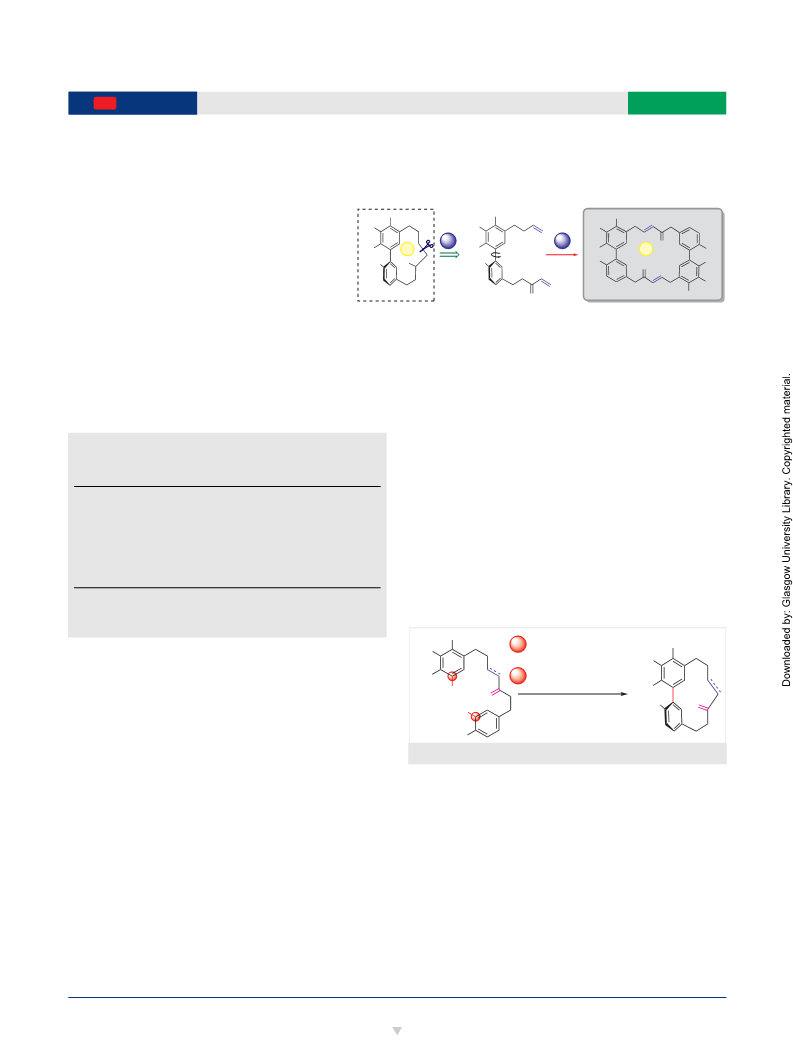
Scheme 8 Synthesis of corresponding 14-membered macrocycle pre-
cursor and subsequent RCM attempts. Reagents and conditions: (a)
butenylbromide, Mg, THF, 0 °C, 0.5 h, 98%; (b) i. TES, TFAA, CH Cl ,
(6) Dai, G. H.; Meng, G. M.; Tong, Y. L.; Chen, X.; Ren, Z. M.; Wang,
K.; Yang, F. Phytomedicine 2014, 21, 1490.
2
2
0
4
°C, 1.5 h; ii. TBAF, THF, 80%; (c) BnBr, NaH, NaI, DMF, 1.5 h, 0 °C to r.t.,
7%; (d) i. t-BuLi, hexane, –78 °C; ii. i-PrOBpin, –78 °C to r.t., 2.5 h; (e)
(7) Martin, M. D.; Calcul, L.; Smith, C.; Jinwal, U. K.; Fontaine, S. N.;
Darling, A.; Seeley, K.; Wojtas, L.; Narayan, M.; Gestwicki, J. E.;
Smith, G. R.; Reitz, A. B.; Baker, B. J.; Dickey, C. A. ACS Chem. Biol.
2015, 10, 1099.
KHF , H O, MeOH, r.t., 1.5 h, 93% (over 2 steps); (f) 4, Pd(PPh ) , K CO ,
TBAB, H O, 100 °C, 16 h; (g) PPTS, MeOH, CH Cl , 60 °C, 3 h, 35% (over
2
2
3
4
2
3
2
2
2
2
1
steps); (h) MnO , CH Cl , 40 °C, 3 h, 37%. (i) GII, CH Cl (1 mM), 16 h,
2
2
2
2
2
(8) Mohamed, S. E. N.; Whiting, D. A. J. Chem. Soc., Perkin Trans. 1
1% (j) GII, PhMe (1 mM), 16 h, 37%.
1983, 2577.
(
9) Bochicchio, A.; Schiavo, L.; Chiummiento, L.; Lupattelli, P.;
Funicello, M.; Hanquet, G.; Choppin, S.; Colobert, F. Org. Biomol.
Chem. 2018, 16, 8859.
In conclusion, a 26-membered macrocycle and two 28-
membered macrocycles have been successfully synthetized
using RCM in high-dilution conditions. No traces of 13- or
(
10) (a) Meidlinger, D.; Marx, L.; Bordeianu, C.; Choppin, S.; Colobert,
F.; Speicher, A. Angew. Chem., Int. Ed. 2018, 57, 9160.
(b) Norouzi-Arasi, H.; Salom-Roig, X. J.; Lanners, S.; Hanquet, G.
1
4-membered cyclized products were spotted even with
Curr. Org. Synth. 2017, 15, 105. (c) Bensoussan, C.; Rival, N.;
Hanquet, G.; Colobert, F.; Reymond, S.; Cossy, J. Nat. Prod. Rep.
RRCM. We can infer that the strain outpaces the reactivity,
even in our attempts to prepare the 14-membered macro-
cycle.
2
014, 31, 468. (d) Chiummiento, L.; Funicello, M.; Lopardo, M.
T.; Lupattelli, P.; Choppin, S.; Colobert, F. Eur. J. Org. Chem. 2012,
88. (e) Ferreiro-Mederos, L.; Lanners, S.; Henchiri, H.; Fekih, A.;
1
Hanquet, G. Nat. Prod. Res. 2009, 23, 256. (f) Bonini, C.;
Chiummiento, L.; Pullez, M.; Solladié, G.; Colobert, F. J. Org.
Chem. 2004, 69, 5015.
Funding Information
We thank the Centre National de la Recherche Scientifique (CNRS),
France as well as the University of Strasbourg, and Paul Massé is
©
2019. Thieme. All rights reserved. Synlett 2019, 30, A–F

 Chiummiento, Lucia
Chiummiento, Lucia
 Choppin, Sabine
Choppin, Sabine
 Colobert, Fran?oise
Colobert, Fran?oise
 Hanquet, Gilles
Hanquet, Gilles
 Massé, Paul
Massé, Paul