Mengxiao Zhang et al. / Chinese Journal of Catalysis 35 (2014) 1874–1882
1875
ids [5,8–10]. However, the stability of the resultant materials in
the above cases is not satisfactory, especially in polar media,
because of the leaching of polycyclic aromatic hydrocar‐
bon‐containing –SO3H groups [8,11]. In addition, a large
amount of concentrated sulfuric acid is used in their prepara‐
tion process, which is harmful to the environment. Further‐
more, catalyst swelling is another important parameter that
can remarkably affect the catalytic activity, indicating that sul‐
fopolycyclic aromatic compounds are not suitable catalysts in
non‐media reactions (e.g., gas‐phase reactions) [8].
ture. The specific surface areas of the samples were calculated
from the adsorption isotherms by the Brunauer‐Emmett‐Teller
(BET) method. FT‐IR spectra of the samples were recorded on a
Nicolet Avatar‐360 spectrometer. TGA was performed using a
PerkinElmer TGA7 instrument under a flowing N2 atmosphere
at a heating rate of 10 °C/min. SEM studies were carried out
with a Philips XL30 using an accelerating voltage of 15 kV. TEM
images were taken using a JEOL JEM 2100 instrument. The
sulfur content was measured by elemental analysis on a
Bruker‐AXS (S4 EXPLORER) elemental analyzer.
Carbon nanotubes (CNTs) have attracted a lot of interest
since their discovery in 1991 [12] because of their unique
structural, mechanical, and electronic properties [13]. Because
CNTs themselves have little catalytic activity, acidic functional
groups need to be introduced, specifically onto the surface of
CNTs, for their application to acid‐catalyzed reactions.
CNT‐based acid catalysts have been synthesized by directly
sulfonating CNTs [14–17], but the activity or the stability of the
obtained catalysts was not ideal [14,17]. Sulfonated multi‐
walled carbon nanotubes have also been obtained by direct
synthesis, resulting in higher stability [18]. CNT‐based acid
catalysts can also be synthesized by directly functionalizing
CNTs with polymers. Poly(styrene sulfonic acid)‐functionalized
CNTs (PSA‐CNTs) have been synthesized by in situ radical
polymerization [19]. High activity and good stability can be
achieved over this catalyst although the stability needs to be
further improved [20].
In this work, perfluorosulfonic acid‐functionalized CNT
(PSFA‐CNT) samples were prepared. Their textural, structural,
and acidic properties were characterized by N2 adsorption,
scanning electron microscopy (SEM), transmission electron
microscopy (TEM), Fourier transform infrared spectroscopy
(FTIR), thermogravimetric analysis (TGA), and titration meth‐
ods. The catalytic activity and stability of the samples for alkyl‐
ation of hydroquinone with tert‐butanol were tested and com‐
pared with PSA‐CNTs.
2.3. Acidity measurement
The density of surface acid sites was measured by a neu‐
tralization titration method [21]. In brief, the sample was added
into an aqueous solution of NaCl (in excess), and HCl formed
because of the exchange of Na+ for protons on sulfonic groups,
which were titrated with a standard solution of NaOH. The
acidity was also measured by potentiometric titration [22,23].
The solid was suspended in acetonitrile, agitated for 3 h, and
then titrated with 0.1 butylamine (mol/L) in acetonitrile. The
electrode potential variation was recorded with a METTLER
TOLEDO FE20 potentiometer.
2.4. Catalytic testing
Alkylation of hydroquinone was carried out in a stainless
steel autoclave with a PTFE liner using magnetic stirring. Typi‐
cally, 0.5 g hydroquinone, 1.0 g tert‐butanol, and 0.2 g catalyst
were added in the autoclave accompanied with 2 g xylene as
solvent. The reaction lasted 4 h. The products were analyzed
with a GC122 gas chromatograph equipped with a SE‐54 capil‐
lary column (30 m 0.25 mm 0.3 m) and a flame ionization
detector.
3. Results and discussion
2. Experimental
3.1. Structure of the catalysts
2.1. Catalyst preparation
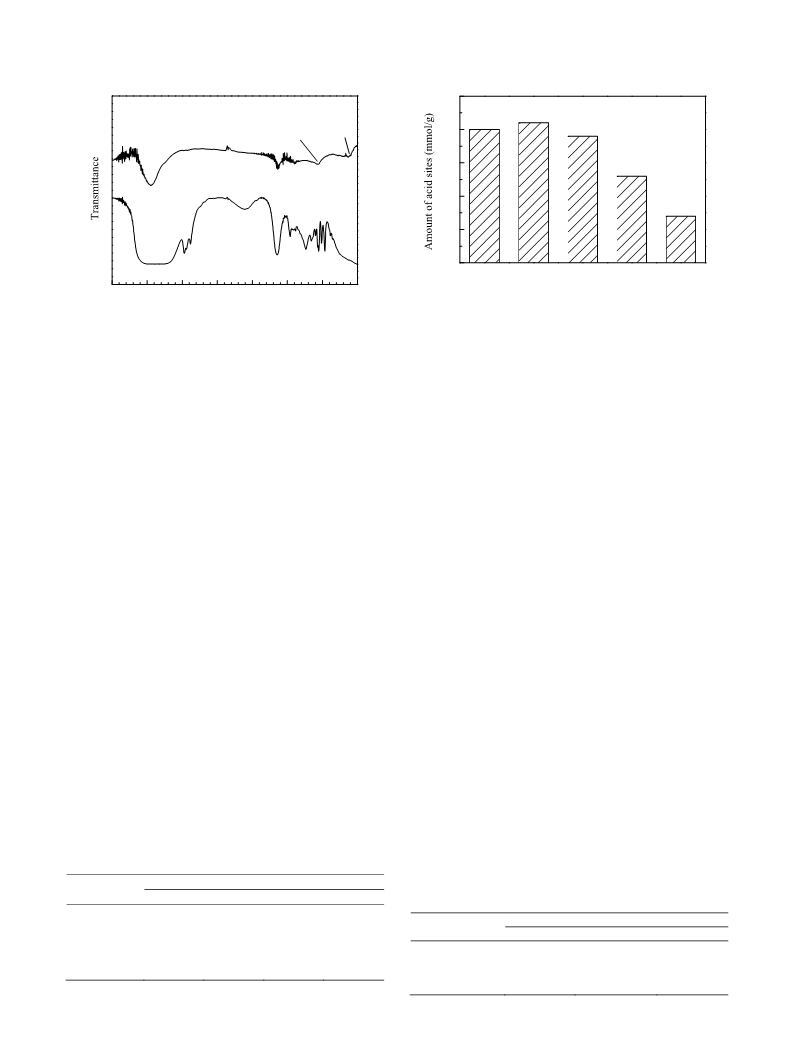
PFSA‐CNT samples with different perfluorosulfonic acid
loadings were prepared. Their N2 adsorption‐desorption iso‐
therms were recorded and are shown in Fig. 1. Similar iso‐
therms were observed for all the samples, showing that modi‐
fication by perfluorosulfonic acid has little effect on the pore
structure of the CNTs.
Table 1 summarizes the textural properties of the prepared
samples. The samples maintained a large surface area and pore
volume after modification although slightly lower than that of
the pristine CNTs.
The SEM images of the CNT materials before and after mod‐
ification are shown in Fig. 2. Significant differences can be ob‐
served in the morphology of the CNTs before and after modifi‐
cation. The PFSA‐CNTs are arranged in ordered sheets, while
the pristine CNTs are disorganized and entwined without any
order. A similar change was observed for PSA‐CNTs [20].
The size and structure of the functionalized CNT materials
can be determined by TEM (Fig. 3). It is clear from the TEM
PFSA‐CNT samples were synthesized by liquid deposition.
Typically, 500 mg of multi‐walled carbon nanotubes (purity
>99.5%, Chengdu Organic Chemical Co. Ltd.) was dispersed in a
certain amount of PFSA‐polytetrafluoroethylene (PTFE) co‐
polymer solution (5 wt% solution, Alfa Aesar), and the mixture
was then diluted with ethanol solution (75%) until the total
mass of the mixture was 25 g. After sonication for 40 min, the
solution was stirring overnight at room temperature. The ob‐
tained black solid was then dried at 100 °C. The final product is
designated as PFSA‐CNT‐x, where x represents the mass ratio
of the PFSA‐PTFE copolymer to CNT.
2.2. Catalyst characterization
The N2 adsorption‐desorption isotherms were measured on
a Micromeritics ASAP 2000 instrument at liquid N2 tempera‐

 Zhang, Mengxiao
Zhang, Mengxiao
 Li, Cuican
Li, Cuican
 Hua, Weiming
Hua, Weiming
 Yue, Yinghong
Yue, Yinghong
 Gao, Zi
Gao, Zi