Miyazawa et al.
45
porcine pancreatic lipase or Candida cylindracea (rugosa) lipase in
organic solvents, it was found that deacylation took place pre-
dominantly at positions other than ortho to the ketonic group,
generally the acetoxy group at the para position being preferen-
tially cleaved over the one at the meta position. The authors con-
cluded that the carbonyl group attached to the benzene ring plays
an important role in the recognition of acetoxy groups in the
polyphenolic peracetates, and they postulated the formation of a
transient Schiff’s base-type complex with the lysine residue in the
active site of porcine pancreatic lipase.4d They have also reported
on the lipase-catalyzed chemo- and regioselective deacylation of
peracetylated aromatic acid esters and ketones4d and perace-
tylated enolic forms of polyphenolic benzyl phenyl ketones.5 In
connection with these studies, Nicolosi and co-workers have re-
ported on the regioselectivity observed in the Pseudomonas cepacia
lipase catalyzed deacylation with 1-butanol of peracetylated fla-
vonoids that contain a carbonyl group attached to the benzene
ring.6 It is worth while to examine how substituents other than
the carbonyl can affect the regioselectivity in the lipase-catalyzed
deacylation of peracylated polyphenols. In this regard, Klibanov
and co-workers have investigated the lipase-catalyzed deacylation
with 1-butanol of octylhydroquinone butanoylated at both phe-
nolic hydroxy groups, and they even found the reversion of the
regioselectivity of Pseudomonas cepacia lipase upon a change from
toluene to acetonitrile as the reaction medium.7
Preparation of the di-O-propanoyl derivatives of substituted
hydroquinones and resorcinols
The 1,4-di-O-propanoates (1) of substituted hydroquinones and
1,3-di-O-propanates (5) of 4-substituted resorcinols were prepared
by the reaction of each hydroquinone or resorcinol with pro-
panoyl chloride in pyridine. The synthetic procedure and the NMR
data for all of the dipropanoyl compounds except 5c were re-
ported before.11
1,3-Di-O-propanoyl-4-t-butylresorcinol (5c)
Oil. 1H NMR ␦: 1.12 (3H, t, J = 7.5 Hz, CH2CH3), 1.16 (3H, t, J = 7.5 Hz,
CH2CH3), 1.29 (9H, s, C(CH3)3), 2.58 (2H, q, J = 7.5 Hz, CH2), 2.64 (2H,
q, J = 7.5 Hz, CH2), 6.90 (1H, d, J = 2.5 Hz, H-2), 6.96 (1H, dd, J = 8.5
and 2.5 Hz, H-6), 7.39 (1H, d, J = 8.5 Hz, H-5). Anal. calcd. for
C16H22O4: C 69.04, H, 7.97; found: C 68.85, H 8.05.
General procedure for the CAL-B-catalyzed deacylation of
the di-O-propanoyl derivatives of substituted
hydroquinones and resorcinols in an analytical scale
A solution of a substituted 1,4-di-O-propanoylhydroquinone (1)
or a 4-substituted 1,3-di-O-propanoylresorcinol (5) (0.1 mmol) and
2-propanol (24 L, 0.3 mmol) in anhydrous diisopropyl ether
(240 L) was stirred with CAL-B (40 mg) at 45 °C in a thermostated
incubator. After a certain period of time, the reaction mixture
was filtered through a glass filter and evaporated to dryness
under reduced pressure. The residue was subjected to NMR
analysis to determine the conversion and product distribution,
These studies have prompted us to investigate the lipase-
catalyzed deacylation of diacylated dihydroxybenzenes, i.e., hy-
droquinones and resorcinols, carrying several substituents other
than the carbonyl. We found that in these deacylations, Candida
antarctica lipase B (CAL-B)8 was more active than other lipases so
far employed and moreover highly regioselective and that some
secondary alcohols acted as better acyl acceptors than the corre-
sponding primary alcohols.9 The present paper reports the results
of our investigation in detail.
1
as the H NMR spectra of the starting dipropanoate, both iso-
meric monopropanoates, and the parent diol are available with
each hydroquinone or resorcinol. In the reactions of substituted
1,4-di-O-propanoylhydroquinones (1a–1h), the proton signals used
for the purpose were different from compound to compound. The
ArCH3 proton signals together with the COCH2CH3 signals were
utilized with 1a. The proton signals appearing as a singlet were
employed in the following cases: 1d, C(CH3)3; 1e, OCH3. The pro-
ton signals (H-5, H-3, or H-6) in the aromatic region were used
with 1b, 1c, and 1f-h. In the reactions of 4-substituted 1,3-di-O-
propanoylresorcinols (5a–5g), the proton signals (H-5, H-2, or H-6)
in the aromatic region were mainly employed for the purpose.
The whole content of the reaction mixture was used up for one
analysis, and several discrete reaction mixtures were used at dif-
ferent reaction times.
Materials and methods
All of the hydroquinones and resorcinols used in this study,
except ethyl-, isopropyl-, and fluorohydroquinoes10 and 4-t-
butylresorcinol, were purchased from Tokyo Chemical Industry
Co. or Aldrich. All of the alcohols were commercially available and
employed after drying over molecular sieves prior to use. CAL-B
was supplied by Boehringer Mannheim (BioCatalytics) as an im-
mobilized form (chirazyme L-2), which had a specific activity of
3.2 U mg–1 lyophilized powder with tributyrin at 25 °C. All organic
solvents were distilled following standard protocols and dried
over molecular sieves prior to use.
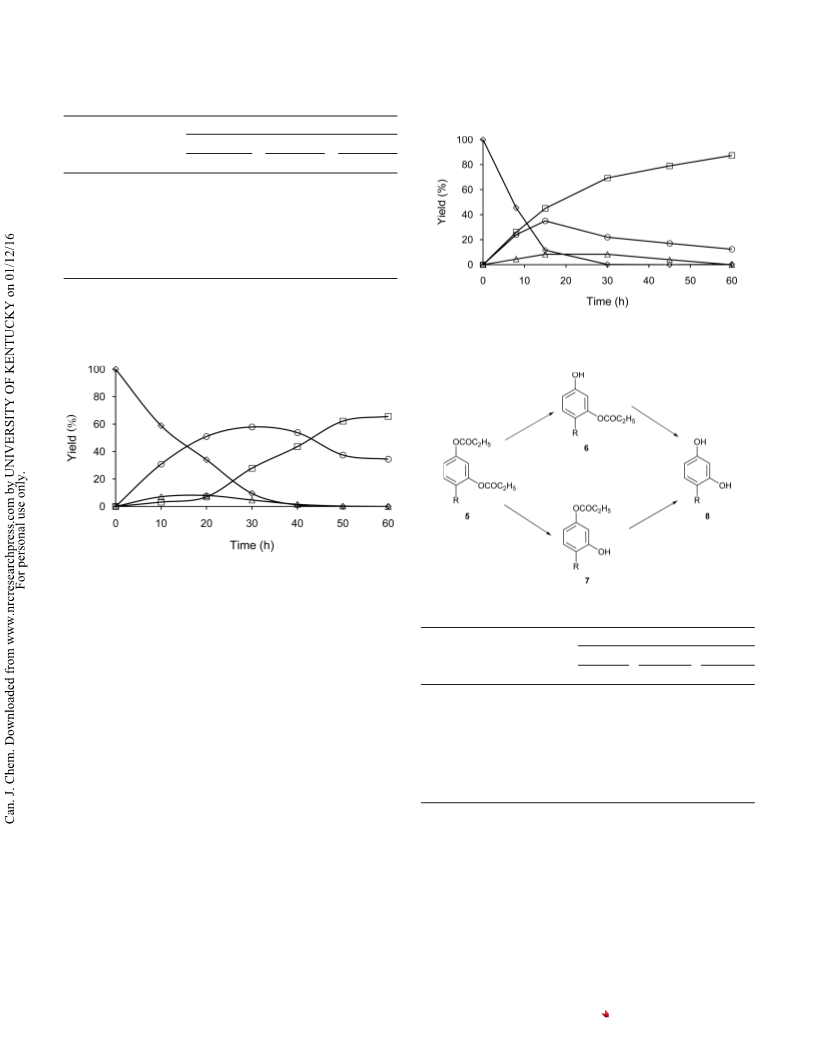
HPLC monitoring of the deacylation of
1,4-di-O-propanoylmethoxyhydroquinone (1e)
A solution of 1e (45 mg, 0.18 mmol) and an alcohol (0.54 mmol)
in diisopropyl ether (450 L) was stirred with CAL-B (75 mg) at
45 °C in a 1-mL vial. Aliquots (approximately 10 L) of the reaction
mixture were withdrawn at frequent intervals, diluted with
diethyl ether, and filtered through a PTFE membrane filter. After
evaporation of ether, the residue was dissolved in acetonitrile
(1 mL) and subjected to HPLC analysis under the following condi-
tions: column, Ascentis RP-Amide (4.6 mm i.d. × 250 mm); mobile
phase, 34% aqueous acetonitrile containing H3PO4 (0.01 M); flow
rate, 1.0 mL min−1; column temperature, 30 °C; detection, UV at
280 nm. The details of the analytical procedure are available else-
where.11
1H NMR (500 MHz) and 13C NMR (125 MHz) spectra were recorded
on a Varian Unity 500 spectrometer using DMSO-d6 as a solvent
with TMS as an internal standard. The liquid chromatograph em-
ployed was a Shimadzu LC-10AD instrument, equipped with a
Rheodyne 7125 sample injector and a Shimadzu SPD-10A variable-
wavelength UV monitor. A Shimadzu C-R8A data processor was
used for data acquisition and processing. TLC was run on Merck
precoated silica gel plates and Wakogel C-300 was used for col-
umn chromatography.
The NMR data for all of the monopropanoates of substituted
hydroquinones and resorcinols used in this study except the 3-O-
propanoate (6c) of 4-t-butylresorcinol are available elsewhere.11
Preparation of mono-O-propanoyl derivatives through
CAL-B-catalyzed gram-scale deacylations
3-O-propanoyl-4-t-butylresorcinol (6c)
Preparation of 4-t-butylresorcinol (8c)
1,3-Di-O-propanoyl-4-t-butylresorcinol (5c) (1.11 g, 4.0 mmol) was
dissolved in anhydrous diisopropyl ether (10 mL) followed by the
addition of 2-propanol (1.0 mL, 132 mmol) and then CAL-B (1.6 g).
The reaction mixture was stirred at 45 °C in an incubator. The
reaction was stopped after 90 min by filtrating the enzyme pow-
der, which was washed with diethyl ether. Evaporation of the
solvent in vacuo from the combined filtrate and the washing af-
This substituted resorcinol was prepared through the reaction
of resorcinol with t-butanol in the presence of concentrated sul-
furic acid in glacial acetic acid according to the literature method.12
Oil. 1H NMR ␦: 1.28 (9H, s, C(CH3)3), 6.10 (1H, dd, J = 8.5 and 2.5 Hz,
H-6), 6.24 (1H, d, J = 2.5 Hz, H-2), 6.86 (1H, d, J = 8.5 Hz, H-5), 8.90 (1H,
s, OH), 9.07 (1H, s, OH).
Published by NRC Research Press

 Miyazawa, Toshifumi
Miyazawa, Toshifumi
 Hamada, Manabu
Hamada, Manabu
 Morimoto, Ryohei
Morimoto, Ryohei