Full Paper
overall yield from 2-amino-5-methoxybenzaldehye, 9 steps)
with simple and inexpensive starting materials and reagents.
Three cascade reactions and one-pot treatments were de-
signed, developed, and successfully applied to this synthesis,
including construction of the E-ring with cascade allylic alcohol
oxidation and intermolecular oxa Diels–Alder reaction under
mild conditions, preparation of the AB-ring system with mild
Compound of 12b. The same procedure for 12a was used for
preparation of 12b (crude yield 100%). Mp: 138–1408C. H NMR
1
(
(
500 MHz, CD OD): d=3.89 (3H, s), 4.84 (2H, s), 4.86 (2H, s), 7.19
3
1H, d, J=2.0 Hz), 7.31 (1H, dd, J =9.5 Hz, J =2.5 Hz), 7.88(1H, d,
1
2
13
J=9.5 Hz), 8.15 (1H, s) ppm. C NMR (125 MHz, CD OD): d=56.1,
3
61.8, 64.3, 106.4, 123.2, 130.2, 130.4, 134.5, 135.4, 143.1, 156.7,
159.6 ppm. IR (KBr): n˜ max =3353, 3130, 3075, 3001, 2971, 2926,
2875, 2835, 1623, 1605, 1500, 1468, 1431, 1385, 1353, 1303, 1226,
cascade pyrrolidine/PhCO H-catalyzed aza Michael addition,
1185, 1169, 1136, 1107, 1052, 1028, 977, 959, 917, 854, 833, 790,
2
À1
+
and intramolecular aldol reaction and in situ MnO oxidation,
759, 628 cm . HRMS (ESI, m/z) calcd. for C12
20.0969; found: 220.0995.
Compound 4a. To a stirred solution of crude 12a (2.0 g) in DMF
H13NO (M+H) :
3
2
2
and closure of the D-ring with cascade TMSCl-promoted con-
densation, olefin migration, and ethanol elimination. The re-
proted protecting group-free total synthesis, with advantages
of a short route, simple operations, and mild conditions, is be-
lieved to be helpful for the future development of industry-
scale syntheses of camptothecin-family alkaloids.
(
33 mL) and CH CN (200 mL) was successively added Et N (5.9 mL,
3
3
4
2 mmol) and MsCl (2.5 mL, 32 mmol) at 08C under N2 atmos-
phere. After being stirred for 5 min, excess concentrated NH OH
4
solution (35%, 300 mL) was added. The reaction was allowed to
warm to room temperature and stirred for additional 1 h. The reac-
tion mixture was extracted with CHCl . The combined organic
3
layers were dried over anhydrous MgSO , filtered, and concentrat-
ed. The residue was purified by flash chromatography on silica gel
4
Experimental Section
(
CH Cl /CH OH=4:1) to afford 4a (1.03 g, 52%, from 6a) as a pale
2 2 3
General. Unless stated otherwise, all solvents were purified and
dried prior to use. IR spectra were recorded on a Bruker TENSOR
[18] 1
yellow solid.
H NMR (300 MHz, CDCl ): d=3.03 (1H, brs), 4.44
3
(2H, s), 4.49 (2H, s), 7.54 (1H, t, J=7.5 Hz), 7.70 (1H, t, J=7.5 Hz),
2
7 or VECTOR 22 FT-IR instrument. Reference peaks for chloroform
1
13
7.81 (1H, d, J=8.1 Hz), 7.98 (1H, s), 8.06 (1H, d, J=8.7 Hz) ppm.
ESIMS (m/z) : 171.25 (M+H) .
in H NMR and C NMR spectra were set at 7.26 ppm and
7.16 ppm, respectively. For [D ]MeOH, the reference peaks in
+
7
4
1
13
H NMR and C NMR spectra were set at 3.31 ppm and 49.00 ppm,
respectively. Flash chromatography was performed on silica gel
Compound 4b. The same procedure for 4a was used for the prep-
aration of 4b. Flash chromatography (CH Cl /MeOH=4:1) afforded
2 2
(
300–400 mesh).
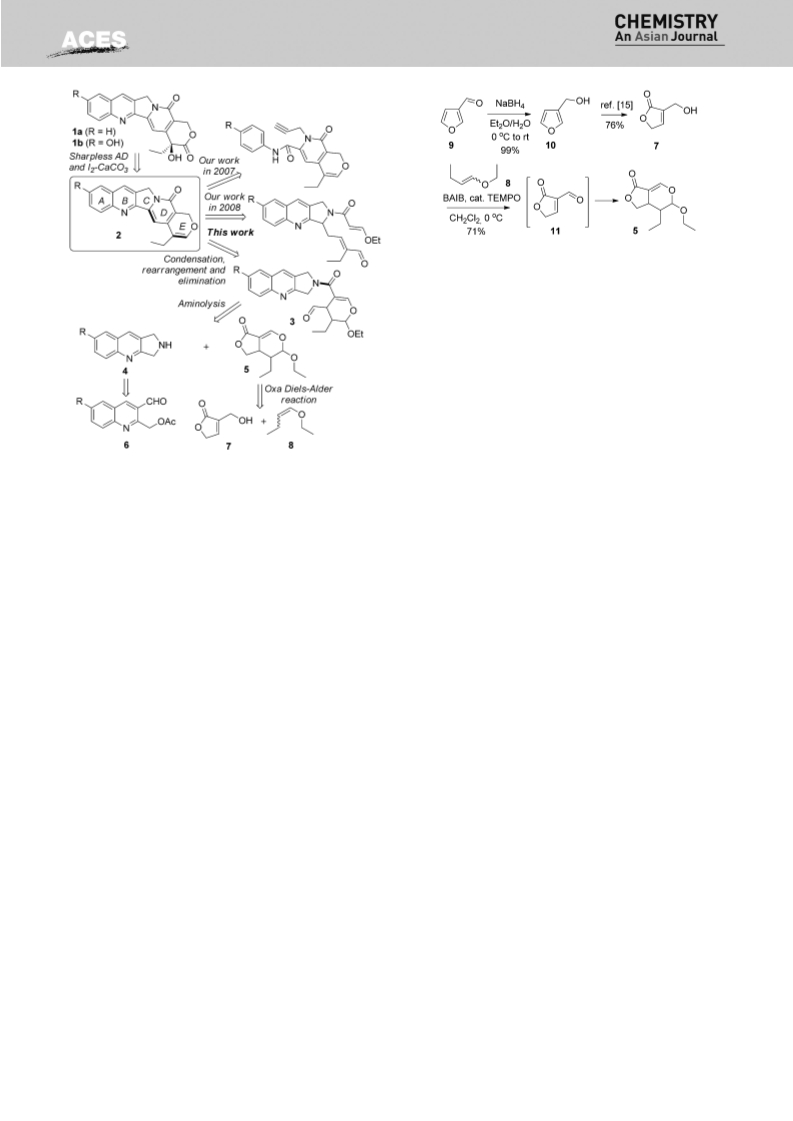
Compound 5. To a stirred solution of 3-(hydroxymethyl)furan-
(5H)-one (7) (8.9 g, 78 mmol) and vinylether 8 (38 g, 379 mmol) in
CH Cl (460 mL) was added bis(acetoxy)iodobenzene (BAIB, 33 g,
4b (52%, from 6b) as a pale yellow solid. Mp: 1138C (dec.).
1
H NMR (500 MHz, CD OD): d=3.92 (3H, s), 4.39 (2H, s), 4.51 (2H,
3
s), 4.88 (1H, s), 7.27 (1H, d, J=2.5 Hz), 7.36 (1H, dd, J =9.5 Hz, J =
1
2
2
1
3
2
.5 Hz), 7.85 (1H, d, J =9.5 Hz), 8.12 (1H, s) ppm. C NMR
1
2
2
(125 MHz, CDCl ): d=50.8, 52.2, 56.1, 107.1, 123.5, 130.0, 130.2,
3
1
02 mmol) and TEMPO (1.8 g, 11.5 mmol) successively at 08C. After
1
30.8, 132.0, 144.6, 159.5, 160.3 ppm. IR (KBr): n˜ max =3421, 3064,
being stirred at the same temperature for 24 h, the reaction mix-
ture was quenched by the addition of saturated aqueous Na S O
solution. The aqueous layer was extracted with ethyl acetate. The
3
003, 2959, 2925, 2851, 1622, 1585, 1502, 1453, 1367, 1299, 1224,
2
2
3
À1
1159, 1127, 1101, 1027, 826 cm . HRMS (EI, m/z) calcd. for
+
.
C H N O (M ): 200.0950; found: 200.0961.
12
12
2
combined organic layers were dried over anhydrous MgSO , fil-
4
tered, and concentrated. The residue was purified by flash chroma-
Compound 13a. To a solution of tricyclic amine 4a (510 mg,
3.0 mmol) in toluene (25 mL) was added Me Al (2.0m in toluene,
1.8 mL, 3.6 mmol) dropwise at 08C under N atmosphere. The reac-
tography on silica gel (petroleum ether/ethyl acetate=5:1) to
3
1
afford 5 (11.8 g, 71%) as a colorless oil. H NMR (300 MHz, CDCl ):
2
3
d=0.81–0.98 (3H, m), 1.16–1.28 (3H, m), 1.33–1.40 (1H, m), 1.50–
tion was then warmed to room temperature. After being stirred for
1 h, the reaction mixture was once again cooled down to 08C. A
solution of lactone 5 (635 mg, 3.0 mmol) in toluene (3 mL) was
then added dropwise. The reaction was allowed to warm to room
temperature and stirred for 16 h. The reaction was quenched suc-
1
.56 (1H, m), 1.71–1.80 (1H, m), 2.95–3.54 (1H, m), 3.60–3.65 (1H,
m), 3.80–4.08 (2H, m), 4.43–4.62 (1H, m), 4.82–5.10 (1H, m), 7.35–
1
3
7
1
6
1
2
8
2
.43 (1H, m) ppm. C NMR (75 MHz, CDCl ): d=10.8, 11.3, 11.9,
3
4.7, 14.8, 14.81, 17.1, 22.6, 22.8, 31.7, 33.8, 35.5, 37.8, 41.4, 41.6,
4.4, 64.6, 65.7, 67.5, 71.4, 71.6, 99.1, 99.2, 103.7, 103.8, 104.4,
47.9, 148.1, 149.6, 169.8 ppm. IR (KBr): n˜ max =3075, 2974, 2935,
898, 2879, 1759, 1670, 1459, 1379, 1341, 1236, 1150, 1105, 1019,
75, 836, 753 cm . HRMS (ESI, m/z) calcd. for C H O (M+H) :
13.1122; found: 213.1135.
cessively with 0.5m HCl and H
was extracted with ethyl acetate. The combined organic layers
were dried over anhydrous MgSO , filtered, and concentrated. The
residue was purified by flash chromatography on silica gel (CH Cl
CH OH=25:1) to afford 13a (1.02 g, 89%) as a pale yellow solid.
H NMR (300 MHz, CDCl ): d=0.91–0.98 (3H, m), 1.21–1.25 (3H, m),
O (30 mL) at 08C. The aqueous layer
2
4
À1
+
/
2
11
16
4
2
3
1
Compound 12a. To a stirred solution of 6a (5.7 g, 25 mmol) in
3
1
.33–1.63 (2H, m), 1.85–1.89 (1H, m), 2.52–2.90 (1H, m), 3.59–3.63
MeOH (110 mL) was added KBH (1.1 g, 20 mmol) in small portions
4
(2H, m), 3.72–3.76 (1H, m), 3.85–4.35 (2H, m), 4.95–5.03 (5H, m),
at À208C. When the substrate was consumed (the reaction usually
6
.74–6.90(1H, m), 7.44–7.49 (1H, m), 7.60–7.71 (2H, m), 7.82 (1H,
completed within 10 min), K CO (6.9 g, 50 mmol) was added. The
2
3
13
s), 7.93–7.96 (1H, m) ppm. C NMR (75 MHz, CDCl ): d=11.60,
reaction was allowed to warm to room temperature and stirred for
additional 10 min. The solvent was removed under vacuum and
3
11.62, 15.22, 15.26, 19.6, 20.2, 35.6, 37.7, 38.8, 41.1, 61.7, 62.8, 64.7,
6
1
5.1, 98.8, 101.3, 111.3, 111.7, 126.6, 127.4, 127.8, 128.3, 128.7,
29.6, 129.8, 145.2, 146.7, 148.0, 159.1, 170.3, 170.6 ppm. IR (KBr):
the residue was dissolved in 55 mL of H O. The aqueous layer was
2
extracted with warmed ethyl acetate. The combined organic layers
n˜ max =3382, 3061, 2965, 2931, 2874, 1641, 1602, 1502, 1437, 1409,
were dried over anhydrous MgSO , filtered, and evaporated to dry-
4
[18]
1379, 1329, 1225, 1203, 1167, 1131, 1109, 1077, 1041, 982, 906, 781,
ness to give a yellow solid 12a (crude yield 100%), which was
À1
+
7
49 cm . HRMS (ESI, m/z) calcd. for C H N O (M+H) : 383.1965;
22 26 2 4
pure enough to use in the next step without further purification.
1
found: 383.1973.
H NMR (300 MHz, CDCl ): d=4.83 (2H, s), 4.92 (2H, s), 7.55–7.60
3
(
6
1H, m), 7.72–7.77 (1H, m), 7.87 (1H, d, J=9.0 Hz), 8.10 (1H, d, J=
.0 Hz), 8.20 (1H, s) ppm. ESIMS (m/z): 190.33 (M+H) .
Compound 13b. The same procedure for 13a was applied for the
preparation of 13b. Flash chromatography (CH Cl /MeOH=25:1)
+
2
2
&
&
Chem. Asian J. 2014, 00, 0 – 0
4
ꢀ 2014 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
ÝÝ These are not the final page numbers!

 Xu, Peng
Xu, Peng
 Chen, Dong-Sheng
Chen, Dong-Sheng
 Xi, Jie
Xi, Jie
 Yao, Zhu-Jun
Yao, Zhu-Jun