
European Journal of Medicinal Chemistry p. 274 - 288 (2015)
Update date:2022-08-18
Topics:
 Sestito, Simona
Sestito, Simona
 Nesi, Giulia
Nesi, Giulia
 Daniele, Simona
Daniele, Simona
 Martelli, Alma
Martelli, Alma
 Digiacomo, Maria
Digiacomo, Maria
 Borghini, Alice
Borghini, Alice
 Pietra, Daniele
Pietra, Daniele
 Calderone, Vincenzo
Calderone, Vincenzo
 Lapucci, Annalina
Lapucci, Annalina
 Falasca, Marco
Falasca, Marco
 Parrella, Paola
Parrella, Paola
 Notarangelo, Angelantonio
Notarangelo, Angelantonio
 Breschi, Maria C.
Breschi, Maria C.
 Macchia, Marco
Macchia, Marco
 Martini, Claudia
Martini, Claudia
 Rapposelli, Simona
Rapposelli, Simona
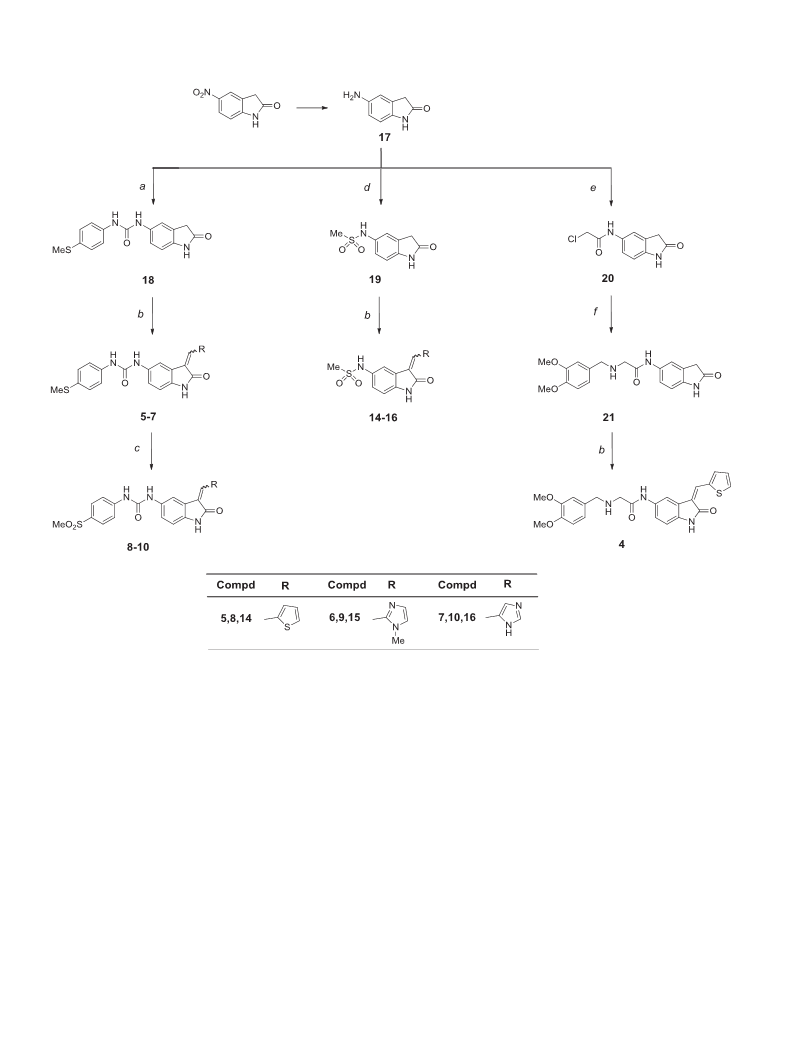
Aggressive behavior and diffuse infiltrative growth are the main features of Glioblastoma multiforme (GBM), together with the high degree of resistance and recurrence. Evidence indicate that GBM-derived stem cells (GSCs), endowed with unlimited proliferative potential, play a critical role in tumor development and maintenance. Among the many signaling pathways involved in maintaining GSC stemness, tumorigenic potential, and anti-apoptotic properties, the PDK1/Akt pathway is a challenging target to develop new potential agents able to affect GBM resistance to chemotherapy. In an effort to find new PDK1/Akt inhibitors, we rationally designed and synthesized a small family of 2-oxindole derivatives. Among them, compound 3 inhibited PDK1 kinase and downstream effectors such as CHK1, GS3Kα and GS3Kβ, which contribute to GCS survival. Compound 3 appeared to be a good tool for studying the role of the PDK1/Akt pathway in GCS self-renewal and tumorigenicity, and might represent the starting point for the development of more potent and focused multi-target therapies for GBM.
View More









Zhejiang Quzhou Jiancheng Silicone Co., Ltd.(Shanghai Jiancheng Industial and Trade Co, Ltd)
Contact:18957018777 +86-570-3888777
Address:The company production base address: Quzhou City, Zhejiang Province high-tech industrial park Nianhua Road 37
website:http://www.lidepharma.com
Contact:+86-25-58409506
Address:11F, Building A1, No.288 North Zhongshan Road, Gulou District, Nanjing,210003, P.R.China.
Shandong Xinke Petrochemical Co., Ltd.
Contact:+86-546-7277016
Address:Gudao Industrial Park, Hekou District, Dongying, Shandong Province, China
SHAANXI TOP PHARM CHEMICAL CO.LTD
Contact:+86-029-85733403
Address:No.108 ,west sector,south er huan,xi'an,china
A.M FOOD CHEMICAL CO., LIMITED
Contact:86-531-87100375
Address:20Floor,Bblock,1Building,pharma-valley,Jinan,China
Doi:10.1016/j.jphotochem.2011.05.003
(2011)Doi:10.1039/c4gc00581c
(2014)Doi:10.1002/prac.19973390127
(1997)Doi:10.1016/S1872-2067(14)60258-8
(2015)Doi:10.1021/ja01150a069
(1951)Doi:10.1016/S0040-4039(01)95651-5
(1973)