260
KHRIPACH et al.
with water (200 ml), and extracted with ethyl acetate.
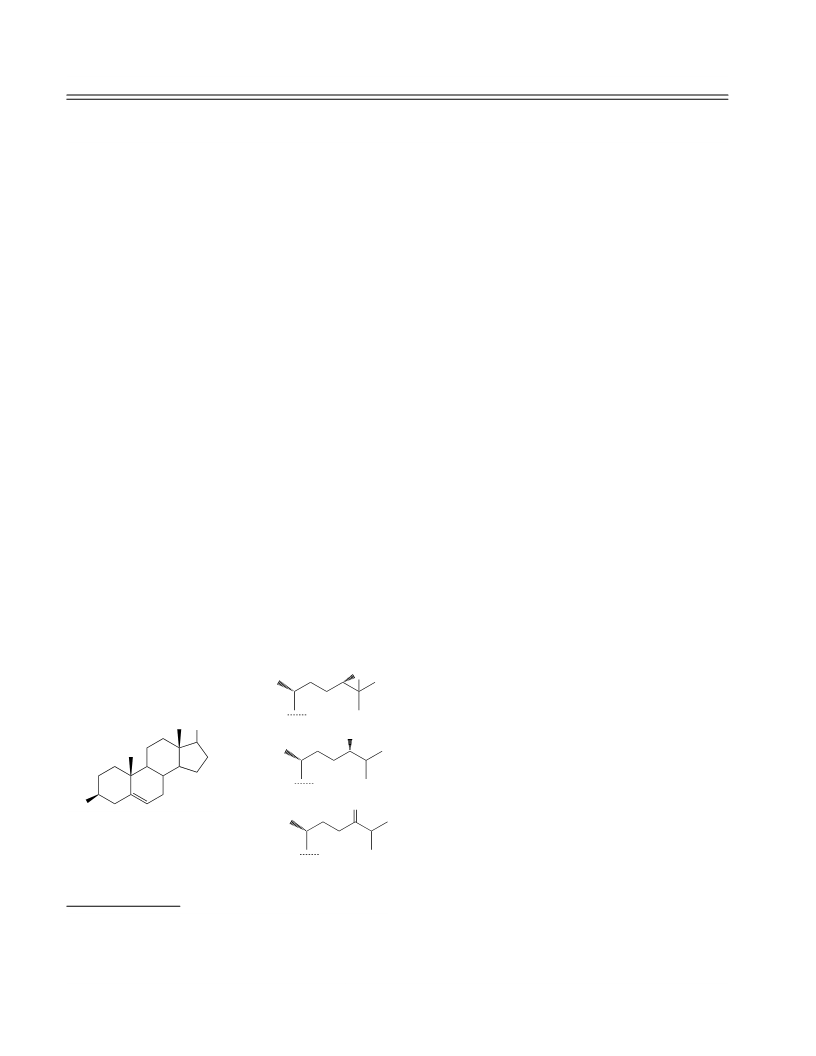
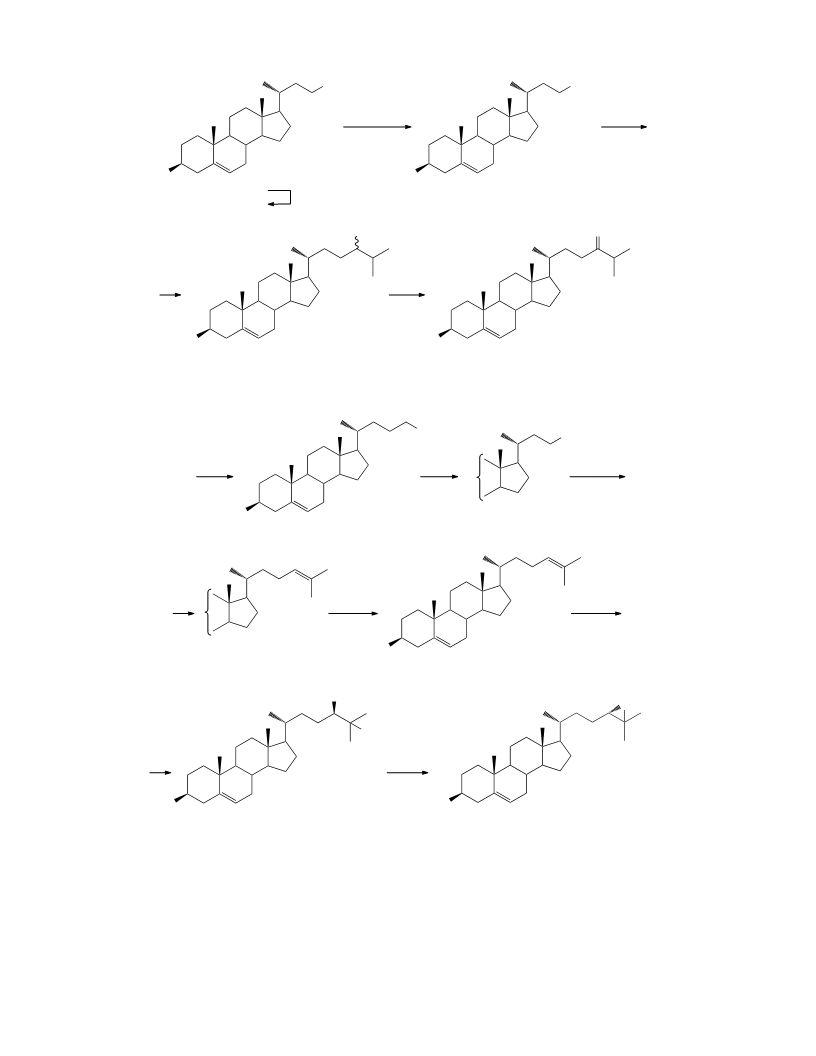
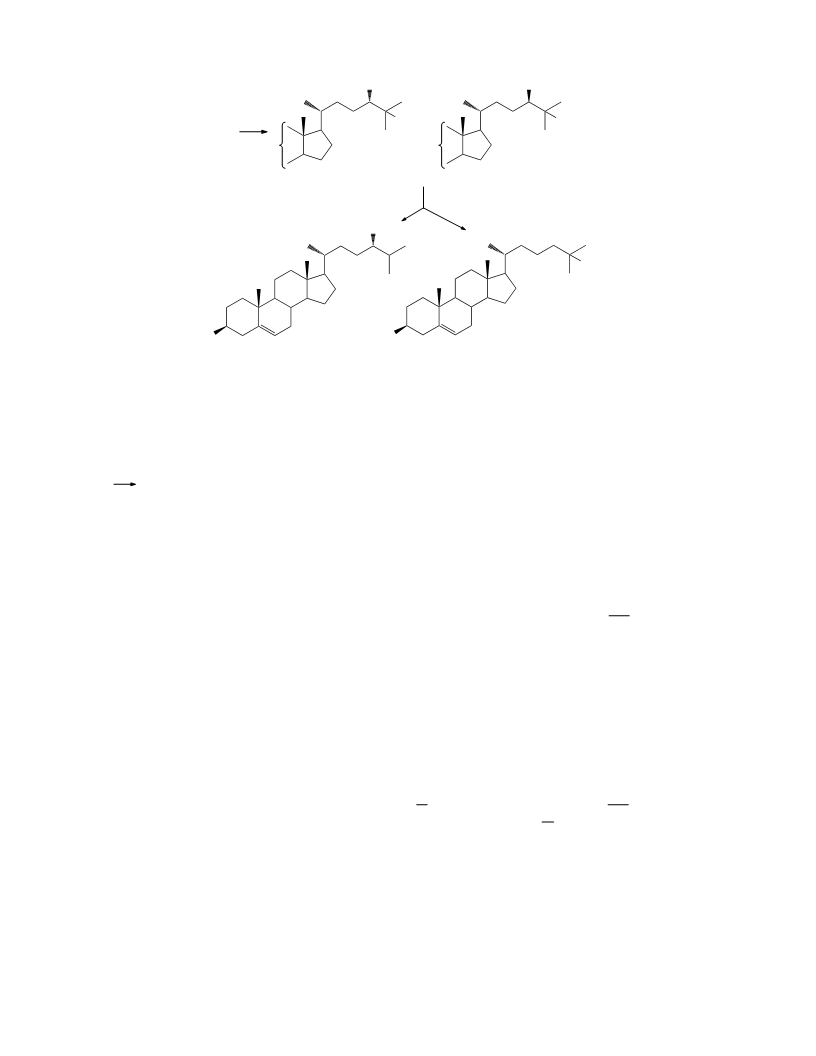
3b-Tetrahydropyranyloxycholesta-5,24-diene (IX).
The extract was dried over Na2SO4, evaporated, and A 2.5 M solution of butyllithium (4 ml, 10 mmol) was
added under nitrogen to a cooled to 0°C stirred suspen-
sion of isopropyltriphenylphosphonium iodide (4.31 g,
10 mmol) in THF (100 ml). After 30 min, a solution of
(VIII) (2.2 g, 5 mmol) in THF was added to the result-
ing bright red solution. The reaction mixture was stirred
for 2 h at room temperature and methyl iodide (0.4 ml)
was added. The resulting precipitate was filtered off,
thoroughly washed, and the filtrate was evaporated in a
vacuum. The residue was chromatographed on a silica
gel column eluted with 10 : 1 petroleum ether–ethyl
chromatographed on a silica gel column to give (VI);
yield 5.5 g (73%); 1H NMR: 0.68 (3 H, s, 18-CH3), 1.01
(3 H, s, 19-CH3), 3.31 (1 H, m, H24), 3.51 (2 H, m,
CH2é in THP), 3.89 (1 H, m, H3), 4.72 (1 H, br. s, ëç
in THP), and 5.35 (1 H, br. s, H6).
3b-Hydroxycholest-5-en-24-one (III). A solution
of (VI) (3.0 g) in pyridine (50 ml) was added to a solu-
tion of CrO3 (5.0 g, 50 mmol) in pyridine (100 ml). The
reaction mixture was kept for 12 h at room temperature,
diluted with diethyl ether, and filtered through a silica
gel layer. The filtrate was evaporated, twice coevapo-
rated with toluene, and dissolved in 2 : 1 methanol–
THF mixture (50 ml). Two milliliters of 36% hydro-
chloric acid was added, the mixture was kept for 30 min
at room temperature, pyridine (12 ml) was added, and
the solvents were removed in a vacuum. The residue
1
acetate to give (IX); yield 1.8 g (77%); H NMR:
0.67 (3 H, s, 18-CH3), 0.93 (3 H, d, J 6.7, 21-CH3), 1.00
(3 H, s, 19-CH3), 1.60 and 1.67 (2 × 3 H, 2 s, 26- and
27-CH3), 3.50 (2 H, m, CH2é in THP), 3.90 (1 H, m,
H3), 4.71 (1 H, br. s, ëç in THP), 5.08 (1 H, br. t, H24),
and 5.34 (1 H, br. s, H6).
was chromatographed on a silica gel column to yield
3b-Acetoxycholest-5,24-diene (X). Concentrate
1
1.8 g (73%) of (III); mp 134–136°ë. ç NMR: 0.67 hydrochloric acid (0.5 ml) was added to a solution of
(3 H, s, 18-CH3), 0.91 (3 H, d, J 6.4, 21-CH3), 1.01 (3H, (IX) (1.8 g, 3.85 mmol) in 2 : 1 THF–methanol mixture
(30 ml). The reaction mixture was kept for 1 h at room
temperature, treated with pyridine (2 ml), and evapo-
rated in a vacuum. The residue was coevaporated with
toluene, dissolved in pyridine (20 ml), and treated with
acetic anhydride (0.5 ml, 5 mmol) and dimethylami-
nopyridine (10 mg). The reaction mixture was kept for
12 h at room temperature and treated by a standard pro-
cedure. The target compound was isolated by column
chromatography on a silica gel column eluted with
10 : 1 petroleum ether–ethyl acetate; yield 1.4 g (85%);
1H NMR: 0.61 (3 H, s, 18-CH3), 0.86 (3 H, d, J 6.7, 21-
CH3), 0.95 (3 H, s, 19-CH3), 1.53 and 1.61 (2 × 3 H, 2
s, 26- and 27-CH3), 1.96 (3 H, s, 3-OCOCH3), 4.53 (1 H,
m, H3), 5.02 (1 H, t, J 6.7, H24), and 5.30 (1 H, d, J 4.2,
H6).
s, 19-CH3), 1.09 (6 H, d, J 6.5, 26- and 27-CH3), 3.62
(1 H, m, H3), and 5.34 (1 H, br. s, H6); 13C NMR: 11.9
q, 18.3 q, 18.4 q, 18.5 q, 19.4 q, 21.1 t, 24.3 t, 28.1 t,
29.9 t, 31.6 s, 31.9 t, 35.4 d, 36.5 s, 37.3 t, 39.8 t, 40.8
d, 42.3 t, 42.4 t, 50.1 d, 55.9 d, 56.7 d, 71.8 d, 121.6 d,
140.8 s, and 215.5 s.
3b-Tetrahydropyranyloxychol-5-en-24-ol (VII).
Lithium aluminum hydride (0.55 g, 10 mmol) was
added in small portions to a stirred solution of (V)
(4.73 g, 10 mmol). The reaction mixture was stirred for
1 h at room temperature; the excess LiAlH4 was
quenched by a standard procedure. The white precipi-
tate was filtered off and thoroughly washed with diethyl
ether. The filtrate was evaporated to give (VII), which
was further used without additional purification; yield
1
4.35 g (98%); H NMR: 0.68 (3H, Ò, 18-CH3), 0.94
3b-Acetoxy-24,25-dihydroxycholest-5-ene (XI).
AD-mix-β (4 g) and methanesulfonamide (0.35 g) were
added to a solution of (X) (2.13 g, 5 mmol) in 5 : 4 tert-
butanol–water mixture (100 ml). The reaction mixture
was stirred for 50 h at room temperature, treated with
sodium sulfite (4 g), and kept for 1 h. tert-Butanol was
removed in a vacuum, the residue was diluted with a
solution of NaHCO3, and the product was extracted
with chloroform. The organic layer was successively
washed with 0.1 M sulfuric acid, water, NaHCO3 solu-
tion, and water; dried; and then evaporated. The residue
was chromatographed on a silica gel column. Elution
with 2 : 1 petroleum ether–ethyl acetate yielded 2.0 g
(87%) of (XI); mp 159–161°ë; 1H NMR: 0.69 (3 H, s,
18-CH3), 0.93 (3 H, d, J 6.1, 21-CH3), 1.02 (3 H, s,
(3 H, d, J 6.7, 21-CH3), 1.00 (3H, s, 19-CH3), 3.50 (2 H,
m, CH2é in THP), 3.60 (2 H, br. t, H23), 3.90 (1 H, m,
H3), 4.72 (1 H, br. s, CH in THP), and 5.36 (1 H, br. s,
H6).
3b-Tetrahydropyranyloxychol-5-en-24-al (VIII).
Chromium (VI) oxide (7 g) was gradually added to a
mixture of pyridine (50 ml) and chloroform (150 ml) at
room temperature. The mixture was stirred for 30 min;
and a solution of (VII) (4.44 g, 10 mmol) in chloroform
(25 ml) was then added. After 12 h, isopropanol (20 ml)
and, after 30 min, diethyl ether (300 ml) were added.
The resulting mixture was filtered through a silica gel
layer, the solvents were removed in a vacuum, and the
residue was chromatographed on a silica gel column.
Elution with 5 : 1 petroleum ether–ethyl acetate led to 19-CH3), 1.15 and 1.20 (2 × 3 H, 2 s, 26- and 27-CH3),
(VIII); yield 3.8 g (86%); H NMR: 0.66 (3H, Ò, 18- 2.03 (3 H, s, 3-OCOCH3), 2.88 (2 H, br. s, 24- and 25-
1
CH3), 0.91 (3 H, d, J 6.1, 21-CH3), 0.99 (3H, s, 19-
CH3), 3.50 (2 H, m, CH2é in THP), 3.90 (1 H, m, H3),
4.70 (1 H, br. s, ëç in THP), 5.31 (1 H, br. s, H6), and 21.0 t, 21.4 q, 23.2 q, 24.2 t, 26.4 q, 27.7 t, 28.1 t, 28.2 t,
9.74 (1 H, s, H23).
OH), 3.31 (1 H, m, H24), 4.59 (1 H, m, H3), and 5.37
(1 H, d, J 4.2, ç6); C NMR: 11.9 q, 18.6 q, 19.3 q,
13
31.8 t, 31.8 d, 32.9 t, 35.7 d, 36.5 s, 36.9 t, 38.0 t, 39.7 t,
RUSSIAN JOURNAL OF BIOORGANIC CHEMISTRY Vol. 28 No. 3 2002

 Khripach
Khripach
 Zhabinskii
Zhabinskii
 Konstantinova
Konstantinova
 Khripach
Khripach
 Antonchick
Antonchick