Total Synthesis of (+)-Quassin
J . Org. Chem., Vol. 65, No. 21, 2000 7067
1
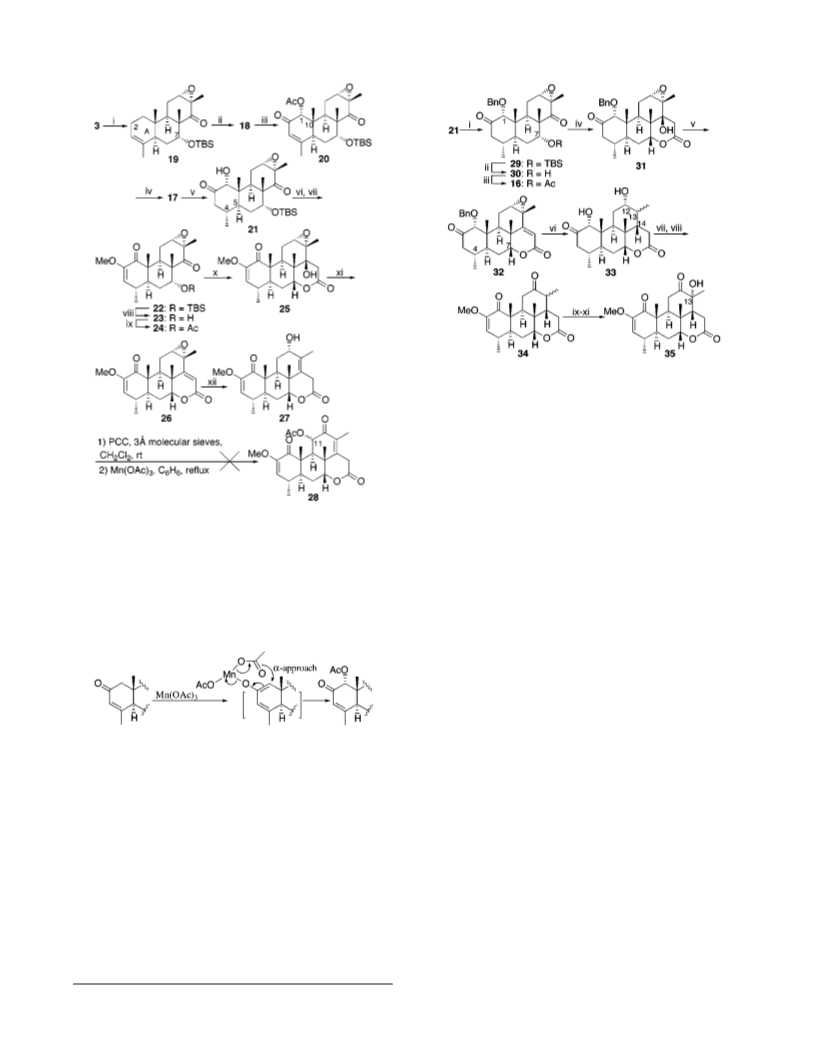
2r-H yd r oxy-2-m e t h oxyp icr a sa -2,13-d ie n e -1,16-d i-
23, the alcohol 30 (30 mg, 0.073 mmol) was converted into the
on e (27). Following the same procedure used to prepare 12
from 11, the R,â-unsaturated lactone 26 (30 mg, 0.084 mmol)
was converted into the alcohol 27 (9 mg, 30%) as a white
acetate 16 (31 mg, 94%) as a white solid: R
f
0.64 (2:1 Et
2
O/
hexane); mp 173-174 °C; [R]
D
) -66.0 (c ) 1.5, CHCl ); MS
3
+
-1
(EI) m/z 455 [M + H] ; IR 1744 (ester CdO), 1706 cm (ketone
1
1
solid: R
f
0.32 (3:1 EtOAc/hexane); mp 201-202 °C; H NMR
CdO); H NMR (300 MHz) δ 0.70 (3 H, s), 0.92 (3 H, d, J )
(
250 MHz) δ 1.09 (3 H,s), 1.14 (3 H, d, J ) 6.9 Hz), 1.17 (3 H,
s), 1.72 (2 H, brs), 1.78 (3 H, s), 1.84 (1 H, d, J ) 7 Hz), 2.05
1 H, dd, J ) 13, 2 Hz), 2.15 (1 H, dd, J ) 14, 5.8 Hz), 2.23 (1
H, d, J ) 6 Hz), 2.35 (1 H, m), 2.65 (1 H, bd, J ) 14 Hz), 3.08
1 H, bd, J ) 15 Hz), 3.38 (1 H, d, J ) 15 Hz), 3.57 (3 H, s),
.09 (1 H, brs), 4.23 (1 H, brs), 5.36 (1 H, d, J ) 2.5 Hz); 13
NMR (62.89 MHz) δ 11.6, 15.7, 19.6, 24.7, 29.7, 30.7, 31.6, 32.7,
6.9, 38.7, 43.0, 45.5, 55.0, 68.8, 78.7, 117.4, 132.2, 132.4, 147.8,
71.8, 199.9.
1
6.6 Hz), 1.01 (3 H, s), 1.40 (3 H, s), 1.44 (1 H, d, J ) 14.7 Hz),
1.62-1.73 (3 H, m), 2.03 (3 H, s), 2.07 (1 H, m), 2.15 (1 H, bd,
J ) 10.8 Hz), 2.25 (1 H, dd, J ) 13.8, 4.2 Hz), 2.50 (1 H, t, J
) 13 Hz), 3.03 (1 H, dd, J ) 11.4, 4.8 Hz), 3.22 (1 H, brs), 3.27
(1 H, brs), 4.25 (1 H, d, J ) 12 Hz), 4.59 (1 H, d, J ) 11.7 Hz),
(
(
4
13
C
5.30 (1 H, brs), 7.30-7.42 (5 H, m); C NMR (62.89 MHz) δ
13.7, 16.4, 18.0, 19.7, 19.9, 21.1, 24.5, 28.6, 32.3, 37.6, 42.7,
46.3, 49.9, 56.5, 59.2, 71.0, 71.9, 83.3, 128.0, 128.4, 128.5, 136.8,
3
1
170.1, 205.7, 211.1; HRMS calcd for C27
455.2433, found 455.2446.
H
35
O
6
(M + H)
r-Ben zyloxy-7r-(ter t-b u t yld im et h ylsiloxy)-12r,13r-
ep oxy-4r,8â,13â-t r im et h yl-19-n or p od oca r p a n e-2,14-d i-
on e (29). To a stirred solution of keto alcohol 21 (20 mg, 0.046
mmol) in dry THF (5 mL) was added NaH (60%, 18 mg, 0.45
1r-Ben zyloxy-12r,13r-ep oxy-14â-h yd r oxyp icr a sa n e-
2,16-d ion e (31). Following the same cyclization procedure
used to prepare 25 from 24, the acetate 16 (24 mg, 0.053 mmol)
was converted into the lactone 31 (21.5 mg, 90%) as a white
2
mmol) at 0 °C under N . After 1 h at 0 °C, the reaction mixture
was added BnBr (0.11 mL, 0.93 mmol) and a catalytic amount
of tetra-n-butylammonium iodide (TBAI). The reaction mixture
was allowed to warm to room temperature and was then
stirred for 8 h. The reaction was quenched by slowly addition
solid: R
f
0.21 (2:1 Et
2
O/hexane); mp 187-188 °C; [R] ) +10.8
D
+
(c ) 1.6, CHCl
3
); MS (EI) m/z 455 [M] ; IR 3418 (OH), 1714
-
1
1
cm (CdO); H NMR (300 MHz) δ 0.74 (3 H, s), 1.00 (3H, d,
J ) 5.4 Hz), 1.01 (3H, s), 1.36 (3H, s), 1.43 (1 H, d, J ) 5.7
Hz), 1.52 (1 H, d, J ) 15 Hz), 1.61 (1H, m), 1.68-1.83 (2H,
m), 2.02-2.17 (2H, m), 2.21 (1H, d, J ) 4.8 Hz), 2.26 (1H, t, J
) 3.3 Hz), 2.51 (1H, t, J ) 13 Hz), 2.68 (1H, d, J ) 18.6 Hz),
3.07 (1H, brs), 3.15 (1H, brs), 3.34 (1H, d, J ) 18.9 Hz), 4.15
of MeOH (0.2 mL) followed by saturated NH
aqueous phase was extracted with CH Cl
(3 ×). The combined
organic extracts were washed with brine, dried (MgSO ), and
4
Cl (2 mL). The
2
2
4
filtered. Concentration of the filtrate followed by flash column
chromatography (9:1 hexane/EtOAc) afforded the ketone 29
(1H, d, J ) 12 Hz), 4.56 (1H, d, J ) 12 Hz), 4.62 (1H, t, J ) 3
Hz), 7.28-7.39 (5H, m); H NMR (300 MHz, CDCl
1
(
20.5 mg, 85%) as a white solid: R
f
0.61 (1:1 Et
); MS (EI) m/z
27 [M + H] ; IR 1709 cm (ketone CdO); H NMR (300 MHz)
δ 0.06 (3 H, s), 0.07 (3 H, s), 0.68 (3 H, s), 0.83 (9 H, s), 0.92
2
O/hexane);
3
+ D O) δ
2
mp 121-122 °C; [R] ) -65.2 (c ) 1.0, CHCl
5
D
3
0.74 (3H, s), 1.00 (3H, d, J ) 5.1 Hz), 1.01 (3H, s), 1.36 (3H,
s), 1.43 (1H, d, J ) 5.7 Hz), 1.52 (1H, d, J ) 15 Hz), 1.61 (1H,
m), 1.68-1.80 (1H, m), 2.02-2.17 (2H, m), 2.21 (1H, d, J )
4.5 Hz), 2.26 (1H, t, J ) 3.3 Hz), 2.51 (1H, t, J ) 13 Hz), 2.68
(1H, d, J ) 18.6 Hz), 3.07 (1H, brs), 3.15 (1H, brs), 3.34 (1H,
d, J ) 18.6 Hz), 4.15 (1H, d, J ) 12 Hz), 4.56 (1H, d, J ) 12
Hz), 4.63 (1H, t, J ) 3 Hz), 7.29-7.39 (5H, m); 13C NMR (62.89
MHz) δ 13.0, 14.3, 19.0, 19.9, 21.5, 25.9, 30.2, 32.6, 36.6, 37.5,
37.6, 42.6, 46.0, 60.4, 60.5, 71.1, 74.1, 81.2, 83.2, 128.1, 128.5,
+
-1
1
(
3 H, s), 0.93 (3 H, d, J ) 6 Hz), 1.37 (3 H, s), 1.64-1.74 (4 H,
m), 1.78 (1 H, dt, J ) 11, 3 Hz), 2.25 (1 H, dd, J ) 13.8, 4.2
Hz), 2.47 (1 H, d, J ) 12.6 Hz), 2.51 (1 H, d, J ) 12.6 Hz), 3.23
(
4
1 H, brs), 3.29 (1 H, brs), 3.35 (1 H, dd, J ) 11.4, 4.2 Hz),
.26 (1 H, brs), 4.30 (1 H, d, J ) 12 Hz), 4.49 (1 H, d, J ) 12
13
Hz), 7.25-7.40 (5 H, m); C NMR (62.89 MHz) δ -5.5, -3.9,
1
4
2
4.0, 16.7, 18.0, 18.9, 19.7, 19.9, 25.7, 27.6, 32.4, 36.7, 42.8,
6.7, 52.7, 57.2, 59.5, 70.7, 71.4, 84.5, 127.7, 128.1, 128.3, 137.2,
136.5, 171.7, 211.4; HRMS calcd for C27
455.2433, found 455.2443.
1r-Ben zyloxy-12r,13r-ep oxyp icr a s-14-en e-2,16-d ion e
(32). Following the same dehydration procedure used in the
preparation of 11, the lactone 31 (15 mg, 0.033 mmol) was
converted to the R,â-unsaturated lactone 32 (13.5 mg, 94%)
H
35
O
6
(M + H)
08.4, 211.9; HRMS calcd for C31
H
47
O
5
Si (M + H) 527.3192,
found 527.3173.
r-Ben zyloxy-12r,13r-ep oxy-7r-h yd r oxy-4r,8â,13â-tr i-
m eth yl-19-n or p od oca r p a n e-2,14-d ion e (30). To a solution
of ketone 29 (53 mg, 0.10 mmol) in dry CH Cl (8 mL) was
added Et O‚BF (63 µL, 0.50 mmol) at 0 °C under N . The
1
2
2
as a white solid. Recrystallization from a mixture of Et
CH Cl gave a single crystal which was analyzed by X-ray to
prove the structure: R 0.24 (2:1 Et O/hexane); mp 193-194
°C; [R] ) -82.8 (c ) 1.1, CHCl ); MS (EI) m/z 437 [M + H] ;
2
O and
2
3
2
2
2
reaction mixture was allowed to warm to about 10 °C and was
then stirred for 6 h. The reaction was quenched with saturated
f
2
+
D
3
-
1
1
NH
Cl
brine, dried (MgSO
followed by flash column chromatography (7:1 hexane/EtOAc)
yielded the alcohol 30 (38 mg, 92%) as a white solid: R 0.31
1:1 Et O/hexane); mp 143-144 °C; [R] ) -36.5 (c ) 2.1,
CHCl ); MS (EI) m/z 413 [M + H] ; IR 3563 (OH), 1703 cm
ketone CdO); H NMR (300 MHz) δ 0.68 (3 H, s), 0.96 (3 H,
s), 1.00 (3 H, d, J ) 6.6 Hz), 1.42 (3 H, s), 1.43-1.53 (2 H, m),
4
Cl (2 mL), and the aqueous phase was extracted with CH
(3×). The combined organic extracts were washed with
), and filtered. Concentration of the filtrate
2
-
IR 1714 cm (CdO); H NMR (300 MHz) δ 0.70 (3H, s), 1.04
(3H, d, J ) 6.6 Hz), 1.10 (3H, s), 1.42 (1H, dd, J ) 15.3 and
5.7 Hz), 1.53 (3H, s), 1.65-1.77 (3H, m), 2.12-2.28 (4H, m),
2
4
1
2.57 (1H, t, J ) 13 Hz), 3.10 (2H, brs), 4.12 (1H, d, J ) 2.3
f
Hz), 4.29 (1H, t, J ) 3 Hz), 4.55 (1H, d, J ) 12 Hz), 6.18 (1H,
s), 7.25-7.38 (5H, m); 13C NMR (62.89 MHz) δ 12.2, 18.5, 20.0,
21.0, 25.8, 33.3, 36.9, 37.3, 42.3, 45.8, 55.8, 58.8, 70.9, 78.4,
82.8, 116.1, 128.1, 128.5, 128.9, 136.2, 164.7, 211.4; HRMS
(
2
D
+
-1
3
1
(
calcd for C27
33 5
H O (M + H) 437.2328, found 437.2320.
1
1
2
4
.61 (1 H, d, J ) 13.5 Hz), 1.70 (1 H, m), 1.97 (1 H, dt, J )
4.4, 3 Hz), 1.99 (1 H, brs), 2.23 (1 H, dd, J ) 13.5, 3.9 Hz),
.33 (1 H, m), 2.55 (1 H, t, J ) 13 Hz), 2.91 (1 H, dd, J ) 12.6,
1r,12r-Dih yd r oxyp icr a sa n e-2,16-d ion e (33). To a sus-
pension of palladium on activated carbon (10%, 2 mg) in EtOH
(1 mL), which had been stirred for 30 min under H atmo-
2
sphere at room temperature, was added a solution of R,â-
unsaturated lactone 32 (15 mg, 0.034 mmol) in EtOH (5 mL).
.2 Hz), 3.20 (2 H, brs), 4.16 (1 H, d, J ) 12 Hz), 4.17 (1 H,
1
brs), 4.61 (1 H, d, J ) 12 Hz), 7.30-7.37 (5 H, m); H NMR
(300 MHz, CDCl
3
+ D
2
O) δ 0.68 (3 H, s), 0.96 (3 H, s), 1.00 (3
The reaction mixture was stirred for 2 days under H
then filtered. Concentration of the filtrate followed by flash
column chromatography (2:1 Et O/CH Cl ) provided the alcohol
33 (11 mg, 92%) as a white solid: R 0.16 (4:1 Et O/hexane);
); MS (EI) m/z
2
and was
H, d, J ) 6.3 Hz), 1.42 (3 H, s), 1.40-1.53 (2 H, m), 1.61 (1 H,
d, J ) 13.2 Hz), 1.71 (1 H, m), 1.97 (1 H, dt, J ) 14.1, 3 Hz),
2
2
2
2
.23 (1 H, dd, J ) 13.8, 4.2 Hz), 2.33 (1 H, m), 2.55 (1 H, t, J
13 Hz), 2.90 (1 H, dd, J ) 12.3, 3.9 Hz), 3.20 (2 H, brs), 4.16
f
2
)
mp 215 °C dec; [R]
D
) +23.3 (c ) 0.5, CHCl
3
+
-1
(
1 H, d, J ) 12 Hz), 4.17 (1 H, brs), 4.61 (1 H, d, J ) 12.3 Hz),
350 [M] ; IR 3390 (OH), 1722 (lactone CdO), 1714 cm
.28-7.37 (5 H, m); 13C NMR (62.89 MHz) δ 13.3, 16.2, 17.3,
9.7, 19.9, 26.3, 27.5, 32.7, 36.5, 43.0, 46.4, 51.7, 56.1, 59.3,
9.3, 70.8, 83.1, 128.0, 128.4, 128.9, 136.7, 209.3, 211.7; HRMS
(ketone CdO); H NMR (300 MHz) δ 0.66 (3H, s), 0.98 (3H, d,
1
7
1
6
J ) 6.3 Hz), 1.01 (3H, d, J ) 7.2 Hz), 1.11 (3H, s), 1.60-1.80
(6H, m), 1.92-1.97 (1H, m), 2.12 (1H, bd, J ) 12.6 Hz), 2.19
(1H, dd, J ) 14, 4 Hz), 2.42 (1H, dd, J ) 11.7, 3.9 Hz), 2.51
(1H, dd, J ) 19.5, 6.6 Hz), 2.58 (1H, dd, J ) 13.8, 12.6 Hz),
3.37 (1H, dd, J ) 19.2, 12.3 Hz), 3.49 (2H, brs), 3.60 (1H, s),
calcd for C25
33 5
H O (M + H) 413.2328, found 413.2321.
7
r-Acetoxy-1r-ben zyloxy-12r,13r-ep oxy-4r,8â,13â-tr i-
m eth yl-19-n or p od oca r p a n e-2,14-d ion e (16). Following the
same acetylation procedure used in the preparation of 24 from
1
4.01 (1H, d, J ) 2.1 Hz), 4.20 (1H, brs); H NMR (300 MHz,

 Shing, Tony K. M.
Shing, Tony K. M.
 Jiang, Qin
Jiang, Qin