
Journal of Heterocyclic Chemistry p. 1221 - 1228 (1993)
Update date:2022-08-16
Topics:
 Mosselhi
Mosselhi
 Pfleiderer
Pfleiderer
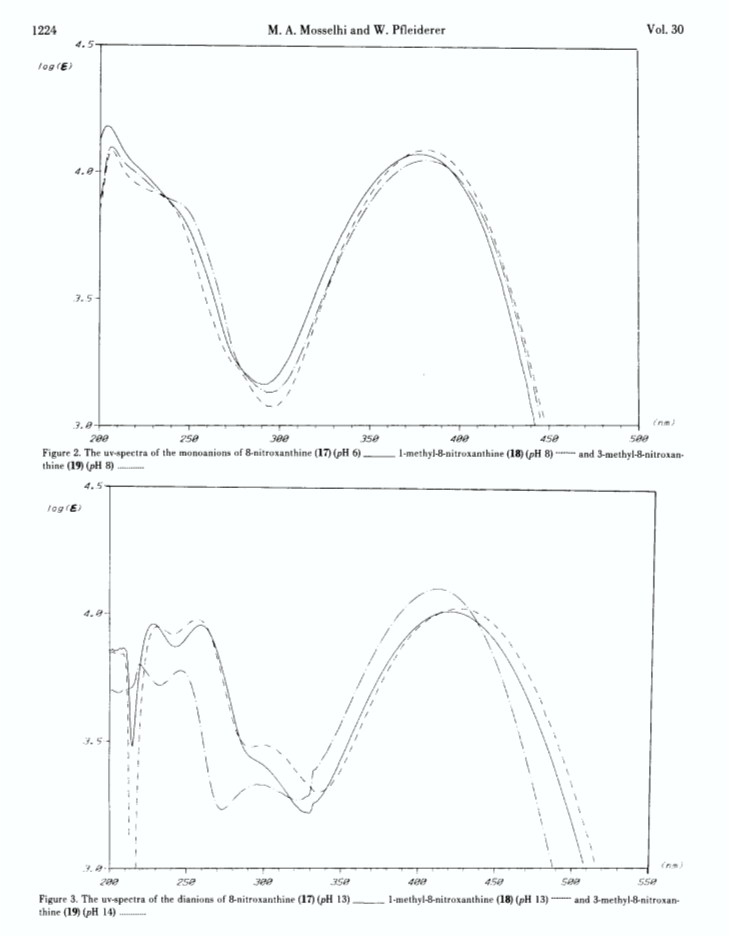
Xanthine (1) and its N-methyl derivatives 2-16 have been nitrated to the corresponding 8-nitro derivatives 17-32 under different reaction conditions. Nitration in glacial acetic acid with nitric acid works well with the N-7 unsubstituted and some of the 9-methylxanthines, respectively, whereas the 7- methylxanthine derivatives react best with nitronium tetrafluoroborate in sulfolane or glacial acetic acid. The 8-nitro group can be displaced nucleophilically to form 8-chloro-, 33, 34, 8-ethoxy-, 35, 36, and uric acid derivatives 37-40, respectively. The newly synthesized 8-nitroxanthines have been characterized by elemental analyses, pK-determinations and uv and 1H- nmr spectra.
View More





Zhonghao (dalian) Research and Design Institute of Chemical Industry Co., Ltd
Contact:+86 411 84674606
Address:201, Huangpu Road , Shahekou District, Dalian ,116023-China
Shanghai KFSL Pharmaceutical Technology Co.,Ltd.
Contact:+86-21-39971718
Address:859 jiadingchengliu shanghai
Tianjin Te-An Chemtech Co., Ltd.
Contact:+86-22-65378638
Address:A5-8, No.80 Haiyun Street, TEDA
Shanghai Norky Pharmaceutical Co., LTD.
website:http://www.norkypharm.com
Contact:86-21-61075300
Address:1165 Jiangning Road, Office 1502, Shanghai, China
Contact:86-21-50966856
Address:Building 5,300 Chuanzhan Road,Pudong New District,Shanghai
Doi:10.1166/jnn.2012.6301
(2012)Doi:10.1002/mabi.201300259
(2014)Doi:10.1016/S0040-4039(97)10386-0
(1997)Doi:10.1039/c7ra02788e
(2017)Doi:10.1111/j.1551-2916.2012.05272.x
(2012)Doi:10.1039/ft9938901001
(1993)