
Helvetica Chimica Acta (2019)
Update date:2022-08-12
Topics:
 Pigot, Corentin
Pigot, Corentin
 Noirbent, Guillaume
Noirbent, Guillaume
 Peralta, Sébastien
Peralta, Sébastien
 Duval, Sylvain
Duval, Sylvain
 Nechab, Malek
Nechab, Malek
 Gigmes, Didier
Gigmes, Didier
 Dumur, Frédéric
Dumur, Frédéric
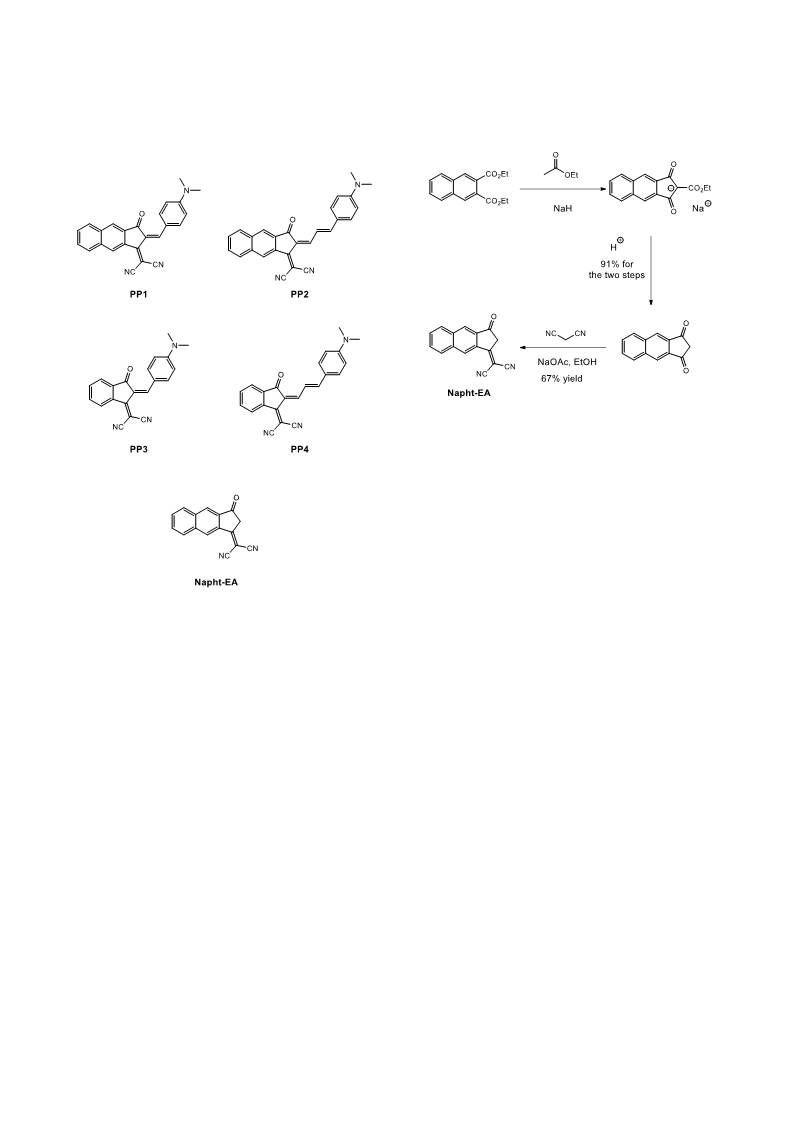
An unprecedented nucleophilic addition of piperidine on an electron acceptor, namely, 2-(3-oxo-2,3-dihydro-1H-cyclopenta[b]naphthalen-1-ylidene)malononitrile is reported. This unexpected behavior was observed during the synthesis of push-pull dyes using the classical Knoevenagel reaction. To overcome this drawback, use of diisopropylethylamine (DIPEA) enabled to produce the expected dyes PP1 and PP2. The optical and electrochemical properties of the different dyes were examined. Theoretical calculations were also carried out to support the experimental results. To evidence the higher electron-withdrawing ability of this electron acceptor, a comparison was established with two dyes (PP3 and PP4) comprising its shorter analogue.
View More


taicang liyuan chemical co,.ltd
website:http://www.tcliyuanchem.com/productse.php
Contact:86-512-53539583
Address:Room 804,Huaxu Building,No.95,Renmin South Road,Taicang city,Jiangsu Province,China
Shanghai Synmedia Chemical Co., Ltd
Contact:+86-21-38681880
Address:6th Floor, 11A Building, No.528 Ruiqing Road, Heqing town, Pudong new district, Shanghai China
Contact:0512-63006287
Address:no.88.YISHENG Road,etdz-WUJIANG,SUZHOU,CHINA
Beijing Mesochem Technology Co.,LTD
website:http://www.mesochem.com
Contact:0086-10-57862036
Address:2301, Floor 23, Building 9 Lippo Plaza, Yard 8 Ronghua Middle Road, ETDZ, Beijing, China
website:http://www.josunpharma.com
Contact:+86-311-80715268 80766839
Address:No.39, Zhaiying Street, Shijaizhuang,Hebei,China
Doi:10.1039/c4cc03699a
(2014)Doi:10.1080/15421406.2015.1107745
(2016)Doi:10.1149/1.1753582
(2004)Doi:10.1016/j.bbalip.2015.11.009
(2016)Doi:10.1002/anie.202103447
(2021)Doi:10.1007/BF02532620
(1974)