
Journal of Organic Chemistry p. 4950 - 4955 (1994)
Update date:2022-08-10
Topics:
 Boger, Dale L.
Boger, Dale L.
 Kochanny, Monica J.
Kochanny, Monica J.
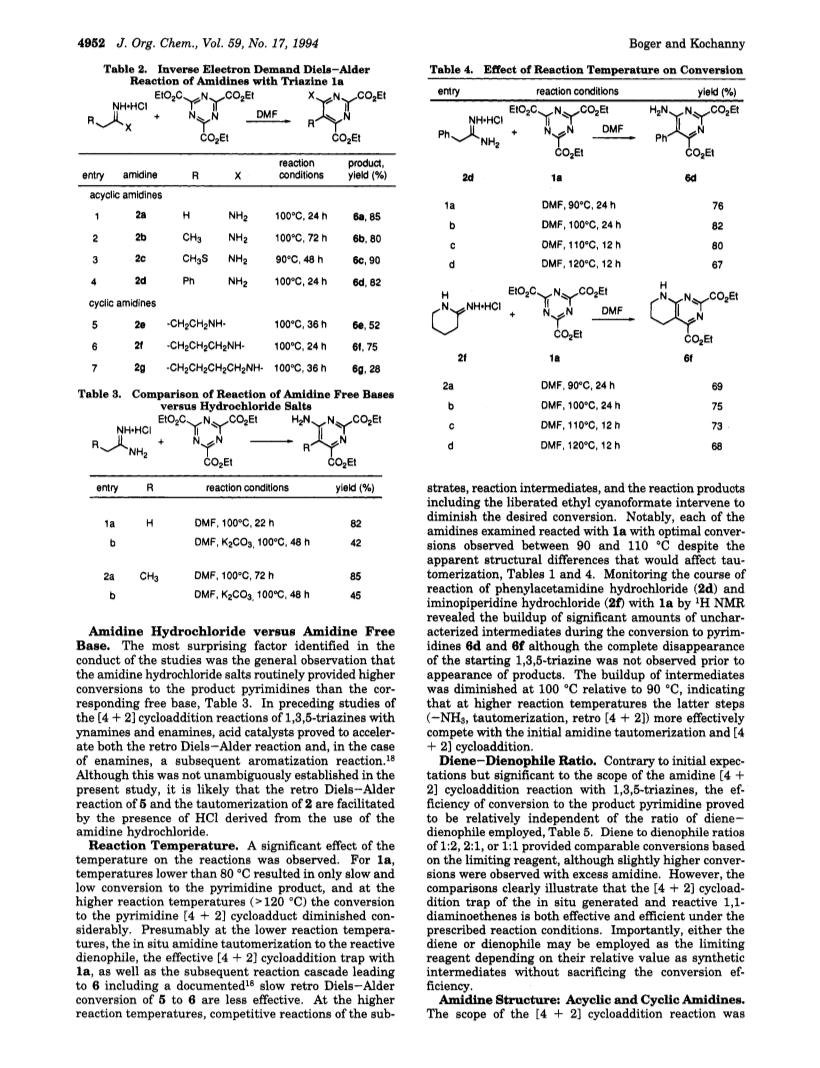
A detailed study of the scope of the amidine Diels-Alder reaction with 1,3,5-triazines is described.The thermal reaction of amidines with symmetrical 1,3,5-triazines proceeds with in situ amidine to 1,1-diaminoethene tautomerization, <4 + 2> cycloaddition with the 1,3,5-triazine, loss of ammonia from the initial Diels-Alder adduct with imine generation, imine to enamine tautomerization, and retro Diels-Alder loss of ethyl cyanoformate to provide substituted 4-aminopyrimidines in excellent conversions.The reaction proceeds best with the amidine hydrochloride salts at intermediate reaction temperatures (90-100 deg C) in polar, aprotic solvents, is rather invariant to the ratio of dienophile-diene used (<1:2<*>1:1<*>2:1), and is subject to triazine substituent effects characteristic of an inverse electron demand Diels-Alder reaction (R = CO2Et > R = H >> R = SCH3).Notably, the generality of the amidine <4 + 2> cycloaddition reaction with 1,3,5-triazines which has been extended to include cyclic amidines effectively addresses the limitations of the alternative ynamine or N,O-ketene acetal dienophiles.A comparative examination of amidines, thioimidates, and imidates revealed that amidines are uniquely suited for use in this reaction cascade.
View More




Jiande City Silibase Silicone New Material Manufacture Co., Ltd.
Contact:15967177856
Address:Genglou Industrial Development Area
Nanjing Chemlin Chemical Co., Ltd.
website:http://www.echemlin.cn
Contact:+86-25-83697070
Address:Rm.902 Longyin Plaza, No. 217 Zhongshan Rd.(N) Nanjing 210009,China
Contact:+86-21-38122007
Address:2, Lane 1123, Kangqiao Road, Pudong New Area, Shanghai
Shanghai Egoal Chemical Co.,Ltd
Contact:+86-21-50333091
Address:Yangming Garden Square 3,YangGao North Road 1188, Pudong New District, Shanghai
Liaoyang hengye Chemical Co., Ltd.
Contact:86-419-5850866
Address:North Old Xiaoxiao Road,Yantai District, Dengta, Liaoyang, Liaoning, China
Doi:10.1002/cjoc.201400403
(2014)Doi:10.1002/(SICI)1096-9888(200002)35:2<218::AID-JMS932>3.0.CO;2-8
(2000)Doi:10.1139/v51-088
(1951)Doi:10.1016/0022-328X(89)87225-0
(1989)Doi:10.1039/b407074g
(2004)Doi:10.1002/chem.201904890
(2020)