
Journal of Porphyrins and Phthalocyanines p. 222 - 230 (2017)
Update date:2022-08-28
Topics:
 Chaudhri, Nivedita
Chaudhri, Nivedita
 Sawhney, Nipun
Sawhney, Nipun
 Madhusudhan, Bijjam
Madhusudhan, Bijjam
 Raghav, Anubhav
Raghav, Anubhav
 Sankar, Muniappan
Sankar, Muniappan
 Satapathi, Soumitra
Satapathi, Soumitra
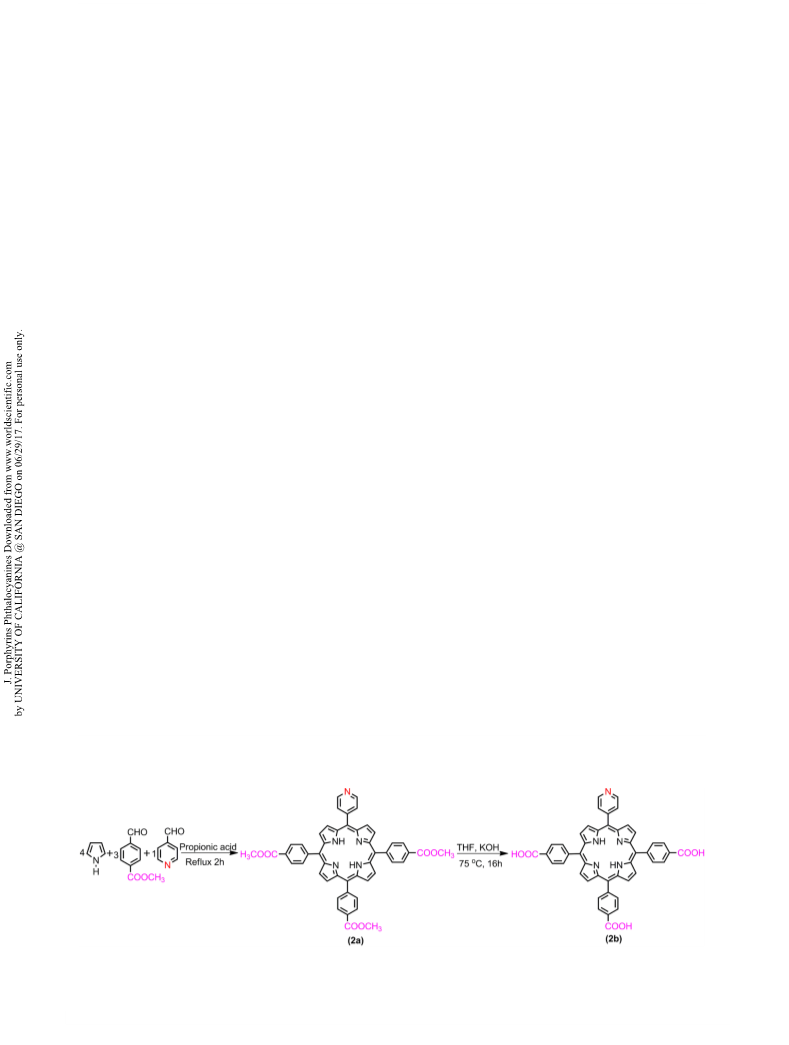
Dye-sensitized solar cells (DSSCs) were fabricated with six meso-substituted A3B and A4 free base porphyrin dyes having different functional groups, as sensitizers. The two step synthesis and the effect of different functional groups and their positions on the photosensitization properties of these porphyrin dyes are reported. The highest power conversion efficiencies (η) of 3.26%, 2.94% and 2.84% were achieved for the DSSC fabricated using 5,10,15-tris(4′-pyridyl)-20-(4′-carboxyphenyl)porphyrin (H2TriPyMCPP), 5,10,15,20-tetrakis(4′-aminophenyl)porphyrin (H2TAPP) and 5-(4′-pyridyl)-10,15,20-tris(4′-carboxyphenyl)porphyrin (H2MPyTriCPP) dyes, respectively. The electron donating amino group is shown to enhance the power conversion efficiency while pyridyl appended porphyrin sensitizers are shown to be superior sensitizers as compared to carboxyphenylporphyrins. The investigation of effect of functional group and position of functional group of porphyrin dye on DSSC can serve as an important tool to guide further selection and synthesis of potential candidates as sensitizers.
View More







Changzhou Sunlight Pharmaceutical Co., Ltd.
Contact:+86-519-83131668;83139028;83138042;83137041
Address:JiuliStreet, Benniu Town Changzhou City, Jiangsu Province
Nanjing Ruizhi Industry & Technologh Co.,Ltd.
Contact:+86-25-86808110
Address:441-4-A5,NO.12 Longzang Avenue,Yuhuatai District,210039,Nanjing
Zibo Linzi Darong Fine Chemical Co., Ltd
Contact:86-532-67773200; 15689126900
Address:Qidu town,Linzi district,Zibo city,Shandong province,China
DANYANG ZHONGTAI TRADE CO .,LTD.
Contact:0086-511-86985511
Address:No.25,Dongfang Rd., Danyang City.Jiangsu P.R.China
Contact:+86-025-52406782
Address:8 Taizishan Rd., Yanjiang Industrial Development Area, Nanjing, Jiangsu, China.
Doi:10.1021/j100669a031
(1972)Doi:10.1002/zaac.19794590110
(1979)Doi:10.1055/s-0040-1705974
(2021)Doi:10.1016/j.jcat.2010.07.004
(2010)Doi:10.1002/(SICI)1097-4601(1997)29:8<607::AID-KIN6>3.0.CO;2-Y
(1997)Doi:10.1002/jps.2600540304
(1965)