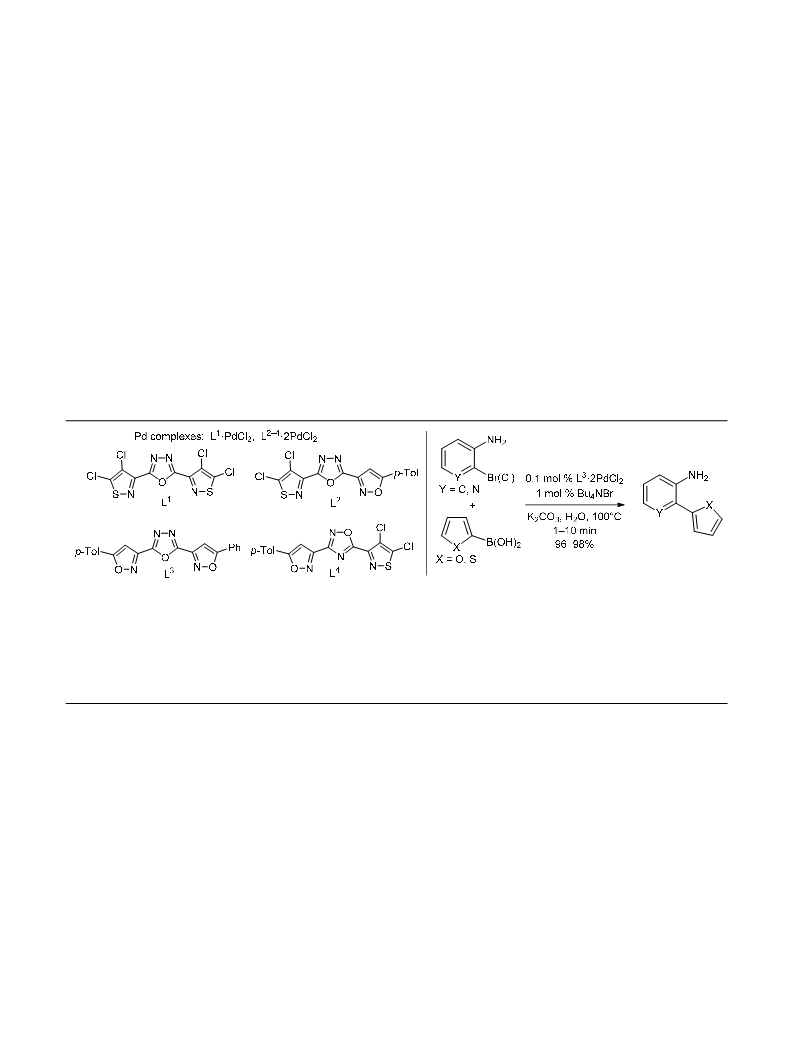
Chemistry of Heterocyclic Compounds 2017, 53(12), 1340–1349
–
1
1
ν, cm : 3503, 3400, 3136, 2919, 1762, 1621, 1570, 1509,
678. H NMR spectrum, δ, ppm: 7.56–7.66 (3Н, m, H Ph);
7.96 (1Н, s, СН isoxazole); 8.02–8.11 (2Н, m, H Ph).
Found, %: C 46.00; H 1.88; Cl 19.28; N 15.47; S 8.66.
1
9
451, 1355, 1299, 1223, 1127, 1108, 1086, 1000, 966, 948,
1
05, 811, 765, 694, 499, 483. H NMR spectrum, δ, ppm
(
(
J, Hz): 2.38 (3H, s, CH
2H, br. s, NH
3
); 7.39 (2Н, d, J = 8.0, H Ar); 7.44
C
14
H
6
Cl
2
N
4
O S. Calculated, %: C 46.05; H 1.66; Cl 19.41;
2
2
); 7.74 (1Н, s, СН isoxazole); 7.81 (2Н, d,
N 15.34; S 8.78.
13
J = 8.0, H Ar). C NMR spectrum, δ, ppm: 21.6 (CH
3
);
01.1 (СН isoxazole); 123.4; 124.1; 126.2 (2CН Ar); 130.5
2CН Ar); 141.6; 149.8; 153.1; 156.4; 156.6; 157.1; 171.7.
Found, %: C 45.25; H 2.31; Cl 17.98; N 14.02; S 8.18.
S. Calculated, %: C 45.36; H 2.54; Cl 17.85;
N 14.10; S 8.07.
,5-Dichloro-N'-{[(4,5-dichloroisothiazol-3-yl)carbonyl]-
oxy}isothiazole-3-carboxyimidamide (19). Yield 1.76 g
3-(4,5-Dichloroisothiazol-3-yl)-5-[5-(4-methylphenyl)-
isoxazol-3-yl]-1,2,4-oxadiazole (10). Yield 2.64 g (58%),
1
(
–
1
white powder, mp 226–228°С. IR spectrum, ν, cm : 3139,
3092, 3060, 2957, 2918, 2853, 1613, 1593, 1588, 1567,
1539, 1509, 1468, 1458, 1423, 1376, 1364, 1344, 1323,
1257, 1234, 1215, 1193, 1151, 1127, 1117, 1015, 998, 956,
15
C H
10Cl
N O
2 4 3
1
4
949, 940, 927, 805, 766, 678, 507. H NMR spectrum,
δ, ppm (J, Hz): 2.40 (3H, s, CH ); 7.42 (2Н, d, J = 8.0,
3
–
1
(
90%), white powder, mp 160–163°С. IR spectrum, ν, cm :
H Ar); 7.88 (1Н, s, СН isoxazole); 7.97 (2Н, d, J = 8.0,
3
1
482, 3307 (NH
2
), 3132, 1758 (C=O), 1624, 1572, 1481,
H Ar). Found, %: C 47.67; H 2.03; Cl 18.71; N 14.71;
452, 1445, 1365, 1345 (C–C, C=C, C=N), 1296, 1224,
204, 1168, 1092, 962, 913 (CCl), 864, 849, 724, 515.
S 8.45. C15
H
Cl
8 2
N
4
2
O S. Calculated, %: C 47.51; H 2.13;
1
Cl 18.70; N 14.77; S 8.45.
1
H NMR spectrum, δ, ppm: 5.63 (2Н, br. s, NH
2
).
3,5-Bis(4,5-dichloroisothiazol-3-yl)-1,2,4-oxadiazole (11).
Yield 4.17 g (93%), white powder, mp 176–176°C.
1
3
C NMR spectrum, δ, ppm: 123.4; 124.7; 149.8; 150.3;
–1
1
53.7; 154.5; 156.0; 157.4. Found, %: C 24.67; H 0.88;
IR spectrum, cm : 1594, 1546, 1468, 1439, 1393, 1379, 1344,
1273, 1254 (C–C, C=C, C=N), 1230, 1204, 1089, 1012,
928, 914 (C–Cl), 836, 776, 667, 528. 13C NMR spectrum,
δ, ppm: 123.4; 124.7; 149.8; 150.3; 153.7; 154.5; 165.3;
167.4. Found, %: C 25.88; Cl 37.73; N 15.07; S 17.03.
Cl 36.49; N 14.37; S 16.48. C
8
H
2
Cl
4
N
4
O
2
2
S . Calculated, %:
C 24.51; H 0.51; Cl 36.17; N 14.29; S 16.36.
Preparation of 3,5-substituted 1,2,4-oxadiazoles 7–11
(
General method). A suspension of the appropriate
carboxyimidamide 15–19 (12 mmol) in formic acid (20 ml)
was prepared and refluxed for 3 h. The reaction mixture
was then poured into water, the precipitate was filtered off,
washed with water, ether, and dried at reduced pressure.
The obtained 3,5-disubstituted 1,2,4-oxadiazoles 7–11 were
further used without additional purification.
C
8
Cl
4
N
4
OS . Calculated, %: C 25.69; Cl 37.91; N 14.98;
2
S 17.14.
Preparation of 2,5-substituted 1,3,4-oxadiazoles 20–23
(General method). A solution of 5-[5-(4-methylphenyl)-
1,2-oxazol-3-yl-2Н-tetrazole (3) or 5-(4,5-dichloro-1,2-thia-
zol-3-yl)-2Н-tetrazole (4) (1 mmol) and 1,2-azolyl-
3-carbonyl chloride 12–14 (1.03 mmol) was refluxed in
anhydrous toluene until the evolution of gas ceased (~8 h).
The precipitate was filtered off, washed with a dilute
3
,5-Bis[5-(4-methylphenyl)isoxazol-3-yl]-1,2,4-oxadiazole
(
7). Yield 4.29 g (93%), white powder, mp 234–236°С.
–
1
IR spectrum, ν, cm : 3138, 3058, 3031, 2921, 2855, 1615,
1
1
7
594, 1557, 1503, 1446, 1417, 1374, 1339, 1300, 1274,
235, 1186, 1155, 1111, 1041, 951, 939, 910, 823, 803,
solution of NaHCO , water, ethanol, dichloromethane, and
3
dried at reduced pressure. The obtained 2,5-isoxazolyl
(isothiazolyl)-substituted 1,3,4-oxadiazoles 20–23 were
further used without additional purification.
1
90, 762, 679, 503. H NMR spectrum, δ, ppm: 2.36 (3Н,
s, CH
3
); 2.40 (3Н, s, CH ); 7.38–7.46 (4Н, m, H Ar); 7.77
3
(
1Н, s, СН isoxazole); 7.78 (1Н, s, СН isoxazole); 7.91–7.96
2-[5-(4-Methylphenyl)isoxazol-3-yl]-5-(5-phenylisox-
azol-3-yl)-1,3,4-oxadiazole (20). Yield 0.34 g (92%),
white powder, mp 256–258°С (decomp.). IR spectrum,
(
C
4Н, m, H Ar). Found, %: C 68.88; H 4.51; N 14.63.
. Calculated, %: C 68.74; H 4.20; N 14.58.
-(4,5-Dichloroisothiazol-3-yl)-3-[5-(4-methylphenyl)-
isoxazol-3-yl]-1,2,4-oxadiazole (8). Yield 3.91 g (86%),
22
5
H
16
N
4
O
3
–
1
ν, cm : 3141, 3090, 3067, 3058, 2922, 2851, 1613, 1560,
1493, 1486, 1450, 1435, 1414, 1227, 1115, 1000, 950, 935,
–1
1
white powder, mp 196–197°С. IR spectrum, ν, cm : 3137,
813, 805, 767, 741, 691, 677. H NMR spectrum, δ, ppm
3
1
1
064, 3030, 2921, 2854, 1614, 1590, 1554, 1499, 1476,
457, 1421, 1349, 1338, 1300, 1274, 1228, 1209, 1138,
(J, Hz): 2.37 (3Н, s, CH ); 7.37 (2Н, d, J = 8.0, H Ar); 7.62
3
(1Н, s, СН isoxazole); 7.65 (1Н, c, СН isoxazole); 7.75–
1
13
039, 999, 927, 914, 828, 801, 767, 680, 505. H NMR
8.00 (5Н, m, H Ph); 8.22–8.36 (2Н, m, H Ar). C NMR
spectrum, δ, ppm (J, Hz): 2.38 (3Н, s, СH
3
); 7.38 (2Н, d,
spectrum, δ, ppm: 21.6 (CH ); 99.6 (СН isoxazole); 99.7
3
J = 7.4, H Ar); 7.65 (1Н, s, СН isoxazole); 7.90 (2Н, d,
(СН isoxazole); 123.9; 124.7; 126.5 (2СН Ar); 128.0 (2СН
Ar); 129.4 (2СН Ph); 130.4 (3СН Ph); 141.7; 145.6; 153.4;
153.5; 162.7; 171.8; 176.0. Found, %: C 68.30, H 3.91,
13
J = 7.4, H Ar). C NMR spectrum, δ, ppm: 21.7 (CH
9.8 (СН isoxazole); 123.9 (2C); 126.5 (2CН Ar); 130.5 (2CН
Ar); 141.8 (2C); 148.5; 151.9; 153.2; 162.0; 169.8. Found,
: C 47.79; H 2.40; Cl 18.91; N 14.61; S 8.69.
Cl S. Calculated, %: C 47.51; H 2.13; Cl 18.70;
N 14.77; S 8.45.
-(4,5-Dichloroisothiazol-3-yl)-5-(5-phenylisoxazol-3-yl)-
,2,4-oxadiazole (9). Yield 2.76 g (63%), white powder,
3
);
9
N 15.07. C21
N 15.13.
H
14
N
4
O . Calculated, %: C 68.10, H 3.81,
3
%
15
C
H
8
2
N
4
O
2
2-(4,5-Dichloroisothiazol-3-yl)-5-(5-phenylisoxazol-3-yl)-
1,3,4-oxadiazole (21). Yield 0.3 g (82%), white powder,
–1
3
mp 224°С. IR spectrum, ν, cm : 3144, 2922, 2853, 1615,
1561, 1498, 1477, 1447, 1427, 1401, 1348, 1199, 1122,
1070, 1037, 993, 949, 923, 844, 814, 768, 689, 679, 526.
1
–
1
mp 192–193°С. IR spectrum, ν, cm : 3146, 2928, 1620,
608, 1590, 1570, 1468, 1459, 1449, 1421, 1379, 1371,
343, 1258, 1233, 1149, 950, 940, 925, 905, 812, 766, 689,
1
1
1
H NMR spectrum, δ, ppm: 7.33 (1Н, s, СН isoxazole);
7.38–7.49 (3Н, m, H Ph); 7.63–7.74 (2Н, m, H Ph).
1
346

 Bumagin, Nikolay A.
Bumagin, Nikolay A.
 Petkevich, Sergey K.
Petkevich, Sergey K.
 Kletskov, Alexey V.
Kletskov, Alexey V.
 Potkin, Vladimir I.
Potkin, Vladimir I.