Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
Original Papers
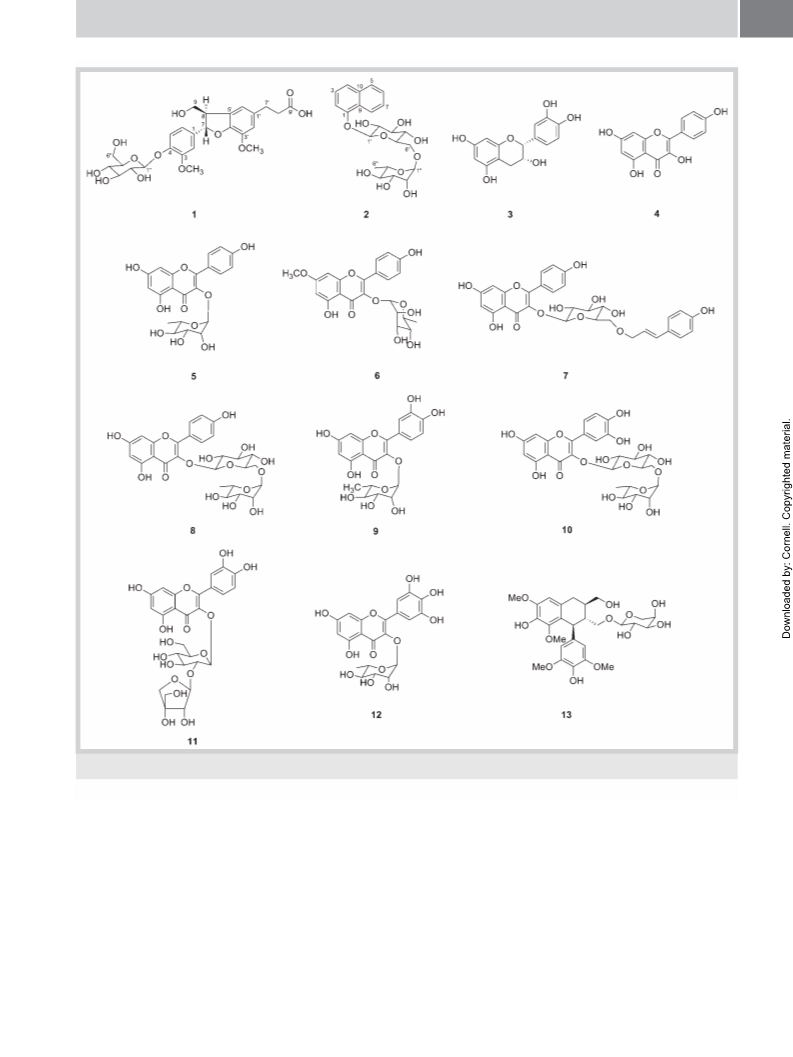
diabetes-related complications. Litsea japonica (Thunb.) Juss.
(Lauraceae) is an evergreen tree that grows in the southern parts
of Korea, Japan, and China. Previous phytochemical studies on
this plant resulted in the isolation of a number of essential oils,
fatty acids, alkaloids, lactones, anthocyanins, and terpenoids [9–
14]. Recently, it was reported that a crude extract from the leaves
of L. japonica induced apoptosis of HL-60 leukemia cells [15] and
significantly inhibited complement activity in vitro [16]. In our
ongoing efforts to identify effective, naturally sourced therapeu-
tic agents for diabetic complications, we found that an extract
from L. japonica reduced the development of diabetic nephropa-
thy via the inhibition of AGEs formation in db/db mice [17] and
prevented diabetes-induced lens opacification via the inhibition
of AR activity [18]. However, the active compounds underlying
these effects of L. japonica remain unknown. Further phytochem-
ical studies of this plant resulted in the isolation of two new (1
and 2) and 11 known compounds (3–13). In this report, we de-
scribe the isolation and structural elucidation of these com-
pounds, as well as the characterization of their inhibitory effects
on AGEs formation and RLAR activity. The effects of kaempferol
(4) and tiliroside (7) on the dilation of HG-induced hyaloid-reti-
nal vessels in larval zebrafish were also investigated.
for the resonance of a carboxyl group (δC 179.72) instead of a hy-
droxymethylene group at C-9′ in DA, suggesting that the dihydro-
coniferyl alcohol group of DA was replaced by a dihydroconi-
ferylic acid group in 1. The presence of a dihydroconiferylic acid
moiety in 1 was further confirmed by the 1H-1H COSY correla-
tions between H-7′ [δH 2.85 (2H, t, J = 7.7 Hz)] and H-8′ [δH 2.49
(2H, t, J = 7.8 Hz)], together with the HMBCs of both H-7′ and H-
"
8′ with a carboxyl carbon signal (δC 179.72; l Fig. 2). The attach-
ment of this moiety at C-1′ was deduced from the HMBC experi-
ment. The six carbon signals at δC 102.89, 78.32, 77.97, 75.04,
71.45, and 62.58 and an anomeric proton signal at δH 4.87 of 1
were typical of a glucose unit, which was identified as D-glucose,
followed by GC analysis of the acid hydrolysate. Moreover, the
large coupling constant (J = 7.5 Hz) of the anomeric proton indi-
cated that the glucose unit was linked in a β-configuration. The
location of a glucose unit at C-4 was elucidated from the HMBC
between the anomeric proton signal at δH 4.87 (H-1′′) and the
"
aglycone carbon signal at δC 147.73 (C-4; l Fig. 2). The large cou-
pling constant (J = 6.4 Hz) between H-7 and H-8 indicated the rel-
ative trans-vicinal coupling of the dihydrobenzofuran ring. This
was confirmed by the cross peaks observed between H-7 and H-
"
9b and between H-8 and H-6 in the NOESY spectrum (l Fig. 3).
The absolute configuration at C-7 and C-8 was determined to be
7S,8R from the circular dichroism spectrum of 1, showing a posi-
tive Cotton effect in the region of 270–290 nm, compared with
those reported for dihydrobenzofuran lignans [26,27]. Thus, the
structure of 1 was determined to be (7S,8R)-dihydrodehydrodi-
coniferylic acid 4-O-β-D-glucopyranoside.
Results and Discussion
!
An 80% ethanol extract of L. japonica leaves was suspended in
water and partitioned successively using n-hexane, EtOAc, and
n-BuOH. The EtOAc-soluble fraction that significantly inhibited
both AGEs formation (IC50 = 8.5 µg/mL) and RLAR activity
(IC50 = 2.3 µg/mL) was subjected to a series of chromatographic
techniques. This led to the isolation of two new (1 and 2) and 11
Compound 2 was obtained as a white powder with the molecular
formula C22H28O10, as established by HRESI‑MS, based on a mo-
lecular ion peak at m/z 453.1753 [M + H]+. Its UV absorption max-
ima (282, 315, 330 nm) suggested the presence of a naphthalene
nucleus [28], which was supported by the analysis of the 1H- and
"
known compounds (3–13; l Fig. 1). By comparing their physico-
13
"
chemical and spectral data with those in literature, the 11 known
compounds were identified as epicatechin (3) [16], kaempferol
(4) [19], afzelin (5) [16], rhamnocitrin-3-O-rhamnoside (6) [20],
tiliroside (7) [16], kaempferol 3-O-rutinoside (8) [21], quercitrin
(9) [16], rutin (10) [16], quercetin 3-O-β-D-apiofuranosyl-
(1 → 2)-β-D-glucopyranoside (11) [22], myricitrin (12) [16], and
lyoniside (13) [23].
C‑NMR spectra (l Table 2). The 13C‑NMR spectrum, together
with the DEPT data, showed that 2 contained 22 carbons, 10 of
which were consistent with those of a 1-naphthol unit [29]. The
12 remaining signals were assigned to glucose (δC 102.86, 78.38,
77.18, 75.22, 71.76, and 68.09) and rhamnose (δC 102.35, 74.17,
72.55, 72.33, 70.01, and 18.06) units, which were identified as
D-glucose and L-rhamnose, respectively, by GC analysis of the ac-
id hydrolysate. The β-configuration of the glucose unit was con-
firmed by the coupling constant of the anomeric proton at δH
5.04 (1H, d, J = 7.8 Hz), and the α-configuration of the rhamnose
unit was determined by the anomeric proton at δH 4.71 (1H, d,
J = 1.5 Hz). The HMBC correlation between H-1′′ (δH 4.71) and C-
6′ (δC 68.09) indicated a rhamnopyranosyl-(1 → 6)-glucopyrano-
Compound 1 was obtained as a white amorphous powder with a
negative specific rotation, [α]2D5 − 43.0 (c 0.1, MeOH). HRESI‑MS
analysis of 1 yielded a molecular ion peak at m/z 537.1964
[M + H]+, in accordance with the molecular formula C26H32O12
.
The UV absorption maxima of 1 in MeOH observed at 276 nm
suggested the presence of aromatic ring(s). Acid hydrolysis of 1
yielded an aglycone and a monosaccharide unit. The 1H NMR
"
syl linkage (l Fig. 2). Furthermore, the attachment of the sugar
"
spectrum (l Table 1) of the aglycone unit displayed five aromatic
chain at C-1 on the 1-naphthol structure was deduced from the
protons, including two meta-coupled doublets [δH 6.77 (1H, d,
J = 2.0 Hz) and 6.73 (1H, d, J = 2.0 Hz)] and ABX aromatic system
protons [δH 7.13 (1H, d, J = 8.4 Hz), 7.02 (1H, d, J = 2.0 Hz), and
6.92 (1H, dd, J = 8.0, 2.0 Hz)], a pair of hydroxymethylene protons
[δH 3.74 (1H, dd, J = 11.0, 7.5 Hz) and 3.70 (1H, dd, J = 11.0,
4.0 Hz)], and two methoxyl protons [δH 3.82 and 3.86 (each 3H,
s)], in addition to aliphatic peaks corresponding to a dihydroconi-
ferylic acid side chain and a dihydrobenzofuran ring. These spec-
troscopic data suggested that 1 is a dihydrobenzofuran-type neo-
lignan, after comparing with reported analogs [24,25]. This pro-
posed skeleton was supported by the 13C‑NMR spectrum
HMBC spectrum, showing HMBC correlations between H-1′ (δH
5.04) and the aglycone carbon C-1 (δC 154.73; l Fig. 2). Thus,
the glycoside linkage was identified as α-L-rhamnopyranosyl-
(1 → 6)-O-β-D-glucopyranosyl located at C-1 of the aglycone.
Finally, the structure of 2 was established as 1-naphthol 1-O-α-
L-rhamnopyranosyl-(1 → 6)-O-β-D-glucopyranoside.
"
Previously, we reported that some phenolic compounds, such as
afzelin (5), quercitrin (9), and rutin (10), exhibit considerable in-
hibitory effects on AGEs formation and RLAR [30]. In this study,
we further investigated the inhibitory effects of isolated com-
pounds, apart from the previously evaluated compounds, on
"
"
(l Table 1), which revealed two carbon signals at δC 88.63 (C-7)
AGEs formation and RLAR activity in vitro (l Table 3). Of the
and 55.85 (C-8) characteristic of a dihydrobenzofuran neolignan.
These spectroscopic data were similar to those for DA [24], except
compounds evaluated, flavonoids, epicatechin (3), kaempferol
(4), rhamnocitrin-3-O-rhamnoside (6), tiliroside (7), kaempferol
Lee IS et al. Flavonoids from Litsea… Planta Med

 Lee, Ik-Soo
Lee, Ik-Soo
 Kim, Yu Jin
Kim, Yu Jin
 Jung, Seung-Hyun
Jung, Seung-Hyun
 Kim, Joo-Hwan
Kim, Joo-Hwan
 Kim, Jin Sook
Kim, Jin Sook