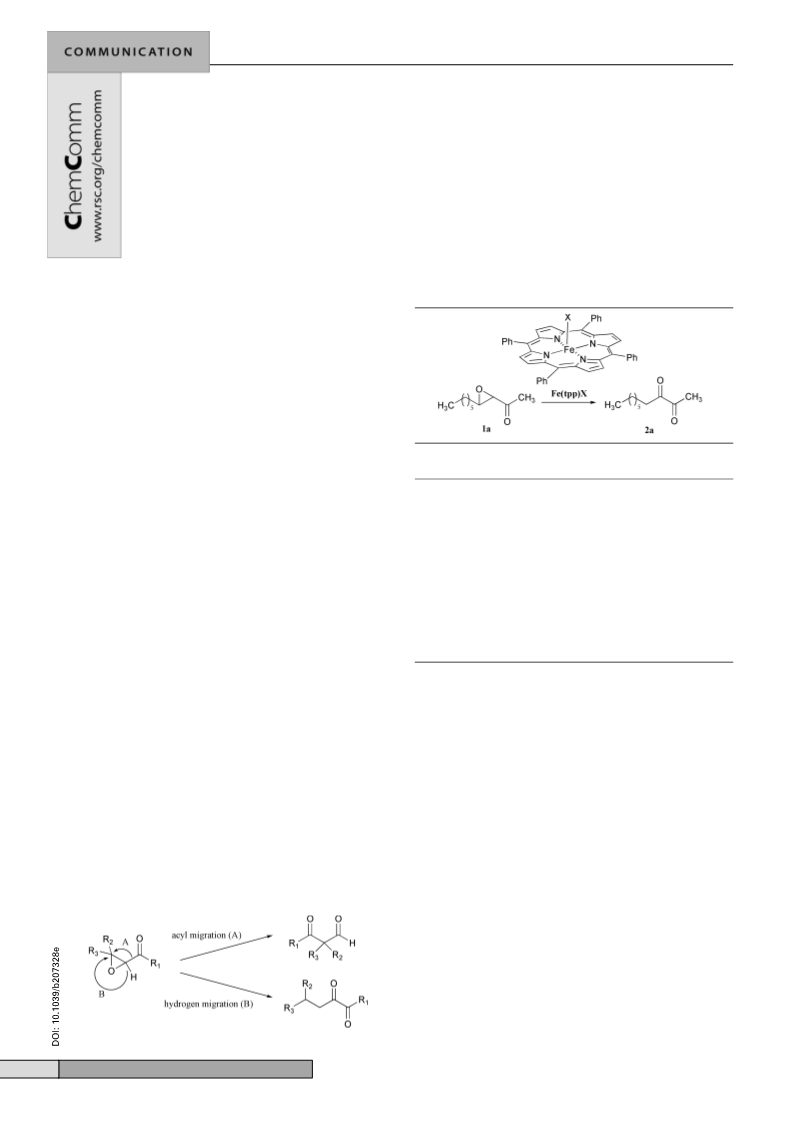
catalyzed reaction proved to be general and applicable to a
variety of a,b-epoxy ketones.‡ As a whole, a facile and highly
selective conversion of epoxy ketones into 1,2-diketones via
hydrogen migration, viz., route B in Scheme 1, was observed.
Acetylepoxides 1b bearing an alkyl group longer than that of 1a
were easily transformed into the corresponding 1,2-diketone 2b
in 88% yield. In the rearrangement of other acetylepoxides 1c,
1d and 1e, olefinic, cycloalkanoic and aromatic moieties in the
substrates were tolerated well, and the corresponding 1,2-dike-
tones 2c, 2d and 2e were obtained in high yields. Both the yield
and the reaction time are independent of the carbon-chain length
of acyl substituents in the substrates: the reaction of non-
anoylepoxide 1f proceeded smoothly, giving the corresponding
1,2-diketone 2f as a sole isolable product. Phenyl epoxides 1g
and 1h were surprisingly susceptible to the catalytic rearrange-
ment. The rearrangements of these epoxides were completed
within 15 min under the same conditions, affording 1,2-dike-
tones 2g and 2h in high yields. This acceleration in reaction rate
should be ascribed to the presence of the b-phenyl group that
can stabilize the carbonium ion generated by the C–O bond
cleavage at the b-position in the rearrangement.
Notes and references
† Fe(tpp)ClO4 and Fe(tpp)OTf were prepared according to the method
described previously, see ref. 8.
‡ The products 2a–2f gave satisfactory NMR, IR and high-resolution mass
spectra. For example, 1H- and 13C-NMR data of 2a and 2e are as follows.
2a: 1H-NMR (270 MHz, CDCl3) d 0.88 (3H, t, J = 6.6 Hz), 1.18–1.35 (8H,
m), 1.58 (2H, m), 2.33 (3H, s), 2.73 (2H, t, J = 7.4 Hz); 13C-NMR (67.8
MHz, CDCl3) d 14.1, 22.6, 23.1, 23.7, 29.0, 29.1, 31.6, 35.7, 197.6, 199.5.
2e: 1H-NMR (270 MHz, CDCl3) d 1.93 (2H, m), 2.29 (3H, s), 2.64 (2H, t,
J = 7.6 Hz), 2.75 (2H, t, J = 7.3 Hz), 7.15–7.31 (m, 5H); 13C-NMR (67.8
MHz, CDCl3) d 23.7, 24.6, 35.0, 126.1, 128.4, 128.5, 141.3, 197.4, 199.1.
The products 2g10a and 2h6a are compared with the literature data.
1 For a recent review, see: C. Lauret, Tetrahedron: Asymmetry, 2001, 12,
2359.
2 H. O. House, J. Am. Chem. Soc., 1954, 76, 1235; H. O. House and G. D.
Ryerson, J. Am. Chem. Soc., 1961, 83, 979 and references cited
therein.
3 R. D. Bach and J. M. Domagala, J. Org. Chem., 1984, 49, 4181; R. D.
Bach and R. C. Klix, J. Org. Chem., 1985, 50, 5440; F. Kunisch, K.
Horbert and P. Welzel, Tetrahedron Lett., 1985, 26, 6039; R. D. Bach
and R. C. Klix, Tetrahedron Lett., 1985, 26, 985; R. C. Klix and R. D.
Bach, J. Org. Chem., 1987, 52, 580; K. Okada, T. Katsura, H. Tanino,
H. Kakoi and S. Inoue, Chem. Lett., 1994, 157; S. Sankararaman and J.
E. Nesakumar, J. Chem. Soc., Perkin Trans. 1, 1999, 3173; J. A. Elings,
H. E. B. Lempers and R. A. Sheldon, Eur. J. Org. Chem., 2000,
1950.
4 Photo irradiation and a Pd(0) catalyst are also effective for the selective
rearrangement of epoxy ketones into 1,3-diketones: S. P. Pappas, R. M.
Gresham and M. J. Miller, J. Am. Chem. Soc., 1970, 92, 5797; M.
Suzuki, A. Watanabe and R. Noyori, J. Am. Chem. Soc., 1980, 102,
2095.
5 K. Okada, K. Murakami and H. Tanino, Tetrahedron, 1997, 53, 14247;
K. Okada, K. Murakami, H. Tanino, H. Kakoi and S. Inoue,
Heterocycles, 1996, 43, 1735; M. Asaoka, S. Hayashibe, S. Sonoda and
H. Takei, Tetrahedron, 1991, 47, 6967; V. Enev and E. Tsankova,
Tetrahedron Lett., 1988, 29, 1829.
6 Although silica gel and AlCl3 mediated rearrangements of chalcone
epoxides into 1,3-diarylpropane-1,2-diones have been reported, the
substrates exemplified in these reports are strictly restricted to chalcone
epoxide derivatives: (a) T. B. Rao and J. M. Rao, Synth. Commun.,
1993, 23, 1527; (b) O. N. Bubel, I. G. Tishchenko and O. A. Grinkevich,
Khim. Geterotsikl. Soedin., 1985, 1624.
In summary, we have developed the first catalytic version of
the Lewis acid mediated rearrangement of a,b-epoxy ketones
into 1,2-diketones. The rearrangement can easily be promoted
by high-valent metalloporphyrin complexes, especially Fe(tp-
p)OTf, at a low catalyst loading (2 mol%) under very mild
conditions. 1,2-Diketones have been found to be versatile
intermediates in organic synthesis, and their preparation has
been widely investigated.10 As for 1,2-diketone syntheses, the
present catalytic process exceeds or competes with other
methods reported so far in view of the simplicity, high yields,
and availability of starting materials.11 Elucidation of the
precise mechanism and the synthetic applications of the present
metalloporphyrin catalyzed rearrangement are in progress.
We thank Mr N. Eguchi, Miss T. Koseki and Miss A. Ohmae
in the Analytical Center of our university for measurements of
NMR and mass spectral data. This research was supported in
part by a Grant-in-Aid for Scientific Research (No. 11119267)
from the Ministry of Education, Culture, Sports, Science and
Technology, Japan and The Science Research Promotion Fund
from Bio Venture Research Center of Meiji Pharmaceutical
University.
7 Sosnovskii et al. have reported that (iPrO)2TiCl2 can promote the
rearrangement. This procedure, however, requires more than a stoichio-
metric amount of (iPrO)2TiCl2: G. M. Sosnovskii and I. V. Astapovich,
Zh. Org. Khim., 1993, 29, 85.
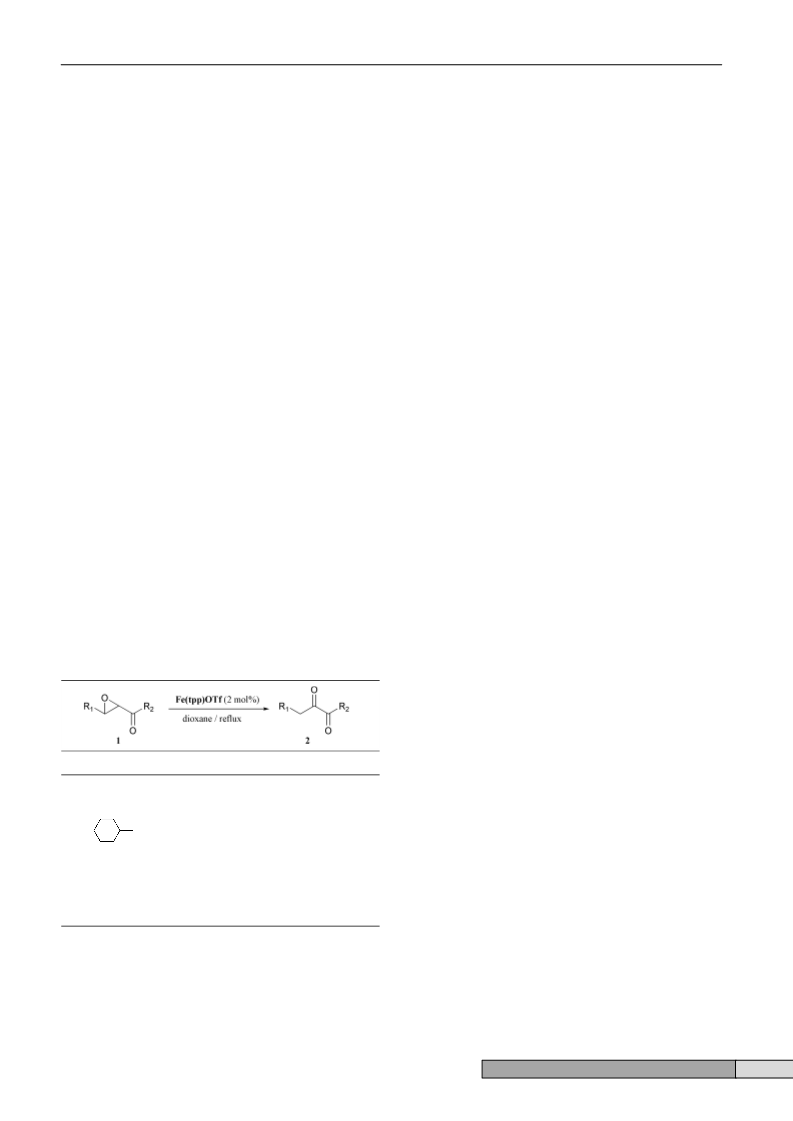
Table 2 Fe(tpp)OTf catalyzed rearrangement of a,b-epoxy ketonesa
8
K Suda, K. Baba, S. Nakajima and T. Takanami, Tetrahedron Lett.,
1999, 40, 7243; T. Takanami, M. Ueno, F. Hino and K. Suda, Chem.
Lett., 1996, 1031.
9 Maruoka et al. have reported that exceptionally bulky, oxygenophilic
organoaluminum reagent, MABR, is among the most effective reagent
for the regio- and stereoselective rearrangement of epoxides into
carbonyl compounds: K. Maruoka, T. Ooi and H. Yamamoto, J. Am.
Chem. Soc., 1989, 111, 6431; K. Maruoka, J. Sato and H. Yamamoto, J.
Am. Chem. Soc., 1991, 113, 5449.
R1
R2
Time
Yield (%)b
10 (a) Z. X. Si, X. Y. Jiao and B. F. Hu, Synthesis, 1990, 509; (b) D.
Seyferth, R. M. Weinstein, R. C. Hui, W. L. Wang and C. M. Archer, J.
Org. Chem., 1991, 56, 5768; (c) F. Babudri, V. Fiandanese, G. Marchese
and A. Punzi, Tetrahedron Lett., 1995, 36, 7305; (d) A. R. Katritzky, Z.
Wang, H. Lang and D. Feng, J. Org. Chem., 1997, 62, 4125; (e) H.
Sakurai, K. Tanabe and K. Narasaka, Chem. Lett., 2000, 168; (f) S.
Antoniotti and E. Duñach, Chem. Commun., 2001, 2566; (g) M. S.
Yusubov, G. A. Zholobova, S. F. Vasilevsky, E. V. Tretyakov and D. W.
Knight, Tetrahedron, 2002, 58, 1607 and references cited therein.
11 Many good methods have been available for the preparation of a,b-
epoxy ketones. For recent reviews, see: M. J. Porter and J. Skidmore,
Chem. Commun., 2000, 1215; T. Nemoto, T. Ohshima and M.
Shibasaki, J. Synth. Org. Chem. Jpn., 2000, 60, 94.
a
b
c
CH3(CH2)5-
CH3(CH2)8-
CH2NCH(CH2)8-
CH3-
CH3-
CH3-
CH3-
1.5 h
1.5 h
2 h
95
88
92
87
d
2.5 h
e
f
g
h
Ph(CH2)2-
CH3(CH2)2-
Ph-
CH3-
CH3(CH2)7-
CH3-
1.5 h
1 h
15 m
15 m
87
91
85
85
Ph-
Ph-
a Conditions: 1 (0.5 mmol), Fe(tpp)OTf (2 mol%), dioxane (3 ml), reflux.
b Isolated yield.
CHEM. COMMUN., 2002, 2570–2571
2571

 Suda, Kohji
Suda, Kohji
 Baba, Kenji
Baba, Kenji
 Nakajima, Shin-Ichiro
Nakajima, Shin-Ichiro
 Takanami, Toshikatsu
Takanami, Toshikatsu