Table 2. Reactions of 1 with sodium or potassium methoxides and sodium ethoxidea
isolated
yield of
2, %
substrate
alkoxylating
agent
reaction
time, h
conversion
of 1, %
selectivity for
2 or 7, %
1
solvent
1a
1a
1a
1b
1c
1d
1e
1a
1b
NaOMe solid
KOMe - in situ
NaOMe - in situ
NaOMe solid
NaOMe solid
NaOMe solid
NaOMe solid
NaOEt solid
n-heptane
n-hexane
toluene
cyclohexane
n-hexane
toluene
toluene
n-hexane
toluene
3.0
2.5
6
2.5
3.0
2.5
24
96.4
100
76
97.8
100
86
52
89.8
93.6
94
97
71
95.3
97.2
12
85
50.5
63.6
82
84
-
81
85
-
-
3.0
1.5
-
NaOEt solid
-
a All reactions were carried out under aerobic conditions with Bu4N+Br- as a PTC.
Scheme 2
Thus, 3-benzyloxybromo- or chlorobenzenes were ob-
tained in yields close to theoretical by treating 1a,b with
benzyl alcohol-potassium hydroxide (KOH) in tetramethy-
lurea, in the presence of a PTC, at 50 °C, for 24 h.8 However,
no information was found in this article, or in other
publications, on the reactions between methoxides and meta-
substituted nitrobenzenes, 1a-e.
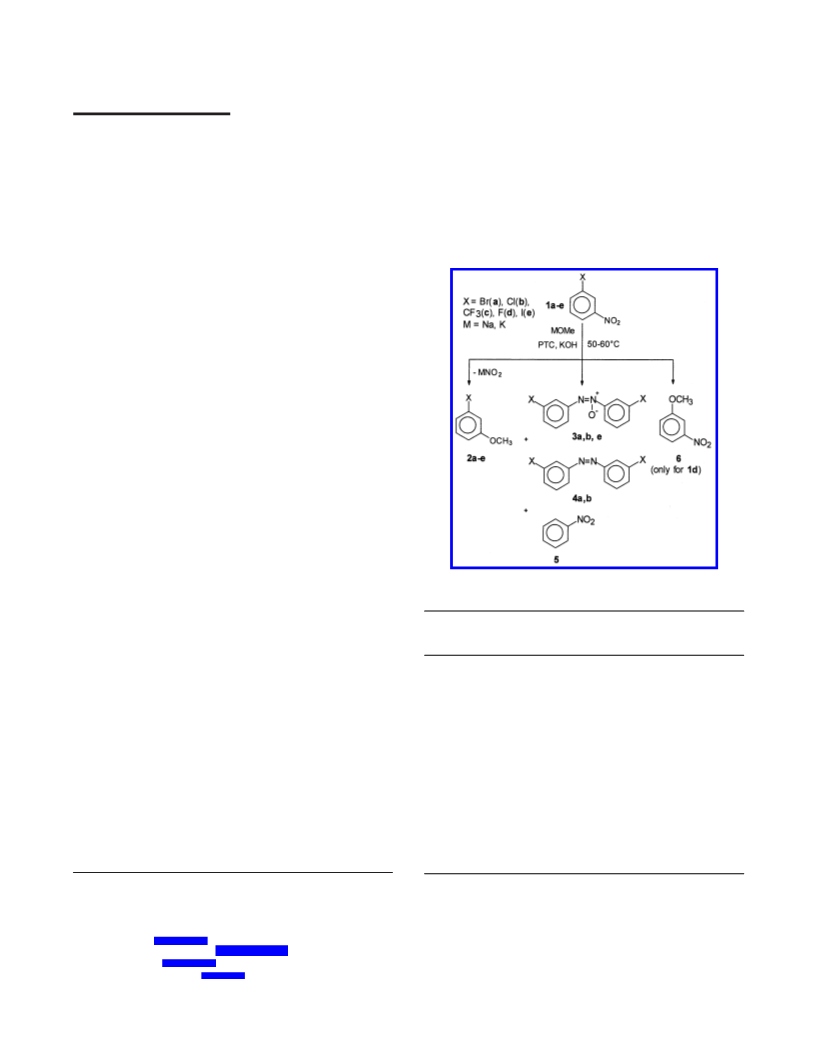
In this work, methoxydenitration is observed in the
reaction of 1 with sodium methoxide (NaOMe) or potassium
methoxide (KOMe), employing an effective amount of a
PTC, selected from quaternary ammonium salts, in the
medium of a water-immiscible nonpolar aprotic solvent, such
as cyclohexane, hexane, heptane, and toluene. No reaction
was observed in dichloromethane. It is generally known that
PTC reactions proceed readily in apolar aprotic solvents. It
can be assumed that the absence of any reaction in dichlo-
romethane is due to its relatively high polarity in comparison
with that of toluene, hexane, or cyclohexane (δ ) 8.9, 2.4,
The second approach to carrying out the methoxydeni-
tration employs KOMe, prepared in situ from methanol and
KOH. The amounts of methanol and KOH are 1.2 and 2.2-
2.4 mol, respectively, per 1 mol of substituted nitrobenzene,
1. The reaction is carried out at a temperature of 55-60 °C
for about 3 h. It should be noted that NaOMe prepared in
situ in the course of the reaction is significantly less reactive
than KOMe obtained in a similar way (Table 2).
In both approaches, the amount of PTC employed is in
the range of 20-30% w/w, based on the initial substituted
nitrobenzene, 1. The reaction proved to be very sensitive to
the type of PTC. As seen from Table 1, tributylmethylam-
monium chloride and all the tetrabutylammonium salts,
regardless of the anion, afforded good results. Neither
hydrophilic tetraethylammonium and benzyltrimethylammo-
nium bromides nor lipophilic, but bulky, tetraoctylammonium
bromide provided satisfactory results. Use of poly(ethylene
glycol) (PEG-1500) as a PTC afforded full consumption of
1a, but the selectivity for 2a over byproducts was low.
The anisoles, 2a-c, were isolated in a yield of more than
80% and a purity of more than 99% by fractional distillation
of the final reaction mixtures (Table 2).
The reactivity of the substituted nitrobenzene, 1e, towards
methoxides turned out to be considerably lower than that of
1a-c (Table 2). Even with a 4-fold molar excess of NaOMe,
the conversion of 1e was below 85%. In the case of
nitrobenzene, 1d, methoxydefluorination prevailed over
methoxydenitration. The main reaction product was m-
nitroanisole, 6, and not the targeted anisole, 2d (Table 2).
We also attempted the preparation of 3-halophenetoles,
7a,b, by a similar route (Scheme 2). For this purpose, 1a,b
were reacted with solid sodium ethoxide (NaOEt) under the
same conditions as used for the reactions with methoxide.
The reactions with NaOEt were characterized by a low
selectivity for the phenetoles, 7a,b (Table 2), while the side
9,10
1.9, and 2.0 respectively).
The methoxydenitration of 1a,b,e is accompanied by
radical reactions leading to the formation of byproducts of
mainly nitro reduction to the corresponding 3,3′-disubstituted
azoxy- and azobenzenes (compounds 3a,b,e and 4a-b,
respectively), and hydrodehalogenation to nitrobenzene, 5.
Therefore, in a nitrogen atmosphere, the selectivity for
anisoles, 2a,b, over byproducts 3, 4, and 5, does not exceed
80-85% (Table 1). To suppress the undesirable radical
processes, the methoxydenitration was performed under
aerobic conditions. As a result, the selectivity for 2a,b
increased to 95-97%.
Two different approaches to using the alkali metal
methoxide are suggested. The first approach consists of
carrying out the reaction using a pre-prepared solid NaOMe
or KOMe, in the presence of an effective amount of KOH.
The amount of methoxide used is 1.1-1.4 mol, based on 1
mol of substituted nitrobenzene, 1. The amount of KOH is
1.5-1.7 mol per 1 mol of 1. The reaction is carried out at a
temperature of 50-55 °C for 2-4 h.
(7) Arca, V.; Paradisi, C.; Scorrano, G. J. Org. Chem. 1990, 55, 3617.
(8) Effenberger, F.; Koch, M.; Streicher, W. Chem. Ber. 1991, 124, 163.
(9) Dehmlow, E. V.; Dehmlow, S. S. Phase Transfer Catalysis; Verlag Chemie
GmbH: Weinheim, 1983.
(10) Weissberger, A.; Proskauer, E. S.; Riddick, J. A.; Toops, E. E. Organic
SolVents. Physical Properties and Methods of Purification; Interscience
Publishers: New York, 1955.
304
•
Vol. 7, No. 3, 2003 / Organic Process Research & Development

 Zilberman, Joseph
Zilberman, Joseph