5
16
J. L. GRENIER ET AL.
4
4
1
(
(
H, J = 1.7 Hz, H2), 8.09 (d, 1H, J = 1.7 Hz, H6), 9.85
C H NO , calculate C 48.01, H 3.13, N 6.22, O 42.64;
9 7 6
Á
s, 1H, CHO); EIMS (60 eV), m/z 197 (100%, M ), 151
38%, [M-NO ] ).
found, C 48.00, H 3.18, N 6.41, O 42.41%; m.p. 240°C
Á
1
3
(decomp); H NMR (DMSO-d ), 6.22 (d, J = 15.7 Hz,
2
6
3
Hb), 7.09 (s, H2), 7.51 (s, H5), 7.93 (d, J = 15.7 Hz, Ha);
C NMR, ꢁ 167.1 (COOH), 151.3 (C3), 146.9 (C4),
13
2
- and 5-nitroferulic acids. Nitrovanillin (3 g) was
dissolved in 15 ml of pyridine and 10 drops of piperidine,
then 3.14 g of malonic acid were added. After complete
dissolution, the solution was kept for 6 days at room
temperature and then heated at 100°C for 4 h. The
solution was cooled and poured into a mixture of crushed
ice (200 g) and concentrated HCl (100 ml). The
precipitate was filtered, washed with ice-cold water and
dried in presence of P O to give 2- or 5-nitroferulic acid.
140.2 (Ca), 139.6 (C6), 123.0 (C1), 121.0 (Cb), 114.5
(C5), 111.0 (C2); EIMS (60 eV), 225 (11%), 208 (6%),
179 (100%).
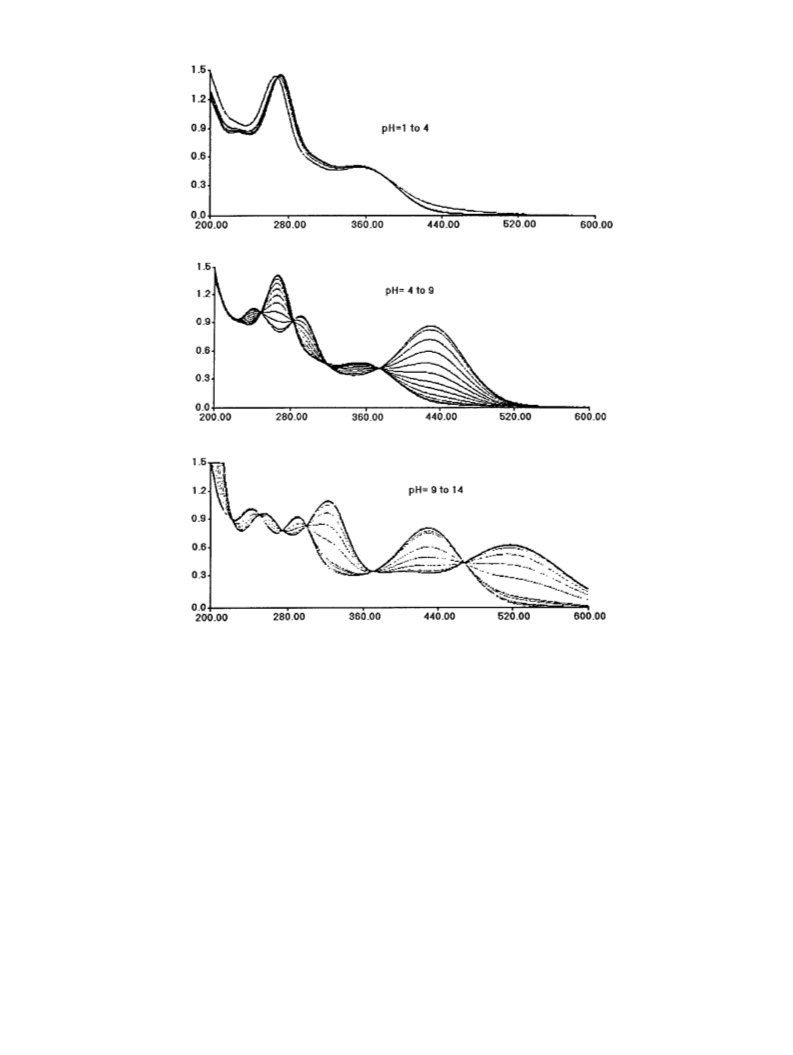
pK measurement. The pK s of the nitro compounds
a
a
were measured in water at 25°C spectrophotometrically
on a Perkin-Elmer Lambda 15 UV–visible spectrometer.
The initial concentration of the solution of NCA was
100 mM. Starting from pH = 1, the pH was adjusted by
adding 50 ml of 1 M NaOH (generally the total volume of
NaOH did not exceed 2 ml for an initial volume of
100 ml). All the manipulations were carried out under an
N2 atmosphere in order to minimize the possible
2
5
2
-Nitroferulic acid (77% yield): m.p. 200–205°C (de-
1
comp.); H NMR (DMSO-d ), ꢁ3.82 (s, 3H, OCH ), 6.48
6
3
3
3
(d, 1H, J = 15.7 Hz, Hb), 7.12 (d, 1H, J = 15.7 Hz, Ha),
3
3
7
.14 (d, 1H, J = 8.7 Hz, H6), 7.64 (d, 1H, J = 8.7 Hz,
Á
H5); EIMS (60 eV), m/z 239 (31%, M ), 193 (90%, [M-
NO ] ), 65 (100%). 5-Nitroferulic acid (75% yield):
m.p. 245°C (decomp.) (lit. 149°C); H NMR (DMSO-
Á
autoxidation of NCA under basic conditions. pK s were
2
a
2
5
1
measured using the Henderson–Hasselbach relation-
3
12
d ), ꢁ3.92 (s, 3H, OCH ), 6.64 (d, 1H, J = 16.0 Hz, Hb),
ship
log(ABH� A)/(A� AB
�
) = pH � pK , where A
6
3
a
BH
3
4
7
.54 (d, 1H, J = 16.0 Hz, Ha), 7.62 (d, 1H, J = 1.7 Hz,
and AB represent the absorbances of the species at
�
4
H2), 7.75 (d, 1H, J = 1.7 Hz, H6); EIMS (60 eV), m/z
acidic and at basic pH, respectively, and A is the
Á
239 (96%, M ), 51 (100%).
measured absorbance. pK s are reported in Table 1.
a
Nitrocaffeic acids. To a suspension of 1 g of 2- or 5-
nitroferulic acid or 1.055 g of 3,4-dimethoxy-6-nitrocin-
namic acid in 50 ml of dichloromethane were added
dropwise at room temperature under a nitrogen atmo-
ESR spectroscopy. ESR spectra were recorded using a
Varian E-109 spectrometer operating at 9.5 GHz with a
100 kHz high-frequency modulation amplitude ranging
from 0.1 to 0.4 G. The sample solutions were examined in
a flat quartz cell inserted in an E-238 cavity operating in
the TM110 mode for the ESR spectra recorded at room
temperature and in a quartz tube inserted in an E-232
cavity operating in the TE104 mode for the ESR spectra
recorded at 77 K.
sphere 20 ml of BBr (20 mmol) in dichloromethane. The
3
mixture was refluxed overnight. To the cooled solution
were added 200 ml of water. After 1 h of hydrolysis, the
organic layer was separated and the aqueous layer
extracted with ethyl acetate (5 Â 50 ml). The organic
layers were collected and refluxed with charcoal for
Nitroaromatic anion radicals were obtained sponta-
neously from a mixture of sodium borohydride (30 mg)
and the nitro compound (1 mg) in distilled water (0.5 ml).
The resulting pH was 8.8 unless indicated otherwise. The
initially yellowish solution turned orange–red after a few
seconds and the ESR spectra were immediately recorded.
Spectral simulations were carried out using a computer
0.5 h. The suspension was filtered over Celite and dried
over MgSO . After evaporation of the solvent the residue
4
was precipitated in a mixture of acetone and water to give
the nitrocaffeic acid. 2-Nitrocaffeic acid (80% yield):
elemental analyses for C H NO , calculated C 48.01, H
9
7
6
3
4
.13, N 6.22, O 42.64; found C 48.14, H 3.21, N 6.34, O
1
29
2.31%; m.p. 220°C (decomp.); H NMR (DMSO-d ),
program.
6
3
3
ꢁ
6.41 (d, J = 15.5 Hz, Hb), 6.97 (d, J = 8.0 Hz, H6),
Superoxide anions were produce from potassium
peroxide in DMSO (0.5 mM KO2 was dissolved in
DMSO in the presence of 5 mM 18-C-6). The nitro
compound was then added and the temperature was
immediately lowered to 77 K. The ESR spectra were
immediately recorded. Attempts to characterize semi-
quinonic or nitroaromatic radicals were made using the
same solution at room temperature.
3
3
13
7
.15 (d, J = 15.5 Hz, Ha), 7.33 (d, J = 8.0 Hz, H5);
C
NMR, ꢁ167.3 (COOH), 146.5 (C4), 144.2 (C3), 141.6
Ca), 138.1 (C2), 122.8 (C1), 120.0 (Cb), 117.2 (C5),
16.3 (C6); EIMS (60 eV), m/z 225 (69%), 179 (56%), 51
100%). 5-Nitrocaffeic acid (80% yield): elemental
analyses for C H NO , calculated C 48.01, H 3.13, N
(
1
(
9
7
6
6
.22, O 42.64; found C 48.08, H 3.15, N 6.07, O 42.70%;
1
m.p. 210°C (decomp.); H NMR (DMSO-d ), ꢁ 6.41 (d,
6
3
3
4
J = 16.0 Hz, Hb), 7.34 (d, J = 2.0 Hz, H6), 7.48 (d,
4
13
J = 16.0 Hz, Ha), 7.66 (d, J = 2.0 Hz, H2); C NMR, ꢁ
67.0 (COOH), 147.0 (C3), 143.8 (C4), 142.0 (Ca), 137.0
C5), 124.9 (C1), 118.0 (Cb), 117.0 (C2), 115.0 (C6);
1
(
REFERENCES
EIMS (60 V), m/z 225 (90%), 179 (17%), 51 (100%). 6-
Nitrocaffeic acid (85% yield); elemental analyses for
1
. Shahidi F, Naczk M. Food Phenolics. Sources, Chemistry, Effects,
Applications. Technomic: Lancaster, PA, 1995.
Copyright 2000 John Wiley & Sons, Ltd.
J. Phys. Org. Chem. 2000; 13: 511–517

 Grenier
Grenier
 Cotelle
Cotelle
 Catteau
Catteau
 Cotelle
Cotelle