10.1002/cctc.201801165
ChemCatChem
FULL PAPER
[10] a) Y.-F. Li, M.-Q. Guo, S.-F. Yin, L. Chen, Y.-B. Zhou, R.-H. Qiu, C.-T. Au,
Carbon, 2013, 55, 269–275; b) Y. Nabae, H. Rokubuichi, M. Mikumi, Y.
B. Kuang, T. Hayakawa, M.-aki Kakimoto, ACS catal. 2013, 3, 230–
236; c) S.-Y. Chen, X.-T. Zhou, J.-X. Wang, R.-C. Luo, Q.-J. Luo, L.-J.
Yu, H.-B. Ji, J. Mol. Catal. 2017, 438, 152–158; d) X. Zhang, H. Yang,
G. Yang, S. Li, X. Wang, J. Ma, ACS Sustainable Chem. Eng. 2018, 6,
5868–5876.
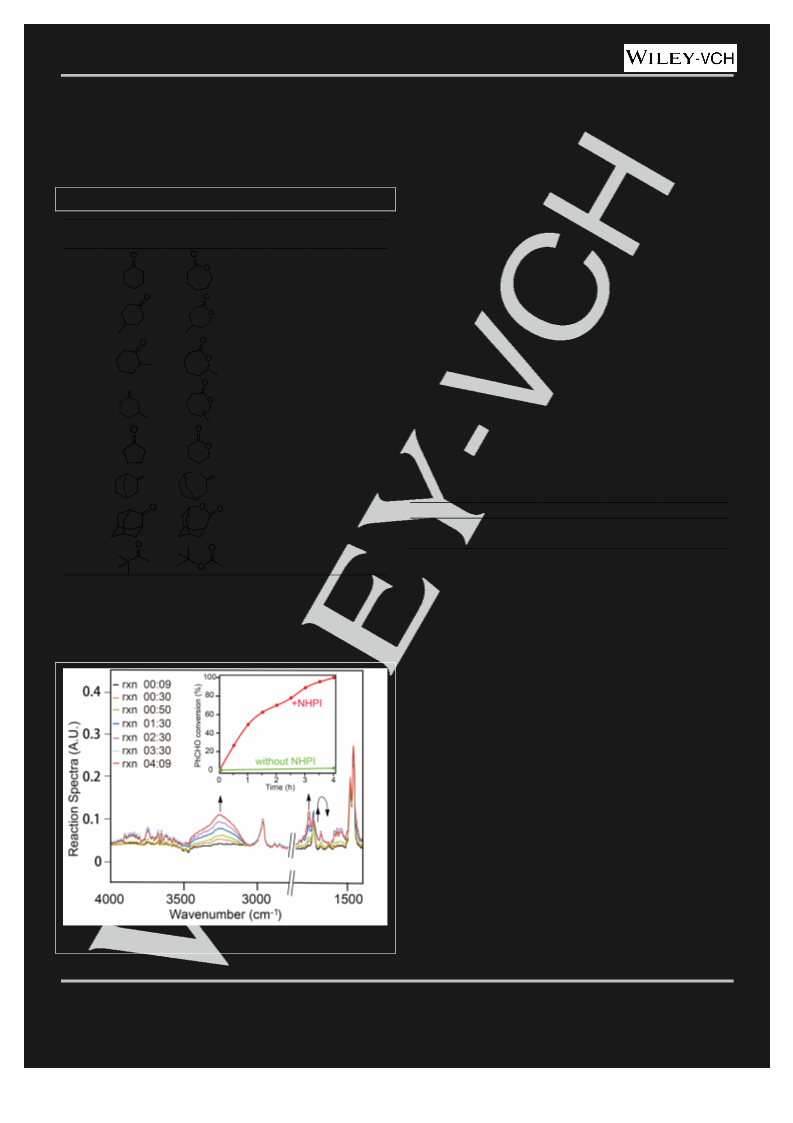
mmol) of benzaldehyde, 0.1962 g (2 mmol) of cyclohexanone and 20 mL
of 1,2-dichloroethane (DCE). The mixture was stirred at 40ºC at
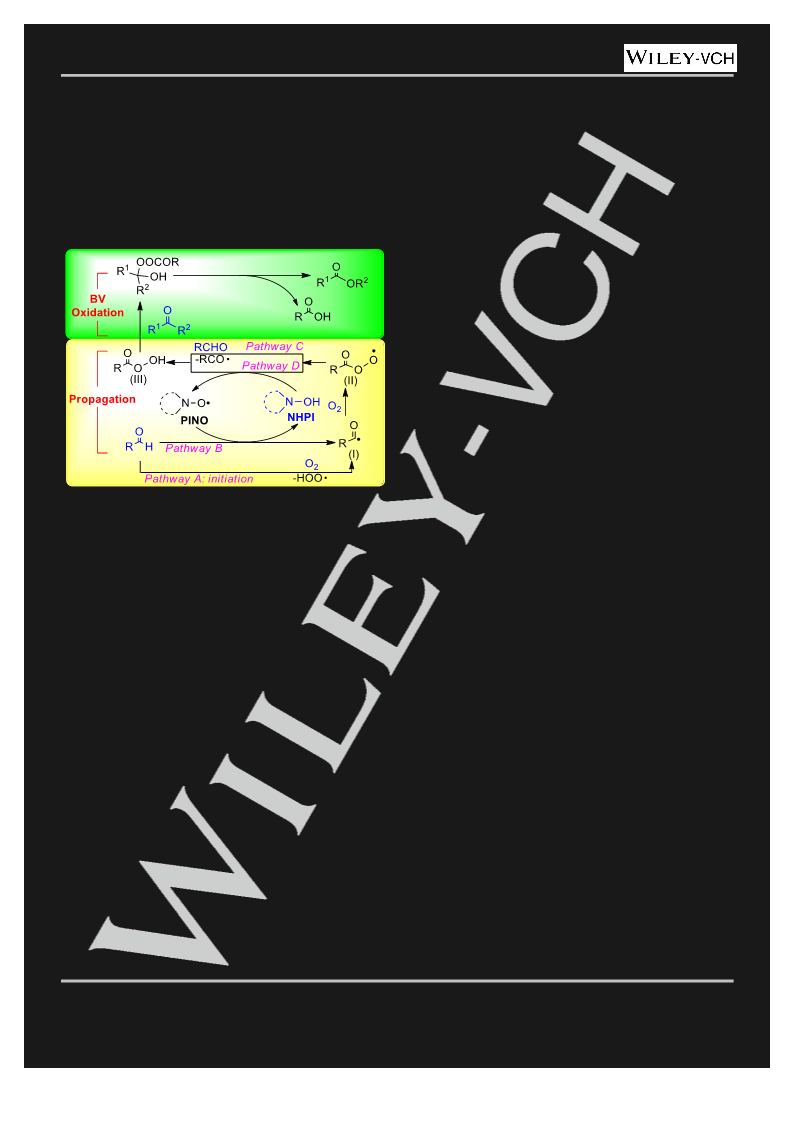
atmospheric pressure. After the typical BV oxidation reacted under the
general condition for an hour, 10 mol% of TEMPO was added to the
system. EPR spectrum were obtained by the X-band (9.5 GHz) EPR
spectrometer (Bruker A300) immediately (Figure 3, black), at t=1 h (red)
and t=2.5 h (blue).
[11] a) K. Kaneda, S. Ueno, T. Imanaka, E. Shimotsuma, Y. Nishiyama, Y.
Ishii, J. Org. Chem. 1994, 11, 2915–2917; b) H. J. Xu, F. F. Zhu, Y. Y.
Shen, X. Wan, Y. S. Feng, Tetrahedron, 2012, 68, 4145-4151.
[12] a) Y. Ishii, S. Sakaguchi, Catal. Today, 2006, 117, 105-113; (b) L. Melone,
C. Punta, Beilstein J. Org. Chem. 2013, 9, 1296–1310; c) Y. F. Liang, X.
Li, X. Wang, Y. Yan, P. Feng, N. Jiao, ACS Catal. 2015, 5, 1956-1963;
d) Y. F. Liang, X. Wang, C.Tang, T. Shen, J. Liu, N. Jiao, Chem.
Commun. 2016, 52, 1416-1419; e) Y. F. Liang, N. Jiao, Acc. Chem.
Res. 2017, 50, 1640-1653.
Acknowledgements ((optional))
This work was supported by the National Natural Science
Foundation of China (No. 21573196), the Fundamental
Research Funds of the Central Universities, and the National
High Technology Research and Development Program (863
Program) of China (Grant No. SS2015AA020601).
[13] a) T. Iwahama, S. Sakaguchi, Y. Nishiyama, Y. Ishii, Tetra. Lett. 1995, 38,
6923–6926; b) Y. Ishiii, T. Iwahama, S. Sakaguchi, K. Nakayama, Y.
Nishiyama, J. Org. Chem. 1996, 61, 4520–526; c) F. Minisci, C. Punta,
F. Recupero, F. Fontana, G. F. Pedulli, Chem. Commun. 2002, 7, 688–
689; d) D. P. Hruszkewycz, K. C. Miles, O. R. Thiel, S.S. Stahl, Chem
Sci. 2017, 8, 1282–1287; e) E. Gaster, S. Kozuch, D. Pappo, Angew
Chem, 2017, 129, 6006-6009; Angew. Chem. Int. Ed. 2017, 56, 5912–
5915.
Keywords: BV oxidation • NHPI • metal-free • molecular oxygen
• mechanistic study
[14] a) Y. Aoki, S. Sakaguchi, Y. Ishii, Adv. Synth. Catal. 2004, 346, 199–202;
b) Y. Hu, L. Chen, B. Li, Catal. Commun. 2016, 83, 82–87.
[1]
[2]
G. R. Krow, in The Baeyer-Villiger Oxidation of ketones and Aldehydes.
Organic Reactions. Wiley-VCH, Weinheim, 2004. 43, 251–798.
a) G. Strukul, Angew. Chem. 1998, 110, 1256–1267; Angew Chem. Int.
Ed. 1998, 37, 1198–1209; (b) M. Renz, Meunier, B. Eur. J. Org. Chem.
1999, 4, 737–750; (c) G.- J. ten Brink, I. W. C. E. Arends, R. A. Sheldon,
Chem. Rev. 2004, 104, 4105–4123; d) R. A. Michelin, P. Sgarbossa, A.
Scarso, G. Strukul, Coor. Chem. Rev. 2010, 254, 646–660; e) H. Leisch,
H. Morley, P. C. K. Lau, Chem. Rev. 2011, 111, 4165–4222.
[15] C. Einhorn, J. Einhorn, C. Marcadal. J-L. Pierre, Chem. Commun. 1997, 5,
447–448.
[16] T. Norihiro, Hiroaki, O. Hiroaki, M. Tsuyoshi, I. Akichika, SYNLETT 2009,
18, 3024–3026.
[17] a) C. Wang, G. Wang, J. Mao, Z. Yao, H. Li, Catal. Commun. 2010, 11,
758–762; b) P. Zhang, C. Wang, Z. Chen, H. Li, Catal. Sci. Technol.
2011, 1, 1133–1137; c) K. Chen, L. Jia, C. Wang, J. Yao, Z. Chen, H. Li,
ChemPhysChem, 2014, 15, 1673–1680; d) K. Chen, P. Zhang, Y.
Wang, H. Li, Green Chem. 2014, 16, 2344–2374.
[3]
A. Baeyer, V. Villiger, Eur. J. Inorg. Chem. 1899, 32, 3625–3633.
[4] a) S. L. Friess, N. Farnham J. Am. Chem. Soc. 1950, 72, 5518–5521; b) J.
Meinwald, E Frauenglass, J. Am Chem. Soc. 1960. 82, 5235–5239.
[5] Y. Imada, H. Iida, S. I. Murahashi, T. Naota, Angew. Chem. 2005, 117,
1732–1734; Angew. Chem. Int. Ed. 2005. 44, 1704–1706.
[18] M. Sankar, E. Nowicka, E. Carter, D. M. Murphy, D. W. Knight, D. Bethell,
G. J. Hutchings, Nat. Commun. 2014, 5, 3332–3337.
[19] a) R. Dao, X. Wang, K. Chen, C. Zhao, J. Yao, H. R. Li, Phys. Chem.
Chem. Phys. 2017, 33, 22309–22320; b) K. Chen, L. Jia, R. N. Dao, J.
Yao, C. M. Wang, Z. R. Chen, H. R. Li, ChemPhysChem 2013, 14,
179–184; c) Y. Sun, W. S. Zhang, X. B. Hu, H. R. Li, J. Phys. Chem. B
2010, 114, 4862–4869; d) R. Amorati, M. Lucarini, V, Mugnaini, G. F.
Pedulli, F. Minisci, F. Recupero, F. Fontana, P. Astolfi, L. Greci, J. Org.
Chem. 2003, 68, 1747–1754; e) S. Wertz, A. Studer, Green Chem.
2013, 15, 3116–3134; f) K. Chen, P. Zhang, Y. Wang, H. Li, Green
Chem. 2014, 16, 2344–2374.
[6] T. Yamada, K. Takahashi, K. Kato, T. Takai, S. Inoki, T. Mukaiyama, Chem.
Lett. 1991, 1, 641–644.
[7] a) A. Corma, L. T. Nemeth, M. Renz,S. Valencia, Nature, 2001, 412,
423–425; b) S. I. Murahashi, Y. Oda, T. Naota, Tetra. Lett. 1992, 49,
7557–7560; c) S. Chen, X. Zhou, Y. Li, R. Luo, H. Ji, Chem. Eng. J.
2014, 241, 138–144.
[8] a) C. Bolm, G. Schlingloff, K. Welckhardt, Tetra. Lett. 1993, 21, 3405–3408;
b) Yan Y., Dong L., Guo J., Huang M., Jiang Y., J. Macromol. Sci A
1997, 34, 1097–1104.
[20] R. K. Solly, S. W. Benson, J. Am. Chem. Soc. 1971, 93, 2127–2131.
[21] a) R. Criegee, Justus Liebigs Ann. Chem. 1948, 560, 127–135; b) R. M.
Goodman, Y. Kishi, J. Am. Chem. Soc. 1998, 120, 9392–9393.
[9] a) K. Kaneda, S. Ueno, T. Imanaka, J Mol. Cata. A: Chem. 1995, 102, 135–
138; b) T. Kawabata, Y. Ohishi, S. Itsuki, N. Fujisaki, T. Shishido, K.
Takaki, Q. Zhang, Y. Wang, K. Takehira, J. Mol. Catal. A: Chem. 2005,
236, 99–106; c) E.-Y. Jeong, M. B. Ansari, S.-E. Park, ACS catal. 2011,
1, 855–863; d) A. Sinhamahapatra, A. Sinha, S. K. Pahari, N. Sutradhar,
H. C. Bajaj, A. B. Panda, Catal. Sci. Technol. 2012, 2, 2375–2382.
This article is protected by copyright. All rights reserved.

 Wang, Lingyao
Wang, Lingyao
 Wang, Yongtao
Wang, Yongtao
 Du, Renfeng
Du, Renfeng
 Dao, Rina
Dao, Rina
 Yuan, Haoran
Yuan, Haoran
 Liang, Cheng
Liang, Cheng
 Yao, Jia
Yao, Jia
 Li, Haoran
Li, Haoran