Paper
Organic & Biomolecular Chemistry
8 (a) K. Steliou, P. Salama and J. Corriveau, J. Org. Chem.,
1985, 50, 4969; (b) D. Bhar and S. Chandrasekaran,
Tetrahedron, 1997, 53, 11835.
9 For papers on the importance of ring size in end-to-end
cyclisation reactions of larger rings, see: (a) G. Illuminati
and L. Mandolini, Acc. Chem. Res., 1981, 14, 95;
(b) J. Fastrez, J. Phys. Chem., 1989, 93, 2635; (c) J. C. Collins
X.-M. Liang, J.-J. Zhang and D.-Q. Wang, Phosphorus, Sulfur
Silicon Relat. Elem., 2014, 189, 1529.
14 Thiolactones are also involved in ring expansion cascade
reactions (known as ‘thia zip reactions’), although are inter-
mediates and not the final products in these reactions, see:
J. P. Tam, Y.-A. Lu and Q. Yu, J. Am. Chem. Soc., 1999, 121,
4316.
and K. James, MedChemComm, 2012, 3, 1489; 15 (a) Y. Nakashita and M. Hesse, Angew. Chem., 1981, 93,
(d) H. Kurouchi and T. Ohwada, J. Org. Chem., 2020, 85,
876.
1077; (b) S. Stanchev and M. Hesse, Helv. Chim. Acta, 1989,
72, 1052.
10 For general perspective on macrocycle synthesis, see: 16 (a) C. Kitsiou, J. J. Hindes, P. I’Anson, P. Jackson,
(a) E. Marsault and M. L. Peterson, J. Med. Chem., 2011, 54,
1961; (b) A. K. Yudin, Chem. Sci., 2015, 6, 30;
(c) K. T. Mortensen, T. J. Osberger, T. A. King, H. F. Sore
and D. R. Spring, Chem. Rev., 2019, 119, 10288; (d) Practical
Medicinal Chemistry with Macrocycles, ed. E. Marsault and
M. L. Peterson, Wiley, 2017; (e) S. D. Appavoo, S. Huh,
D. B. Diaz and A. K. Yudin, Chem. Rev., 2019, 119, 9724;
(f) I. V. Smolyar, A. K. Yudin and V. G. Nenajdenko, Chem.
Rev., 2019, 119, 10032; (g) I. Saridakis, D. Kaiser and
N. Maulide, ACS Cent. Sci., 2020, 6, 1869.
T. C. Wilson, E. K. Daly, H. R. Felstead, P. Hearnshaw and
W. P. Unsworth, Angew. Chem., Int. Ed., 2015, 54, 15794;
(b) L. G. Baud, M. A. Manning, H. L. Arkless, T. C. Stephens
and W. P. Unsworth, Chem. – Eur. J., 2017, 23, 2225;
(c) T. C. Stephens, M. Lodi, A. Steer, Y. Lin, M. Gill and
W. P. Unsworth, Chem.
– Eur. J., 2017, 23, 13314;
(d) T. C. Stephens, A. Lawer, T. French and W. P. Unsworth,
Chem. – Eur. J., 2018, 24, 13947 For the application of SuRE
reactions in the work of another group, see: (e) C. Zhao,
Z. Ye, Z.-X. Ma, S. A. Wildman, S. A. Blaszczyk, L. Hu,
I. A. Guizei and W. Tang, Nat. Commun., 2019, 10, 4015,
DOI: 10.1038/s41467-019-11976-2.
11 For reviews of ring expansion chemistry, see: (a) M. Hesse,
in Ring Enlargement in Organic Chemistry, Wiley-VCH,
Weinheim, 1991; (b) W. P. Unsworth and J. R. Donald, 17 This method was introduced by our group in ref. 16d, and
Chem. – Eur. J., 2017, 23, 8780; (c) K. Prantz and J. Mulzer,
Chem. Rev., 2010, 110, 3741; (d) T. C. Stephens and
W. P. Unsworth, Synlett, 2020, 31, 133; (e) A. K. Clarke and
W. P. Unsworth, Chem. Sci., 2020, 11, 2876.
more fully explored and benchmarked against other com-
putational methods in: A. Lawer, R. G. Epton,
T. C. Stephens, K. Y. Palate, M. Lodi, E. Marotte,
K. J. Lamb, J. K. Sangha, J. Lynam and W. P. Unsworth,
Chem. – Eur. J., 2020, 26, 12674 See ESI for full details of
the new computational chemistry results reported in this
paper.†
12 (a) L. Li, Z.-L. Li, F.-L. Wang, Z. Guo, Y.-F. Cheng, N. Wang,
X.-W. Dong, C. Fang, J. Liu, C. Hou, B. Tan and X.-Y. Liu,
Nat. Commun., 2016, 7, 13852; (b) J. E. Hall, J. V. Matlock,
J. W. Ward and J. Clayden, Angew. Chem., Int. Ed., 2016, 55, 18 For computational chemistry studies on related ring expan-
11153; (c) Z.-L. Li, X.-H. Li, N. Wang, N.-Y. Yang and sion processes, see ref. 12d and i.
X.-Y. Liu, Angew. Chem., Int. Ed., 2016, 55, 15100; 19 A guideline established in our earlier work (ref. 17) is that
(d) R. Mendoza-Sanchez, V. B. Corless, Q. N. N. Nguyen,
M. Bergeron-Brlek, J. Frost, S. Adachi, D. J. Tantillo and
A. K. Yudin, Chem. – Eur. J., 2017, 23, 13319; (e) R. Costil,
the ring expanded isomer should be the most stable by
>3kcal mol−1 for ring expansion to be the predicted
outcome.
Q. Lefebvre and J. Clayden, Angew. Chem., Int. Ed., 2017, 56, 20 P. Majer, P. F. Jackson, G. Delahanty, B. S. Grella, Y.-S. Ko,
14602; (f) D. R. Loya, A. Jean, M. Cormier, C. Fressigné,
S. Nejrotti, J. Blanchet, J. Maddaluno and M. De Paolis,
Chem. – Eur. J., 2018, 24, 2080; (g) N. Wang, Q.-S. Gu,
Z.-L. Li, Z. Li, Y.-L. Guo, Z. Guo and X.-Y. Liu, Angew.
Chem., Int. Ed., 2018, 57, 14225; (h) Y. Zhou, Y.-L. Wei,
W. Li, Q. Liu, K. M. Maclin, J. Poláková, K. A. Shaffer,
D. Stoermer, D. Vitharana, E. Y. Wang, A. Zakrzewski,
C. Rojas, B. S. Slusher, K. M. Wozniak, E. Burak,
T. Limsakun and T. Tsukamoto, J. Med. Chem., 2003, 46,
1989.
J. Rodriguez and Y. Coquerel, Angew. Chem., Int. Ed., 2019, 21 Another important consideration when using this approach
58, 456; (i) E. Reutskaya, A. Osipyan, A. Sapegin,
A. S. Novikov and M. Krasavin, J. Org. Chem., 2019, 84,
1693; ( j) A. Lawer, J. A. Rossi-Ashton, T. C. Stephens,
B. J. Challis, R. G. Epton, J. M. Lynam and W. P. Unsworth,
Angew. Chem., Int. Ed., 2019, 58, 13942; (k) S. Grintsevich,
A. Sapegin, E. Reutskaya, S. Peintner, M. Erdélyi and
M. Krasavin, Eur. J. Org. Chem., 2020, 5664.
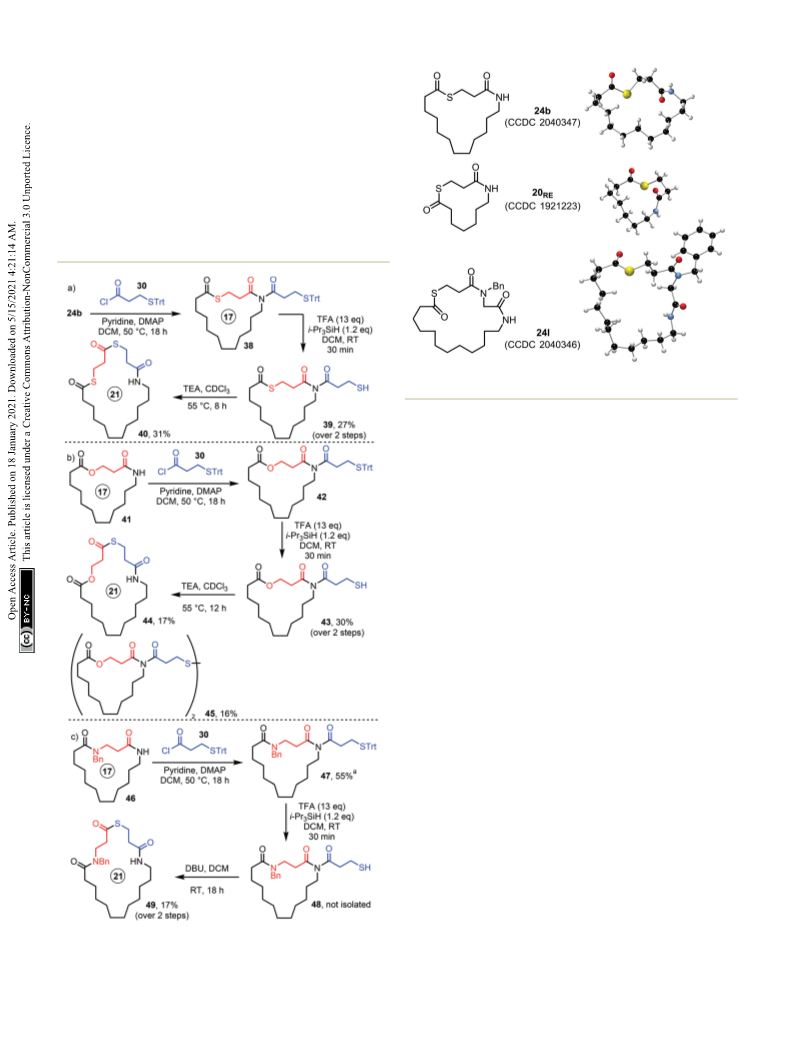
is that in our hands, the N-acylation step (i.e. 21b → 31b
was somewhat capricious, which we think is a consequence
of the requirement to form an acid chloride in the presence
of an acid labile protecting group. Efficient N-acylation can
still be achieved by forming and reacting acid chloride 30
quickly, but compared with other SuRE variants, this is an
additional complication.
13 (a) H. C. De Araújo and J. R. Mahajan, Synthesis, 1978, 228; 22 (a) M. Ruiz-Gayo, F. Albericio, E. Pedroso and E. J. Giralt,
(b) J. R. Mahajan and H. C. De Araújo, Synthesis, 1980, 64;
(c) R. Malherbe, G. Rist and D. Bellus, J. Org. Chem., 1982,
48, 860; (d) E. Vedejs, J. M. Dolphin and H. Mastalerz,
J. Am. Chem. Soc., 1983, 105, 127; (e) C. Meng, J.-J. Li,
Chem. Soc., Chem. Commun., 1986, 1501; (b) E. J. Corey,
D. Y. Gin and R. S. Kania, J. Am. Chem. Soc., 1996, 118,
9202; (c) B. Ponsati, E. Giralt and D. Andreu, Tetrahedron,
1990, 46, 8255.
1410 | Org. Biomol. Chem., 2021, 19, 1404–1411
This journal is © The Royal Society of Chemistry 2021

 Palate, Kleopas Y.
Palate, Kleopas Y.
 Epton, Ryan G.
Epton, Ryan G.
 Whitwood, Adrian C.
Whitwood, Adrian C.
 Lynam, Jason M.
Lynam, Jason M.
 Unsworth, William P.
Unsworth, William P.