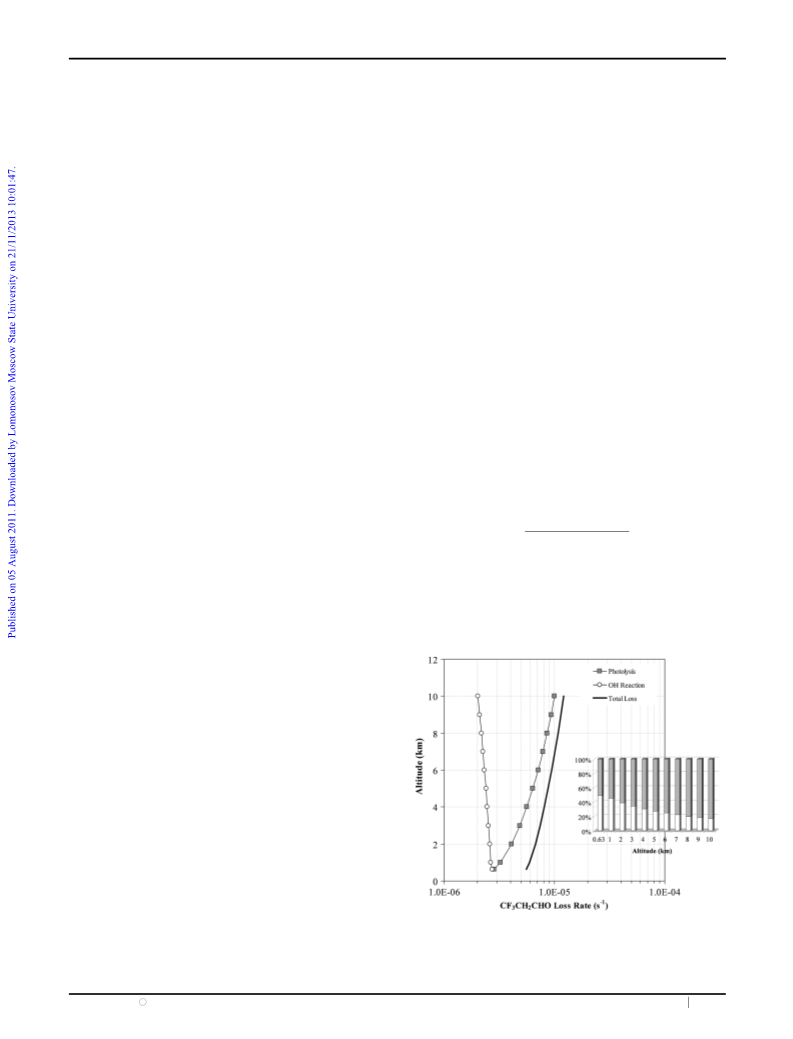
of CF3CH2CHO compared with its removal by OH radicals.
In Fig. 6, a comparison of the first-order rate for the
homogeneous removal and the UV photolysis process of
CF3CH2CHO is shown together with the overall rate loss,
2 E. Jimenez, M. Antinolo, B. Ballesteros, E. Martınez and
´ ´
J. Albaladejo, ChemPhysChem, 2010, 11, 4079–4087.
3 T. Kelly, V. Bossoutrot, I. Magneron, K. Wirtz, J. Treacy,
A. Mellouki, H. Sidebottom and G. Le Bras, J. Phys. Chem. A,
2005, 109, 347–355.
4 M. D. Hurley, J. A. Misner, J. C. Ball, T. J. Wallington,
D. A. Ellis, J. W. Martin, S. A. Mabury and M. P. Sulbaek
Andersen, J. Phys. Chem. A, 2005, 109, 9816–9826.
5 M. D. Hurley, T. J. Wallington, M. P. Sulbaek Andersen,
D. A. Ellis, J. W. Martin and S. A. Mabury, J. Phys. Chem. A,
2004, 108, 1973–1979.
6 L. Chen, N. Takenaka, H. Bandow and Y. Maeda, Atmos.
Environ., 2003, 37, 4817–4822.
7 R. Atkinson, D. L. Baulch, R. A. Cox, J. N. Crowley,
R. F. Hampson, R. G. Hynes, M. E. Jenkin, M. J. Rossi, J. Troe
and T. J. Wallington, Atmos. Chem. Phys., 2008, 8, 4141–4496.
kloss (=
J + kOH[OH]avg). The overall lifetime of
CF3CH2CHO is estimated to be B2 days at the ground level
and 1 day at higher altitudes in the troposphere. The upper limit
for the contribution of the photolysis of CF3CH2CHO to kloss
could range from 50% at the ground level up to 80% at 10 km.
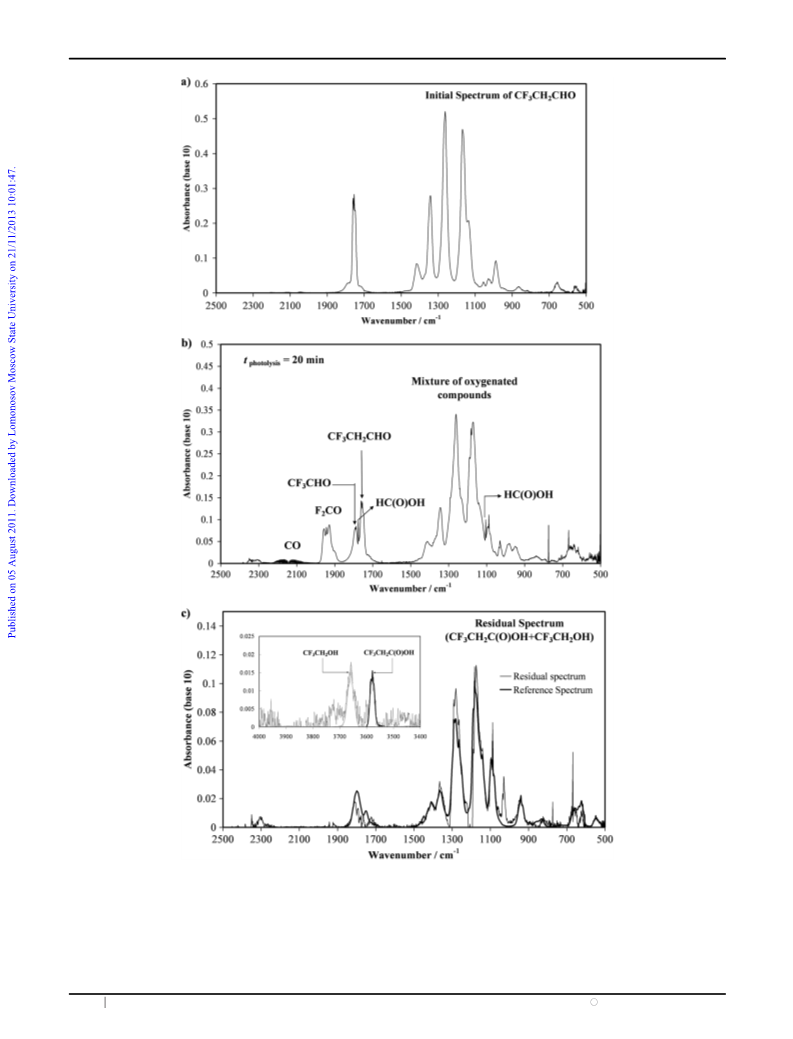
According to this study, in unpolluted atmospheres the
expected products of the photodegradation of CF3CH2CHO
can be CO, F2CO, HC(O)OH, CF3CHO, CF3CH2C(O)OH,
and CF3CH2OH. However, the volume mixing ratio of
CF3CH2CHO used in this work is several orders of magnitude
higher than that expected in the atmosphere and, therefore, at
the expected levels of CF3CH2CHO in the atmosphere, none of
photodegradation products pose any environmental threat.
8 M. Antinolo, E. Jimenez, A. Notario, E. Martınez and
´ ´
J. Albaladejo, Atmos. Chem. Phys., 2010, 10, 1911–1922.
9 S. R. Sellevag, T. Kelly, H. Sidebottom and C. J. Nielsen, Phys.
Chem. Chem. Phys., 2004, 6, 1243–1252.
10 M. S. Chiappero, F. E. Malanca, G. A. Arguello,
¨
S. T. Wooldridge, M. D. Hurley, J. C. Ball, T. J. Wallington,
R. L. Waterland and R. C. Buck, J. Phys. Chem. A, 2006, 110,
11944–11953.
Conclusions
11 E. Jime
Chem. Phys., 2007, 7, 1565–1574.
12 B. Lanza, E. Jimenez, B. Ballesteros and J. Albaladejo, Chem.
Phys. Lett., 2008, 454, 184–189.
13 E. Jimenez, B. Lanza, M. Antinolo and J. Albaladejo, Environ. Sci.
nez, B. Lanza, E. Martınez and J. Albaladejo, Atmos.
´ ´
In this work, we report (i) the temperature dependence of
the UV absorption cross sections of CF3CH2CHO between
269 and 323 K; (ii) the first study on the pressure dependence
of Fl=308nm in CF3CH2CHO/air/cyclohexane mixtures,
(iii) the quantification of the final products (CO, HC(O)OH,
CF3CHO, F2CO, and CF3CH2OH) by FTIR spectroscopy,
and (iv) a proposed mechanism that justifies the observed final
products and their temporal evolution.
´
´
Technol., 2009, 43, 1831–1837.
14 Y. Hashikawa, M. Kawasaki, R. L. Waterland, M. D. Hurley,
J. C. Ball, T. J. Wallington, M. P. Sulbaek Andersen and
O. J. Nielsen, J. Fluorine Chem., 2004, 125, 1925–1932.
15 R. Atkinson, D. L. Baulch, R. A. Cox, J. N. Crowley,
R. F. Hampson, R. G. Hynes, M. E. Jenkin, M. J. Rossi and
J. Troe, Atmos. Chem. Phys., 2006, 6, 3625–4055.
16 I. Barnes. Personal communication.
17 H. Somnitz, M. Fida, T. Ufer and R. Zellner, Phys. Chem. Chem.
Phys., 2005, 7, 3342–3352.
18 H. Somnitz, T. Ufer and R. Zellner, Phys. Chem. Chem. Phys.,
2009, 11, 8522–8531.
19 A. Tomas, F. Caralp and R. Lesclaux, Z. Phys. Chem., 2000, 214,
1349–1365.
20 M. M. Maricq, J. J. Szente, G. A. Khitrov, T. S. Dibble and
J. S. Francisco, J. Phys. Chem., 1995, 99, 11875–11882.
21 C. G. Ci, H. B. Yu, S. Q. Wan, J. Y. Liu and C. C. Sun, Bull.
Korean Chem. Soc., 2011, 32, 1187–1194.
22 S. P. Sander, J. Abbatt, J. R. Barker, J. B. Burkholder,
R. R. Friedl, D. M. Golden, R. E. Huie, C. E. Kolb,
M. J. Kurylo, G. K. Moortgat, V. L. Orkin and P. H. Wine,
Chemical Kinetics and Photochemical Data for Use in Atmospheric
Studies, Evaluation No. 17, JPL Publication 10-6, Jet Propulsion
Laboratory, Pasadena, 2011.
Knowing the photolysis quantum yield at tropospheric
pressures is of great importance in order to better determine
the photolysis lifetime of this fluorinated aldehyde. On the
other hand, the wavelength dependence of Fl in the actinic
region (l Z 290 nm) is also needed to better quantify any
photolysis process in the troposphere. In this work, the
photolysis quantum yield of CF3CH2CHO at 308 nm has
been reported to decrease with total pressure. If a similar
behaviour is considered for Fl at other wavelengths, the photo-
lysis rate J at higher altitudes in the troposphere would be more
important than reported, since Fl and Fl increases with altitude.
Acknowledgements
23 T. L. Osif and J. Heicklen, J. Phys. Chem., 1976, 80, 1526–1531.
24 J. E. Carruthers and R. G. W. Norrish, J. Chem. Soc., 1936,
1036–1042.
The authors would like to thank the Spanish Ministerio de
Ciencia e Innovacio
´
n (MICINN) (CGL2007-61835/CLI and
a de Educacion y Ciencia
25 J. W. Bozelli and A. M. Dean, J. Phys. Chem., 1993, 97, 4427–4441.
26 M. Martı
´
CGL2010-19066) and the Consejerı
´
´
´
nez-Avila, J. Peiro
´
I. Nebot-Gil, Chem. Phys. Lett., 2003, 370, 313–318.
-Garcı
´
a, V. M. Ramı
´
´
rez-Ramırez and
de la Junta de Comunidades de Castilla-La Mancha (PEII11-
0279-8538) for supporting this Project. M. Antinolo wishes to
thank MICINN for providing her a grant (AP2007-02706). We
would also like to thank Prof. Ian Barnes for providing a
reference spectrum of F2CO and Dr Christa Fittschen and
Dr Mary K. Gilles for helpful discussions.
27 T. J. Dillon and J. N. Crowley, Atmos. Chem. Phys., 2008, 8,
4877–4889.
29 M. A. Blitz, D. E. Heard and M. J. Pilling, Chem. Phys. Lett.,
2002, 365, 374–379.
30 S. A. Carr, M. T. Baeza-Romero, M. A. Blitz, M. J. Pilling,
D. E. Heard and P. W. Seakins, Chem. Phys. Lett., 2007, 445, 108–112.
31 M. T. Baeza Romero, M. A. Blitz, D. E. Heard, M. J. Pilling, B. Price,
P. W. Seakins and L. Wang, Faraday Discuss., 2005, 130, 73–88.
32 K. L. Demerjian, J. A. Kerr and J. G. Calvert, Adv. Environ. Sci.
Technol., 1974, 4, 1–262.
33 S. Madronich and S. Flocke, The role of solar radiation in atmo-
spheric chemistry, in Handbook of Environmental Chemistry, ed.
P. Boule, Springer-Verlag, Heidelberg, 1999, pp. 1–26.
34 M. Krol, P. J. van Leeuwen and J. Lelieveld, J. Geophys. Res.,
[Atmos.], 1998, 103, 10697–10711.
References
1 J. S. Daniel and G. J. M. Velders (Lead Authors). Halocarbon
Scenarios, Ozone Depletion Potentials, and Global Warming
Potentials, in Scientific Assessment of Ozone Depletion: 2006,
Global Ozone Research and Monitoring Project-Report No. 50,
World Meteorological Organization, Geneva, Switzerland,
2007, ch. 8.
c
15946 Phys. Chem. Chem. Phys., 2011, 13, 15936–15946
This journal is the Owner Societies 2011

 Antinolo, Maria
Antinolo, Maria
 Jimenez, Elena
Jimenez, Elena
 Albaladejo, Jose
Albaladejo, Jose