10.1002/cctc.201900304
ChemCatChem
The adsorption energy of the ammonia is calculated by the following
equitation:
∆E = −(Eꢀꢁꢀꢂꢃ − Eꢄꢅꢀꢂꢃ(ꢆꢆꢆ) − Eꢇꢈꢉ) (1)
Neubert, H. Neumann, M. Beller, Angew. Chem. Int. Ed. 2010, 49,
8126-8129; d) R. Kawahara, K. Fujita, R. Yamaguchi, J. Am. Chem.
Soc. 2010, 132, 15108-15111; e) J. Gallardo-Donaire, M. Ernst, O.
Trapp, T. Schaub, Adv. Synth. Catal. 2016, 358, 358-363.
a) M. V. Klyuev, M. L. Khidekel, Russ. Chem. Rev. 1980, 49, 28-53; b)
A. Fischer, T. Mallat, A. Baiker, Catal. Today 1997, 37, 167-189.
a) T. Takanashi, M. Tamura, Y. Nakagawa, K. Tomishige, RSC Adv.
2014, 4, 28664; b) M. Ousmane, G. Perrussel, Z. Yan, J. M. Clacens, F.
De Campo, M. Pera-Titus, J. Catal. 2014, 309, 439-452; c) X. Cui, X.
Dai, Y. Deng, F. Shi, Chem. Eur. J. 2013, 19, 3665-3675; d) R.
Pfützenreuter, M. Rose, ChemCatChem 2016, 8, 251-255; e) J. Krupka,
L. Dluhoš, L. Mrózek, Chem. Eng. Technol. 2017, 40, 870-877; f) F. Q.
Qi, L. Hu, S. L. Lu, X. Q. Cao, H. W. Gu, Chem. Commun. 2012, 48,
9631-9633.
[5]
[6]
The adsorption energy of the hydrogen is calculated by the following
equitation:
∆E = −(Eꢀꢁꢀꢂꢃ − Eꢄꢅꢀꢂꢃ(ꢆꢆꢆ) − 0.5Eꢈꢊ) (2)
Etotal is the energy of the ammonia or hydrogen on the surface, Emetal(111)
is the energy of the bare metal without adsorption of hydrogen or
ammonia, ENH3and EH2are the energy of ammonia and hydrogen in
gaseous sate, respectively.
[7]
a) Q. Yang, Q. Wang, Z. Yu, Chem. Soc. Rev. 2015, 44, 2305-2329; b)
K. Shimizu, K. Kon, W. Onodera, H. Yamazaki, J. N. Kondo, ACS Catal.
2013, 3, 112-117.
Models
DFT calculations were carried out on Rh, Ru, Pt, Pd, Co and Ni metal
surfaces. A hcp (0001) surface was applied to model the flat surface of
Ru, while for other metals, fcc (111) surface was applied because these
structures are mostly exposed and therefore are the most representative
surfaces.[21] For all the calculations, a 2×2 supercell model including five
atomic layers were employed. A 15 Å vacuum layer between the slabs
was used to prevent spurious interactions generated by periodic
boundary conditions. The three bottom metal layers were fixed and the
top two layers were allowed to relax.
[8]
[9]
A. Fischer, M. Maciejewski, T. Bürgi, T. Mallat, A. Baiker, J. Catal. 1999,
183, 373-383.
a) Y. X. Liu, K. Zhou, H. M. Shu, H. Y. Liu, J. T. Lou, D. C. Guo, Z. J.
Wei, X. N. Li, Catal. Sci. Technol. 2017, 7, 4129-4135; b) D. Ruiz, A.
Aho, T. Saloranta, K. Eränen, J. Wärnå, R. Leino, D. Y. Murzin, Chem.
Eng. J. 2016, 307, 739-749.
[10] S. Nishimura, K. Mizuhori, K. Ebitani, Res. Chem. Intermed. 2015, 42,
19-30.
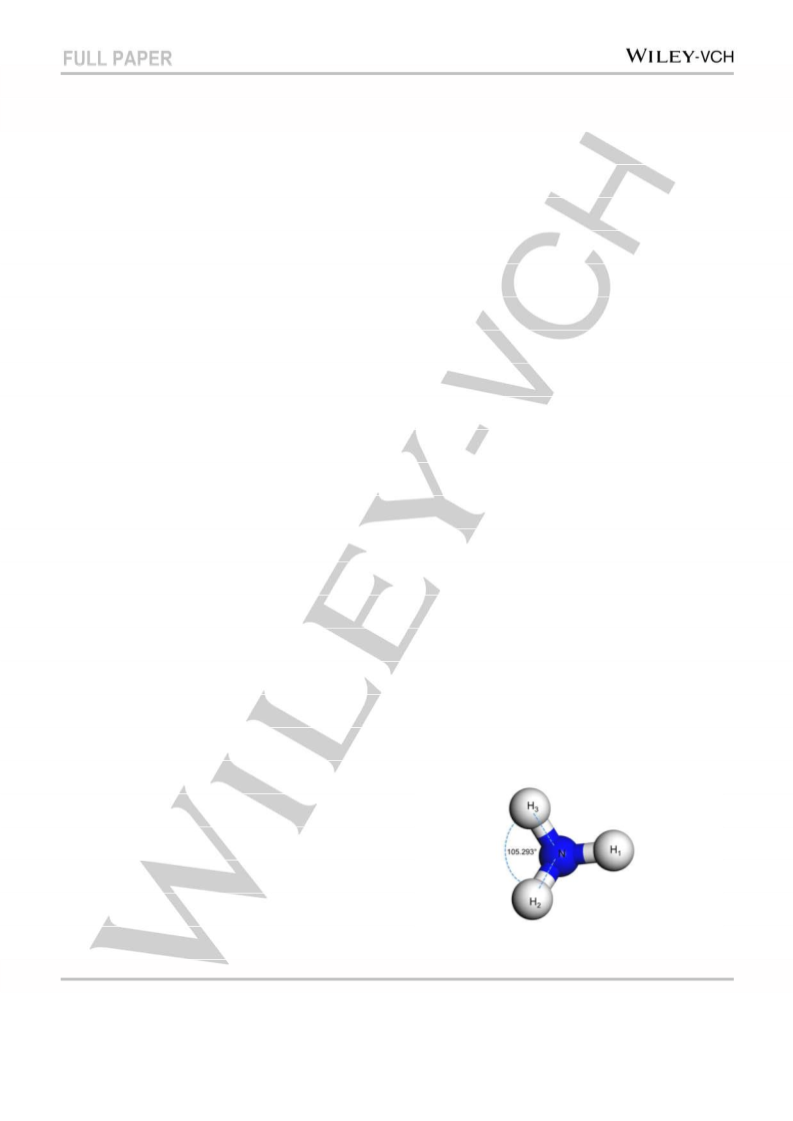
The lattice parameter of the optimized cell of Pt, Pd, and Ni were 0.3924
nm, 0.3891 nm and 0.3524 nm, respectively, which agree well with the
experimental values of 0.3923 nm, 0.3890 nm and 0.3524 nm,
respectively.[30] The free ammonia and hydrogen molecule were
optimized using the same methods. The equilibrium bond lengths of N-H
and H-H were 0.1022 nm and 0.0741 nm respectively, while the bonding
angle of H-N-H is 105.3° (Figure 5.) which were close to the
experimental value (0.1012 nm, 0.0741 nm, 106.7°).[30] These results
indicate that our model is applicable and reliable.
[11] M. Chatterjee, T. Ishizaka, H. Kawanami, Green Chem. 2016, 18, 487-
496.
[12] D. Pingen, O. Diebolt, D. Vogt, ChemCatChem 2013, 5, 2905-2912.
[13] T. Komanoya, T. Kinemura, Y. Kita, K. Kamata, M. Hara, J. Am. Chem.
Soc. 2017, 139, 11493-11499.
[14] X. Wang, W. Chen, Z. Li, X. Zeng, X. Tang, Y. Sun, T. Lei, L. Lin, J.
Energy Chem. 2018, 27, 209-214.
[15] X. F. Li, J. P. Ma, X. Q. Jia, F. Xia, Y. Z. Huang, Y. M. Xu, J. Xu, Acs
Sus. Chem. Eng. 2018, 6, 8048-8054.
[16] J. H. Cho, S. H. An, T.-S. Chang, C.-H. Shin, Catal. Lett. 2016, 146,
811-819.
[17] N.-T. Le, A. Byun, Y. Han, K.-I. Lee, H. Kim, Green Sustainable Chem.
2015, 05, 115-127.
Acknowledgements
[18] D. Pingen, J. B. Schwaderer, J. Walter, J. Wen, G. Murray, D. Vogt, S.
Mecking, ChemCatChem 2018, 10, 3027-3033.
This research was supported by the National Natural Science
Foundation of China (21878269 and 21476211) and the Natural
Science Foundation of Zhejiang province (LY16B060004 and
LY18B060016).
[19] D. Pingen, C. Müller, D. Vogt, Angew. Chem. Int. Ed. 2010, 49, 8130–
8133.
[20] T. Takanashi, Y. Nakagawa, K. Tomishige, Chem. Lett. 2014, 43, 822-
824.
[21] a) G. W. Watson, R. P. K. Wells, D. J. Willock, G. J. Hutchings, J. Phys.
Chem. B 2001, 105, 4889-4894; b) P. Ferrin, S. Kandoi, A. U. Nilekar,
M. Mavrikakis, Surf. Sci. 2012, 606, 679-689.
Keywords: Biomass • Furfuryl alcohol • 5-Hydroxymethylfurfural
• Raney Ni • Reductive amination
[22] Z. P. Liu, P. Hu, M. H. Lee, J. Chem. Phys. 2003, 119, 6282-6289.
[23] a) J.-H. Park, E. Hong, S. H. An, D.-H. Lim, C.-H. Shin, Korean J.
Chem. Eng. 2017, 29, 1-9; b) Y. X. Liu, K. Zhou, M. Lu, L. C. Wang, Z.
J. Wei, X. N. Li, Ind. Eng. Chem. Res. 2015, 54, 9124-9132.
[24] a) B. Delley, J. Phys. Chem. 1996, 100, 6107-6110; b) B. Delley, J.
Chem. Phys. 2000, 113, 7756-7764; c) B. Delley, Phys. Rev. B 2002,
66, 155125.
[1]
[2]
[3]
a) K. S. Hayes, Appl. Catal. A: Gen. 2001, 221, 187-195; b) K.
Yamaguchi, J. He, T. Oishi, N. Mizuno, Chem.Eur. J. 2010, 16, 7199-
7207.
a) Z. J. Wei, Y. Li, D. Thushara, Y. X. Liu, Q. L. Ren, J. Taiwan Ins.
Chem. Eng. 2011, 42, 363-370; b) Z. J. Wei, Y. X. Liu, T. Dilantha, Q. L.
Ren, Green Chem. 2012, 14, 1220-1226.
[25] P. Hu, D. A. King, S. Crampin, M. H. Lee, M. C. Payne, Chem. Phys.
Lett. 1994, 230, 501-506.
a) G. Liang, A. Wang, L. Li, G. Xu, N. Yan, T. Zhang, Angew. Chem. Int.
Ed. 2017, 56, 3050-3054; b) M. Pera-Titus, F. Shi, ChemSusChem
2014, 7, 720-722; c) Y. Li, Z. J. Wei, C. J. Chen, Y. X. Liu, Prog.Chem.
(CHN) 2010, 22, 1603-1609; d) Z. J. Wei, J. T. Lou, Z. B. Li, Y. X. Liu,
Catal. Sci. Technol. 2016, 6, 6217-6225; e) Z. J. Wei, J. T. Lou, C. M.
Su, D. C. Guo, Y. X. liu, S. G. Deng, ChemSusChem 2017, 10, 1720-
1732.
[26] J. P. Perdew, K. Burke, M. Ernzerhof, Phys. Rev. Lett. 1996, 77, 3865-
3868.
[27] Y. Inada, H. Orita, J. Comput. Chem. 2008, 29, 225-232.
[28] H. J. Monkhorst, J. D. Pack, Phys. Rev. B 1976, 13, 5188-5192.
[29] a) S. Chen, S. Sun, B. Lian, Y. Ma, Y. Yan, S. Hu, Surf. Sci. 2014, 620,
51-58; b) L. M. Cam, L. Van Khu, N. N. Ha, J. Mol. Model. 2013, 19,
4395-4402.
[4]
a) C. Gunanathan, D. Milstein, Angew. Chem., Int. Ed. 2008, 47, 8661-
8664; b) O. Saidi, A. J. Blacker, M. M. Farah, S. P. Marsden, J. M.
Williams, Chem. Commun. 2010, 46, 1541-1543; c) S. Imm, S. Bahn, L.
[30] D. R. Lide, Handbook of Chemistry and Physics, CRC Press, London,
2001.
This article is protected by copyright. All rights reserved.

 Zhou, Kuo
Zhou, Kuo
 Liu, Haiyan
Liu, Haiyan
 Shu, Huimin
Shu, Huimin
 Xiao, Shuwen
Xiao, Shuwen
 Guo, Dechao
Guo, Dechao
 Liu, Yingxin
Liu, Yingxin
 Wei, Zuojun
Wei, Zuojun
 Li, Xiaonian
Li, Xiaonian